Advanced Manufacturing of Cyclopenta[d]pyrimidin-4-yl Piperazine Compounds for AKT Inhibitors
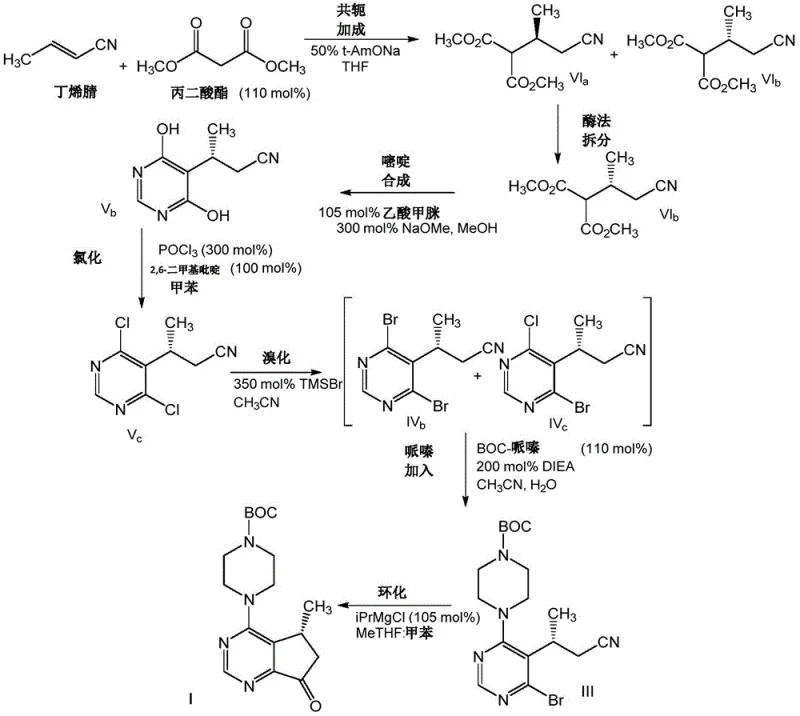
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for critical oncology drug intermediates, particularly for AKT inhibitors like Ipatasertib. Patent CN107001287B discloses a groundbreaking methodology for the preparation of (cyclopenta[d]pyrimidin-4-yl)piperazine compounds, specifically focusing on the efficient synthesis of (R)-4-(5-methyl-7-oxo-6,7-dihydro-5H-cyclopenta[d]pyrimidin-4-yl)piperazine and its N-protected derivatives. This technical disclosure represents a significant leap forward in process chemistry by replacing traditional iodination protocols with a more cost-effective and environmentally friendly bromination strategy. The innovation lies in the strategic use of nitrile-substituted intermediates which withstand harsher reaction conditions that would typically degrade ester-based analogues. By integrating enzymatic resolution with telescoped halogenation steps, this process addresses key pain points in impurity control and production throughput. For R&D directors and procurement specialists, understanding these mechanistic advantages is crucial for securing a reliable pharmaceutical intermediates supplier capable of delivering high-purity materials at scale.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic pathways for constructing the cyclopenta[d]pyrimidine core often rely heavily on iodination reactions to facilitate subsequent cyclization steps. These conventional methods typically involve the use of iodo-ester or iodo-acid substituted compounds, which present significant challenges in terms of reagent cost and environmental impact. The reliance on iodine-containing reagents necessitates strict handling protocols and generates hazardous waste streams that complicate downstream processing and disposal. Furthermore, ester-substituted intermediates are chemically fragile under the rigorous conditions required for effective halogen exchange, often leading to unwanted ester cleavage and concomitant lactone formation. This susceptibility to degradation limits the choice of brominating agents and forces reactions to proceed at lower temperatures, thereby slowing down reaction kinetics and extending the overall production cycle. Consequently, manufacturers face reduced yields and increased operational complexity when attempting to scale these legacy processes for commercial demand.
The Novel Approach
The novel approach detailed in the patent fundamentally shifts the paradigm by utilizing nitrile-substituted compounds as the key intermediates for ring construction. By replacing the labile ester group with a robust nitrile functionality, the synthesis gains access to a much broader range of reaction conditions and reagents. This structural modification allows for the use of stronger and more reactive brominating agents, such as trimethylsilyl bromide, at elevated temperatures ranging from 70°C to 75°C without compromising the integrity of the molecule. The process further innovates by implementing a telescoped sequence where chlorination, bromination, and piperazine addition are performed consecutively without isolating the intermediate reaction products. This end-to-end methodology drastically simplifies the unit operations required, eliminating multiple filtration and drying steps that traditionally bottleneck production capacity. The result is a streamlined workflow that not only enhances safety by minimizing operator exposure to intermediates but also significantly improves the overall mass balance and throughput of the manufacturing line.
