Revolutionizing L-Glufosinate Production: High-Efficiency Biocatalysis and Green Substrate Synthesis
Introduction to Next-Generation Herbicide Manufacturing
The global demand for effective, environmentally sustainable herbicides is driving a significant shift in agrochemical manufacturing strategies, particularly for broad-spectrum agents like L-glufosinate-ammonium. As detailed in the groundbreaking patent CN114958934A, a novel biocatalytic approach has emerged that addresses the critical limitations of traditional chemical synthesis. This technology leverages a specifically engineered glutamate dehydrogenase derived from Alkallimnicola ehrlichii AK93 to catalyze the asymmetric reductive amination of a keto acid substrate. Unlike conventional methods that struggle with low substrate tolerance and complex purification, this innovation enables high-conversion reactions at substrate concentrations exceeding 1.5M. For industry leaders seeking a reliable agrochemical intermediate supplier, this patent represents a pivotal advancement in achieving both economic efficiency and environmental compliance.
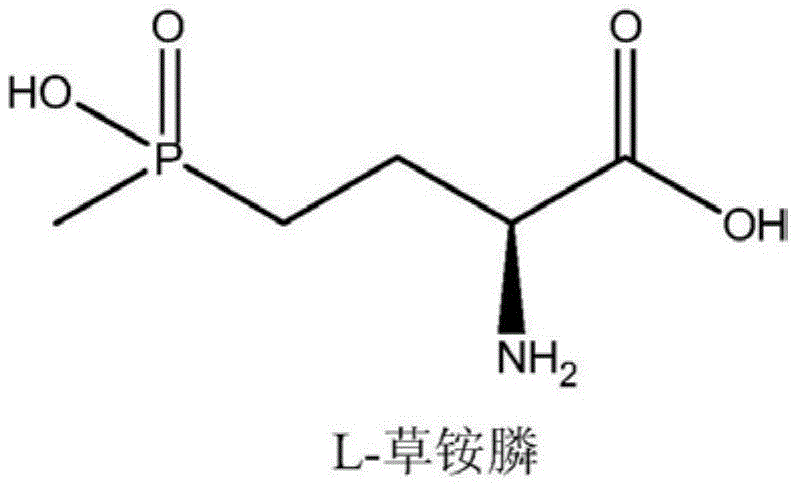
The significance of this technology extends beyond mere yield improvements; it fundamentally alters the impurity profile and safety landscape of production. By utilizing an enzymatic pathway, manufacturers can bypass the harsh conditions and toxic reagents associated with older chemical routes. The resulting L-glufosinate-ammonium possesses superior herbicidal activity compared to racemic mixtures, allowing for reduced application rates in the field. This aligns perfectly with modern regulatory pressures demanding lower environmental burdens. For procurement and supply chain executives, understanding the mechanistic underpinnings of this process is essential for evaluating long-term sourcing strategies and ensuring the continuity of high-purity raw material supplies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of L-glufosinate-ammonium has been plagued by significant technical and safety hurdles inherent to chemical synthesis and chiral resolution. Traditional chiral resolution methods suffer from a theoretical maximum yield of only 50%, necessitating the recycling of unwanted enantiomers and driving up operational costs. Furthermore, chemical synthesis routes often rely on asymmetric hydrogenation using expensive chiral catalysts or the Strecker reaction, which requires the handling of highly toxic cyanide compounds. As illustrated in prior art pathways, the use of cyanide introduces severe safety risks and generates hazardous waste streams that are costly to treat and dispose of in compliance with environmental regulations.
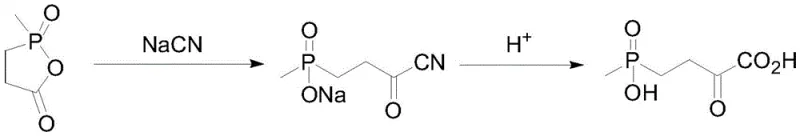
Additionally, existing enzymatic methods using transaminases have faced their own set of challenges, primarily regarding substrate inhibition and equilibrium limitations. Previous reports indicate that typical glutamate dehydrogenases used in similar contexts exhibit low substrate tolerance, often capping out at concentrations around 100 mM. This limitation forces manufacturers to process large volumes of solvent to produce relatively small amounts of product, drastically reducing reactor throughput and increasing energy consumption for downstream separation. The generation of difficult-to-separate byproducts in transaminase-catalyzed reactions further complicates purification, leading to lower overall yields and higher production costs that erode profit margins in a competitive market.
