Revolutionizing Triazole Manufacturing: A Novel Iodine-Promoted Route for Commercial Scale-Up
Revolutionizing Triazole Manufacturing: A Novel Iodine-Promoted Route for Commercial Scale-Up
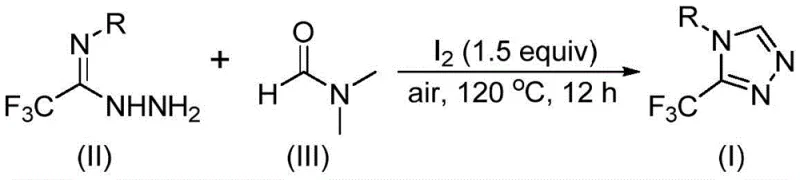
The pharmaceutical and agrochemical industries are constantly seeking robust, scalable, and cost-effective methodologies for constructing privileged heterocyclic scaffolds. A recent technological breakthrough, documented in patent CN114920707B, introduces a highly efficient preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds. These structural motifs are critical components in numerous high-value active pharmaceutical ingredients (APIs), including notable examples like Factor IXa inhibitors and GlyT1 inhibitors, where the trifluoromethyl group enhances metabolic stability and lipophilicity. The core innovation of this patent lies in its ingenious utilization of N,N-dimethylformamide (DMF), a ubiquitous polar aprotic solvent, serving a dual role as both the reaction medium and the essential carbon source. This approach fundamentally shifts the paradigm of triazole synthesis by eliminating the need for specialized, expensive carbon donors, thereby offering a streamlined pathway that is exceptionally attractive for reliable pharmaceutical intermediate supplier networks aiming to optimize their production portfolios.
Furthermore, the operational simplicity of this method addresses several long-standing pain points in fine chemical manufacturing. Unlike traditional routes that often demand stringent exclusion of moisture and oxygen, this novel protocol operates effectively under ambient air conditions. The reaction utilizes molecular iodine as a promoter, which activates the DMF solvent to participate in a tandem cyclization with trifluoroethyliminohydrazide derivatives. This results in the formation of the target 1,2,4-triazole ring with high efficiency and broad substrate tolerance. For procurement and supply chain leaders, this translates to a process that is not only chemically elegant but also logistically superior, reducing the dependency on complex infrastructure and specialized handling protocols that typically inflate the cost of goods sold (COGS) for complex heterocycles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted 1,2,4-triazoles has been fraught with synthetic challenges that hinder large-scale adoption. Conventional methodologies frequently rely on multi-step sequences involving hazardous reagents, expensive transition metal catalysts, or unstable precursors that require cryogenic storage and handling. Many established protocols necessitate strictly anhydrous and anaerobic environments to prevent catalyst deactivation or side reactions, imposing a heavy burden on reactor design and operational safety protocols. Additionally, the use of stoichiometric amounts of specialized carbon sources often leads to poor atom economy and generates significant volumes of chemical waste, complicating downstream purification and environmental compliance. These factors collectively contribute to extended lead times and elevated production costs, making it difficult for manufacturers to achieve the cost reduction in pharmaceutical intermediate manufacturing required to remain competitive in the global market.
The Novel Approach
In stark contrast, the method disclosed in CN114920707B offers a transformative solution by leveraging the inherent reactivity of DMF. By employing molecular iodine to activate the solvent, the process facilitates a direct cyclization that incorporates a carbon atom from the DMF molecule directly into the triazole ring. This strategy drastically simplifies the reaction setup, as it removes the necessity for external carbon donors and allows the reaction to proceed in open vessels under air. The conditions are remarkably mild yet effective, typically requiring temperatures between 110°C and 130°C for 10 to 15 hours. This robustness ensures that the process is highly amenable to commercial scale-up of complex pharmaceutical intermediates, as it mitigates the risks associated with scaling sensitive organometallic reactions. The ability to synthesize a diverse array of substituted triazoles from readily available starting materials further underscores the practical utility of this approach for industrial applications.
Mechanistic Insights into Iodine-Promoted Tandem Cyclization
The mechanistic elegance of this transformation lies in the dual activation pathways facilitated by molecular iodine. As illustrated in the reaction scheme below, the process begins with the interaction between iodine and DMF. Depending on which functional group of the DMF participates, two distinct pathways can be envisioned, both converging on the desired triazole product. In one pathway, the formyl group of DMF acts as the carbon source, undergoing condensation with the trifluoroethyliminohydrazide to form a hydrazone intermediate. This is followed by an intramolecular cyclization and the elimination of dimethylamine to yield the final 3-trifluoromethyl-1,2,4-triazole. Alternatively, the N-methyl group of DMF can serve as the carbon donor, where iodine promotes the formation of an amine salt species that subsequently reacts with the hydrazide. This pathway involves the elimination of nitrogen-methylformamide to generate an azadiene intermediate, which then undergoes oxidative aromatization to furnish the target heterocycle.