Mechanistic Insights into Grignard-Mediated Cyclization and Enzymatic Resolution
A critical component of this advanced synthesis is the stereoselective formation of the chiral center, achieved through a highly specific enzymatic resolution step. The process begins with a conjugate addition of crotononitrile to a malonate ester, generating an isomeric mixture that is subsequently subjected to enzymatic hydrolysis. Unlike traditional chemical resolution methods that may require multiple recrystallizations, this biological approach utilizes nitrilase enzymes to selectively hydrolyze the unwanted isomer, leaving the desired (R)-nitrile intact with high enantiomeric excess. The patent highlights that operating at an elevated pH of approximately 9.2 can double the reaction rate compared to neutral conditions, demonstrating a sophisticated optimization of biocatalytic parameters. This precision ensures that the downstream cyclization steps proceed with a substrate of defined stereochemistry, which is paramount for the biological activity of the final AKT inhibitor. The ability to perform this resolution directly on the reaction mixture without prior isolation of the isomeric blend further underscores the efficiency of the design.
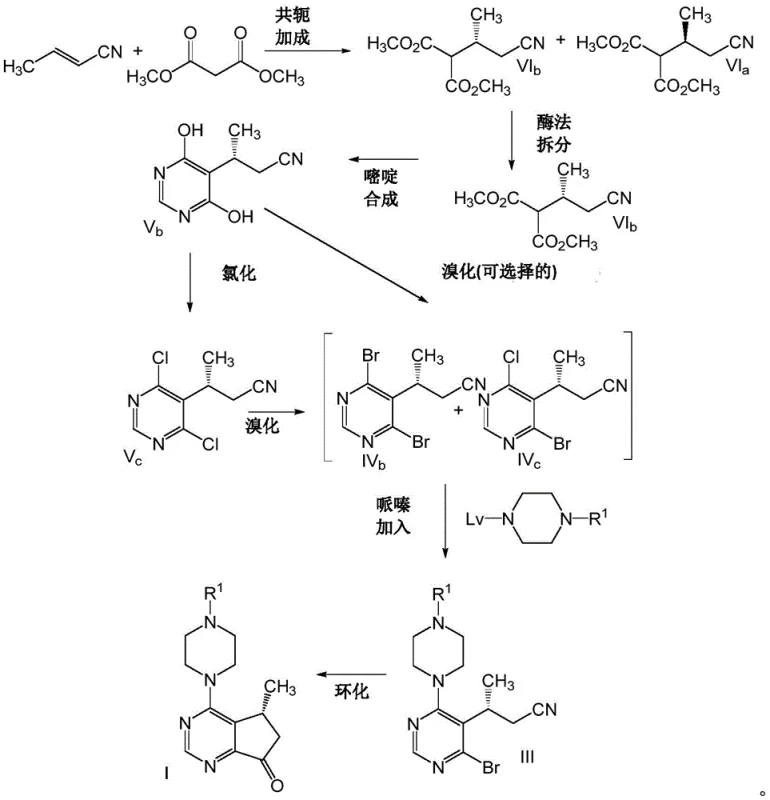
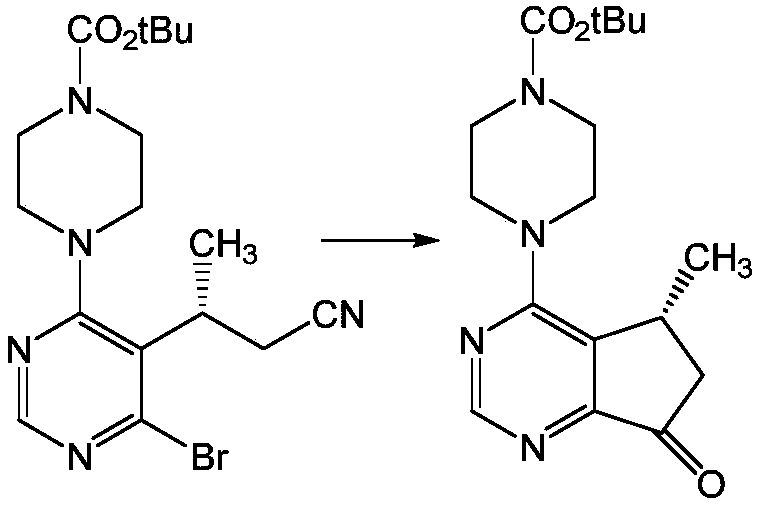
The final ring closure is accomplished via a Grignard-mediated cyclization, a transformation that requires precise control over organometallic reactivity. The method employs organomagnesium reagents, such as isopropylmagnesium chloride, to effect an intramolecular attack on the nitrile group, forming the five-membered cyclopentyl ring fused to the pyrimidine core. This step is particularly sensitive to moisture and temperature, necessitating anhydrous conditions and controlled addition rates to prevent side reactions. The use of nitrile-substituted precursors is again advantageous here, as they facilitate the cyclization more effectively than their chloro-nitrile counterparts, which have been shown to yield significant amounts of dechlorinated by-products. The reaction is quenched carefully to hydrolyze the resulting imine intermediate to the final ketone product, achieving conversion rates exceeding 90% in optimized embodiments. This mechanistic robustness ensures that the final API intermediate meets stringent purity specifications required for clinical and commercial applications.