The Novel Approach
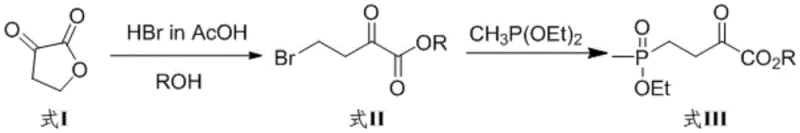
The innovative process disclosed in the patent data offers a transformative solution by combining a green chemical synthesis of the substrate with a highly efficient biocatalytic step. The new route for synthesizing the precursor, 4-(hydroxy-(methyl)phosphinyl)-2-oxobutyric acid (PPO), begins with dihydro-2,3-furandione, avoiding cyanide entirely. This substrate is then subjected to asymmetric reductive amination using the engineered glutamate dehydrogenase SEQ ID NO: 2 or its V375 mutants. This enzyme demonstrates remarkable robustness, tolerating substrate concentrations as high as 1.5M without significant inhibition, which represents a more than tenfold increase over prior art capabilities.
Moreover, this novel approach utilizes inorganic ammonium salts as the amino donor, which simplifies the reaction stoichiometry and eliminates the formation of organic byproducts that are difficult to remove. The integration of a coenzyme regeneration system ensures that expensive cofactors like NADPH are continuously recycled, making the process economically viable on a large scale. By streamlining the synthesis from cheap, readily available starting materials and employing a biocatalyst with superior kinetics, this method achieves high atom economy and reduces the overall carbon footprint. For stakeholders focused on cost reduction in agrochemical intermediate manufacturing, this dual innovation in both substrate preparation and enzymatic conversion offers a compelling value proposition.
Mechanistic Insights into Glutamate Dehydrogenase Catalyzed Reductive Amination
The core of this technological breakthrough lies in the specific catalytic mechanism of the engineered glutamate dehydrogenase (GluDH). The enzyme facilitates the transfer of a hydride ion from the reduced cofactor NADPH to the carbonyl carbon of the keto acid substrate, while simultaneously incorporating an ammonia molecule to form the chiral amine. The patent highlights specific mutations at the 375th amino acid position (Valine to Serine, Alanine, Glycine, or Cysteine), which appear to optimize the active site geometry for the bulky phosphinic acid-containing substrate. These modifications reduce steric hindrance and enhance the binding affinity, thereby mitigating the substrate inhibition effects that typically plague wild-type enzymes at high concentrations.
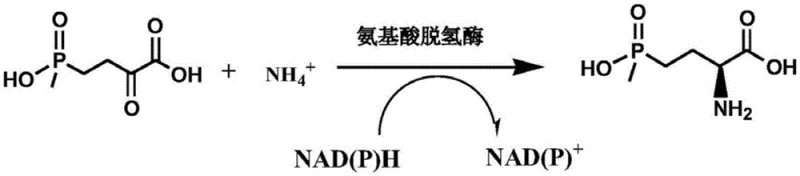
From an impurity control perspective, the high stereoselectivity of the mutant enzymes is paramount. The biocatalytic reaction proceeds with an enantiomeric excess (ee) value exceeding 99%, effectively suppressing the formation of the inactive D-enantiomer. This level of precision is difficult to achieve with chemical catalysts without extensive optimization and purification steps. The use of a coenzyme regeneration system, such as glucose dehydrogenase or formate dehydrogenase, maintains the redox balance within the reaction mixture, ensuring that the reduction potential remains favorable throughout the conversion. This mechanistic efficiency translates directly to process reliability, minimizing batch-to-batch variability and ensuring consistent product quality for downstream formulation.
