Advanced Synthesis of Glufosinate-Ammonium Salt Intermediates via Palladium Catalysis
Introduction to Patent CN108383870B
The global demand for non-selective herbicides has surged following regulatory restrictions on older chemistries like paraquat, positioning glufosinate-ammonium as a critical agricultural input. Patent CN108383870B introduces a transformative synthetic methodology for producing glufosinate-ammonium salt intermediates, addressing long-standing safety and efficiency bottlenecks in agrochemical manufacturing. This technology leverages a sophisticated palladium-catalyzed oxidation strategy to construct the carbon backbone, offering a distinct advantage over traditional routes that rely on hazardous reagents. By shifting the synthetic paradigm towards catalytic oxidation and reductive amination, the patent outlines a pathway that is not only chemically elegant but also commercially viable for high-volume production. The core innovation lies in the precise control of oxidation states using molecular oxygen and permanganate, ensuring high purity profiles essential for downstream formulation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of glufosinate-ammonium has been dominated by the Strecker method, as exemplified in prior art such as US6359162. This legacy approach typically involves the reaction of diethyl methylphosphite with acrolein, followed by a series of transformations including acidolysis and, most critically, cyanidation. The reliance on highly toxic cyanide reagents presents severe operational risks, necessitating expensive containment infrastructure and complex waste treatment protocols to manage cyanide residues. Furthermore, the multi-step nature of the Strecker route often leads to cumulative yield losses and the generation of difficult-to-remove impurities that compromise the quality of the final active ingredient. Alternative routes, such as those involving Grignard reactions, introduce additional complexities regarding moisture sensitivity and exothermic control, making them less attractive for safe, continuous manufacturing environments.
The Novel Approach
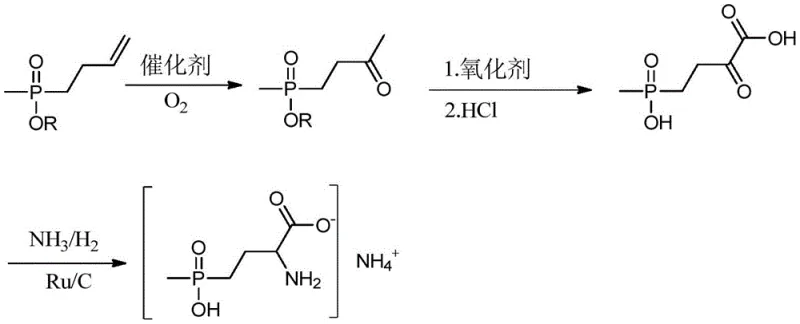
In stark contrast, the methodology disclosed in CN108383870B utilizes a streamlined sequence initiated by the direct oxidation of an allyl phosphonate derivative. As illustrated in the reaction scheme below, the process converts Compound I into a ketone intermediate (Compound II) using a Pd(II) catalytic system under oxygen pressure, effectively bypassing the need for carbon-carbon bond formation via toxic nucleophiles. This is followed by a controlled oxidation and hydrolysis step to generate the key keto-acid intermediate (Compound III), which is subsequently converted to the final herbicide via reductive amination. This route significantly reduces the number of unit operations and eliminates the handling of pyrophoric or acutely toxic substances, thereby simplifying the engineering controls required for plant operation.
Mechanistic Insights into Pd(II)-Catalyzed Oxidation
The cornerstone of this synthetic strategy is the initial transformation of the terminal alkene in Compound I to a methyl ketone in Compound II, a process mechanistically analogous to the Wacker oxidation. In this catalytic cycle, a Pd(II) species coordinates with the olefinic double bond, facilitating nucleophilic attack by water or hydroxide to form a hydroxy-alkyl-palladium intermediate. Subsequent beta-hydride elimination releases the ketone product and generates a Pd(0) species, which is rapidly re-oxidized to the active Pd(II) state by a co-catalyst system, typically involving copper salts like CuCl2 under an oxygen atmosphere. The patent specifies precise molar ratios for the Pd/Cu system (e.g., 1:5 to 1:15) and the inclusion of acidic co-catalysts such as acetic acid or trifluoroacetic acid to maintain catalyst activity and prevent palladium black precipitation. This rigorous control over the redox environment ensures high selectivity for the methyl ketone, minimizing over-oxidation or isomerization byproducts that could complicate downstream purification.
Following the formation of the ketone, the synthesis proceeds to the critical intermediate Compound III through an oxidative cleavage or functionalization of the phosphonate ester side chain using potassium permanganate in alkaline media. This step effectively hydrolyzes the ethyl ester on the phosphorus atom to the free acid while simultaneously managing the oxidation state of the carbon chain to prepare it for amination. The subsequent reductive amination step employs a heterogeneous ruthenium on carbon (Ru/C) catalyst under ammonia and hydrogen pressure. This heterogeneous catalysis is particularly advantageous for commercial scale-up as it allows for easy filtration and recovery of the precious metal catalyst, reducing metal contamination in the final agrochemical product. The mechanism involves the formation of an imine intermediate from the ketone and ammonia, which is immediately hydrogenated to the primary amine, locking in the stereochemistry and structural integrity required for herbicidal activity.