How to Synthesize (R)-4-(5-methyl-7-oxo-6,7-dihydro-5H-cyclopenta[d]pyrimidin-4-yl)piperazine Efficiently
Implementing this synthesis requires a coordinated sequence of biocatalytic and organometallic steps designed to maximize yield while minimizing waste. The process initiates with the preparation of the chiral nitrile building block, followed by a telescoped halogenation sequence that builds the functionalized pyrimidine core. The final stage involves the critical Grignard cyclization which closes the ring system to form the target ketone. Operators must adhere to strict anhydrous protocols during the metalation phase and maintain precise temperature controls during the enzymatic resolution to ensure optimal enantioselectivity. The detailed standardized synthesis steps see the guide below.
- Perform conjugate addition of crotononitrile with dimethyl malonate followed by enzymatic resolution using nitrilase to isolate the (R)-isomer.
- Execute a telescoped chlorination and bromination sequence on the pyrimidine intermediate without isolating the dichloro species.
- Conduct nucleophilic substitution with N-protected piperazine followed by Grignard cyclization using iPrMgCl to form the final cyclopentyl ring.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers substantial strategic benefits beyond mere technical elegance. The elimination of iodine reagents and the reduction in isolation steps translate directly into a more resilient and cost-efficient supply chain model. By simplifying the manufacturing workflow, producers can mitigate risks associated with complex multi-step syntheses and reduce the dependency on scarce or volatile raw materials. This process stability is essential for maintaining consistent supply volumes in the face of fluctuating market demands for oncology therapeutics.
- Cost Reduction in Manufacturing: The removal of expensive iodination reagents and the associated waste treatment costs leads to a significant decrease in the overall cost of goods sold. Furthermore, the telescoped nature of the halogenation steps reduces solvent consumption and energy usage by eliminating intermediate drying and heating cycles. This lean manufacturing approach allows for competitive pricing structures without compromising on the quality of the high-purity pharmaceutical intermediates delivered to clients.
- Enhanced Supply Chain Reliability: By utilizing readily available starting materials like crotononitrile and dimethyl malonate, the process reduces dependency on specialized custom synthons that often suffer from long lead times. The robustness of the nitrile chemistry allows for wider operating windows, decreasing the likelihood of batch failures due to minor process deviations. This reliability ensures a steady flow of materials, crucial for partners managing just-in-time inventory for clinical trials and commercial launches.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, with fewer unit operations meaning less equipment footprint and lower capital expenditure for production facilities. The shift away from heavy metal catalysts and hazardous iodine waste aligns with increasingly strict global environmental regulations, reducing the regulatory burden on manufacturing sites. This sustainability profile enhances the long-term viability of the supply source and supports the corporate social responsibility goals of downstream pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this patented synthesis route. These answers are derived directly from the experimental data and process descriptions found within the patent documentation to ensure accuracy and relevance for technical stakeholders.
Q: How does this process improve upon conventional iodination methods?
A: This method eliminates the need for expensive and environmentally burdensome iodination steps by utilizing a bromination strategy on nitrile-substituted intermediates, which allows for harsher reaction conditions and higher conversion rates.
Q: What is the advantage of using nitrile substituents over esters in this synthesis?
A: The presence of the nitrile moiety prevents ester cleavage and lactone formation during bromination, enabling the use of more reactive brominating agents and higher temperatures for improved efficiency.
Q: Can the intermediate reaction products be isolated between halogenation steps?
A: No, the process is designed as a 'through-process' where chlorination, bromination, and piperazine addition occur sequentially without isolating intermediates, significantly reducing production cycle time.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ipatasertib Intermediate Supplier
At NINGBO INNO PHARMCHEM, we understand that the successful development of AKT inhibitors depends on the consistent availability of high-quality intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements regardless of the project phase. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of cyclopenta[d]pyrimidin-4-yl piperazine compounds meets the exacting standards required for oncology drug synthesis. Our commitment to technical excellence means we can adapt this advanced patented chemistry to our manufacturing lines efficiently.
We invite you to contact our technical procurement team to discuss how we can support your specific project needs with a Customized Cost-Saving Analysis. By leveraging our expertise in process optimization, we can help you secure specific COA data and route feasibility assessments tailored to your timeline. Partner with us to ensure a stable, compliant, and cost-effective supply chain for your next-generation cancer therapeutics.