How to Synthesize L-Glufosinate-Ammonium Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for implementing this technology at an industrial scale. It begins with the preparation of the keto acid substrate through a safe, cyanide-free chemical sequence, followed by the biocatalytic transformation using resting cells or purified enzyme preparations. The process is designed to be operationally simple, requiring standard fermentation and reaction equipment without the need for high-pressure or extreme temperature conditions. Detailed standardized synthesis steps are provided below to guide technical teams in replicating these results.
- Synthesize the keto acid substrate PPO by reacting dihydro-2,3-furandione with hydrobromic acid and alcohol, followed by phosphonation with diethyl methylphosphite.
- Deprotect the intermediate ester using concentrated hydrochloric acid to obtain 4-(hydroxy-(methyl)phosphinyl)-2-oxobutyric acid.
- Perform asymmetric reductive amination using the engineered glutamate dehydrogenase (SEQ ID NO: 2 or V375 mutants) in the presence of an amino donor and coenzyme regeneration system.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this biocatalytic platform offers substantial strategic benefits that extend beyond simple unit cost metrics. The elimination of toxic cyanide reagents significantly reduces the regulatory burden and insurance costs associated with hazardous material handling and storage. Furthermore, the ability to run reactions at high substrate concentrations means that existing reactor infrastructure can produce significantly more product per batch, effectively increasing capacity without capital expenditure. This operational efficiency leads to a more resilient supply chain capable of meeting fluctuating market demands with greater agility.
- Cost Reduction in Manufacturing: The process achieves cost optimization through multiple vectors, primarily by removing the need for expensive chiral resolving agents and precious metal catalysts. The high substrate tolerance of the enzyme allows for smaller reactor volumes to produce the same output, reducing solvent usage and energy costs for heating and cooling. Additionally, the simplified downstream processing, driven by the absence of complex organic byproducts, lowers the cost of purification and waste disposal. These factors combine to create a leaner manufacturing model with improved margin potential.
- Enhanced Supply Chain Reliability: Reliance on hazardous raw materials like cyanide creates vulnerability in the supply chain due to strict transportation regulations and potential supply disruptions. By switching to benign starting materials such as dihydro-2,3-furandione and ammonium salts, manufacturers can secure a more stable and diverse supplier base. The robustness of the engineered enzyme also ensures consistent production schedules, minimizing the risk of batch failures that could lead to stockouts and contractual penalties.
- Scalability and Environmental Compliance: The green chemistry principles embedded in this process facilitate easier scale-up from pilot to commercial production. The avoidance of heavy metals and toxic reagents simplifies wastewater treatment, ensuring compliance with increasingly stringent environmental standards globally. This environmental stewardship not only mitigates regulatory risk but also enhances the brand reputation of the final agrochemical product among environmentally conscious consumers and distributors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. They are derived from the specific experimental data and beneficial effects described in the patent documentation, providing clarity on performance metrics and operational parameters. Understanding these details is crucial for technical due diligence and feasibility assessments.
Q: What are the advantages of the new GluDH enzyme over traditional transaminases?
A: The patented glutamate dehydrogenase exhibits significantly higher substrate tolerance, allowing concentrations up to 1.5M compared to the typical 100mM limit of prior art enzymes. Additionally, it uses inorganic ammonium as a direct amino donor, eliminating difficult-to-separate organic byproducts common in transaminase processes.
Q: How does the new substrate synthesis route improve safety?
A: The novel route starts from dihydro-2,3-furandione and avoids the use of highly toxic cyanide reagents required in traditional Strecker synthesis methods. This eliminates severe safety hazards and simplifies wastewater treatment protocols.
Q: What optical purity can be achieved with this biocatalytic method?
A: The process utilizes highly stereoselective enzyme mutants (such as V375S) that consistently deliver L-glufosinate-ammonium with an enantiomeric excess (ee) value greater than 99%, meeting the stringent quality requirements for modern herbicide formulations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-Glufosinate-Ammonium Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the biocatalytic routes described in patent CN114958934A for the production of high-quality herbicides. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes are successfully translated into robust industrial operations. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the critical enantiomeric excess values required for L-glufosinate-ammonium.
We invite you to collaborate with our technical procurement team to explore how this advanced manufacturing technology can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this green biocatalytic process. We encourage you to contact us today to discuss specific COA data and route feasibility assessments tailored to your unique production requirements and market goals.