How to Synthesize Glufosinate-Ammonium Salt Efficiently
The execution of this synthesis requires careful attention to reaction parameters, particularly the oxygen pressure and temperature during the initial palladium-catalyzed step to maximize turnover frequency. The patent details a robust protocol where Compound I is reacted in a binary solvent system of water and organic solvents like acetonitrile or DMF, ensuring adequate solubility for both the organic substrate and the inorganic catalyst components. Following the oxidation, the workup involves standard extraction and distillation techniques to isolate the ketone intermediate with high purity before proceeding to the permanganate oxidation. For the final amination, the use of methanol as a solvent and precise control of ammonia partial pressure are critical to driving the equilibrium towards the desired amine product without forming secondary amine byproducts.
- Perform Pd(II)-catalyzed oxidation of the allyl phosphonate precursor (Compound I) using oxygen at 20-60°C and 0.4-0.7 MPa to form the ketone intermediate (Compound II).
- React Compound II with an oxidant (e.g., KMnO4) in alkaline aqueous solution at 0-50°C, followed by acidification to yield the keto-acid intermediate (Compound III).
- Dissolve Compound III in solvent, add Ru/C catalyst, and react sequentially with ammonia gas and hydrogen gas to obtain the final glufosinate ammonium salt (Compound IV).
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this patented route offers substantial strategic benefits by de-risking the manufacturing process and optimizing cost structures. The elimination of cyanide and Grignard reagents removes the need for specialized hazardous material handling certifications and reduces the liability insurance costs associated with toxic inventory storage. Furthermore, the use of molecular oxygen as the primary oxidant in the first step represents a significant cost reduction in agrochemical intermediate manufacturing, as oxygen is vastly cheaper and more atom-economical than stoichiometric organic oxidants. The simplified workflow, characterized by fewer reaction steps and easier isolation procedures, translates directly into reduced utility consumption and shorter batch cycle times, enhancing overall plant throughput capacity without requiring capital-intensive expansion.
- Cost Reduction in Manufacturing: The process achieves cost efficiency primarily through the substitution of expensive and hazardous reagents with catalytic systems and commodity chemicals like oxygen and ammonia. By avoiding the complex waste treatment streams associated with cyanide detoxification, facilities can realize significant operational expenditure savings. Additionally, the high yields reported in the patent examples suggest a reduction in raw material consumption per kilogram of finished product, further improving the gross margin profile for manufacturers adopting this technology.
- Enhanced Supply Chain Reliability: The reliance on stable, commercially available starting materials such as allyl phosphonates and common transition metal salts ensures a resilient supply chain less susceptible to the volatility of niche reagent markets. The robustness of the reaction conditions, operating at moderate temperatures and pressures, minimizes the risk of unplanned shutdowns due to thermal runaways or equipment failures. This stability allows for more predictable production scheduling and shorter lead times for high-purity agrochemical intermediates, enabling suppliers to respond more agilely to seasonal demand fluctuations in the farming sector.
- Scalability and Environmental Compliance: The transition to a catalytic oxidation pathway aligns perfectly with modern environmental, social, and governance (ESG) goals by drastically reducing the generation of toxic byproducts. The ability to scale this process from laboratory to commercial tonnage is facilitated by the use of standard reactor configurations capable of handling gas-liquid reactions, avoiding the need for cryogenic conditions or exotic metallurgy. This ease of scale-up ensures that suppliers can rapidly ramp up production volumes to meet global herbicide demand while maintaining strict compliance with increasingly stringent environmental regulations regarding heavy metal and cyanide discharge.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route, derived directly from the patent specifications and experimental data. Understanding these nuances is vital for technical teams evaluating the feasibility of technology transfer or licensing agreements. The answers provided reflect the specific advantages of the Pd-catalyzed route over legacy methodologies, focusing on safety, yield, and operational simplicity.
Q: How does this patent avoid the use of toxic cyanide?
A: Unlike the traditional Strecker method which requires highly toxic cyanide for carbon chain extension, this patented route utilizes a palladium-catalyzed Wacker-type oxidation to functionalize the allyl group directly, followed by oxidative cleavage/hydrolysis, completely eliminating cyanide hazards.
Q: What are the typical reaction conditions for the key oxidation step?
A: The critical first step operates under mild conditions, specifically between 20-60°C and an oxygen pressure of 0.4-0.7 MPa, utilizing a Pd(II) catalytic system often paired with copper salts or acidic co-catalysts to ensure high conversion rates.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is designed for industrial scalability with short reaction steps, mild temperature requirements, and the use of robust heterogeneous catalysts like Ru/C in the final step, facilitating easier product isolation and purification compared to legacy methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Glufosinate-ammonium Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes like CN108383870B to maintain competitiveness in the global agrochemical market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot scale to full manufacturing is seamless and efficient. We are committed to delivering high-purity intermediates that meet stringent purity specifications, utilizing our rigorous QC labs to verify that every batch conforms to the highest standards of quality and safety required by international regulatory bodies.
We invite you to collaborate with us to leverage this innovative technology for your supply chain needs. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this cyanide-free route can optimize your total cost of ownership. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us partner with you to secure a sustainable and efficient supply of glufosinate-ammonium intermediates.