This mechanistic flexibility contributes to the high reaction efficiency observed across a wide range of substrates. From an impurity control perspective, the use of DMF as both solvent and reactant minimizes the introduction of extraneous organic fragments that could complicate the impurity profile. The primary byproducts, such as dimethylamine or nitrogen-methylformamide, are volatile or easily separable, facilitating straightforward purification via standard silica gel column chromatography. For R&D directors focused on purity specifications, this clean reaction profile is a significant advantage, as it reduces the burden on analytical quality control and ensures that the final high-purity pharmaceutical intermediates meet the rigorous standards required for subsequent coupling reactions in API synthesis. The tolerance for various functional groups on the aromatic ring further demonstrates the robustness of the catalytic cycle against electronic and steric variations.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting is straightforward, requiring minimal specialized equipment beyond standard heating and stirring capabilities. The procedure involves charging a reaction vessel with the trifluoroethyliminohydrazide substrate, molecular iodine, and DMF solvent. The mixture is then heated to the specified temperature range under air until conversion is complete, as monitored by standard analytical techniques such as TLC or HPLC. The detailed standardized synthetic steps, including precise molar ratios and workup procedures derived from the patent examples, are outlined in the guide below to ensure reproducibility and optimal yield.
- Combine molecular iodine, trifluoroethyliminohydrazide, and organic solvent DMF in a reaction vessel under air atmosphere.
- Heat the reaction mixture to a temperature range of 110-130°C and maintain stirring for 10-15 hours to ensure complete conversion.
- Upon completion, perform post-treatment including filtration, washing, drying, and purification via silica gel column chromatography to isolate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this DMF-based methodology presents a compelling value proposition centered on cost efficiency and operational resilience. The primary driver of cost reduction is the elimination of expensive, specialized reagents in favor of commodity chemicals. DMF is one of the most widely produced and inexpensive solvents in the chemical industry, and its dual usage here means that no additional carbon source needs to be purchased, stored, or handled. Furthermore, the use of molecular iodine as a promoter is significantly more economical than relying on precious metal catalysts like palladium or rhodium, which are subject to volatile market pricing and supply constraints. This shift to base-metal-free catalysis inherently lowers the raw material cost baseline, enabling substantial savings that can be passed down the supply chain or reinvested into process optimization.
- Cost Reduction in Manufacturing: The economic benefits extend beyond mere reagent costs to include significant savings in operational expenditures. Because the reaction does not require an inert atmosphere, manufacturers can utilize standard glass-lined or stainless steel reactors without the need for nitrogen purging systems or glovebox setups. This simplification reduces energy consumption associated with gas handling and lowers the capital expenditure required for reactor modification. Additionally, the high atom economy of using the solvent as a reactant minimizes waste generation, which in turn reduces the costs associated with waste disposal and environmental compliance. These cumulative effects create a leaner manufacturing process that is highly resistant to inflationary pressures on raw materials.
- Enhanced Supply Chain Reliability: Supply chain continuity is critically dependent on the availability of starting materials. The substrates required for this synthesis, specifically the trifluoroethyliminohydrazides, can be rapidly synthesized from commercially available aromatic amines, triphenylphosphine, and hydrazine hydrate. This reliance on bulk commodity chemicals ensures that production is not bottlenecked by the scarcity of exotic reagents. Moreover, the robustness of the reaction conditions means that the process is less susceptible to variability caused by minor fluctuations in environmental conditions or reagent quality. This reliability allows for more accurate production planning and shorter lead times, ensuring that downstream API manufacturing schedules are met without interruption.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to the tonne scale often introduces new safety and environmental challenges. However, the absence of pyrophoric reagents and the operation under air atmosphere make this triazole synthesis inherently safer and easier to scale. The simplified workup procedure, involving filtration and chromatography, is compatible with existing purification infrastructure in most fine chemical facilities. From an environmental standpoint, the reduced use of hazardous reagents and the minimization of waste streams align with green chemistry principles. This facilitates smoother regulatory approvals and supports corporate sustainability goals, making the technology attractive for companies aiming to reduce their environmental footprint while maintaining high production volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel triazole synthesis method. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical aspects of adopting this technology for industrial production.
Q: What are the key advantages of using DMF as a carbon source in this synthesis?
A: Using DMF eliminates the need for separate, expensive carbon sources as it acts as both the solvent and the reactant. This dual functionality significantly simplifies the reaction setup, reduces raw material costs, and streamlines the purification process by minimizing extraneous byproducts.
Q: Does this method require strict anhydrous or anaerobic conditions?
A: No, one of the most significant operational benefits of this patented method is that it proceeds efficiently under standard air atmosphere without the need for rigorous anhydrous or oxygen-free environments, greatly facilitating industrial scale-up and reducing equipment complexity.
Q: What is the substrate scope for the R group in this triazole synthesis?
A: The method demonstrates excellent versatility, accommodating a wide range of substituents on the aromatic ring including alkyl, alkoxy, alkylthio, halogens, and trifluoromethyl groups at ortho, meta, or para positions, allowing for the synthesis of diverse bioactive analogs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient heterocycle synthesis in the development of next-generation therapeutics. Our team of expert chemists has thoroughly analyzed the potential of the iodine-promoted DMF cyclization route and is fully prepared to integrate this technology into our manufacturing portfolio. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial supply is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 3-trifluoromethyl-1,2,4-triazole intermediate delivered meets the exacting standards required for pharmaceutical applications.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project needs. By leveraging our expertise in process optimization, we can provide a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this method for your specific supply chain. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and efficiency in your drug development programs.
