Advanced Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles for Pharmaceutical Applications
Advanced Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles for Pharmaceutical Applications
The pharmaceutical industry continuously seeks robust and efficient synthetic routes for heterocyclic compounds that serve as critical scaffolds in modern drug design. A significant breakthrough in this domain is detailed in patent CN113307778A, which discloses a novel preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds. These structures are ubiquitous in bioactive molecules, appearing in prominent drugs such as Sitagliptin and various anticonvulsants, where the trifluoromethyl group enhances metabolic stability and lipophilicity. The disclosed technology offers a streamlined pathway to access these valuable intermediates, addressing long-standing challenges in yield and operational complexity faced by reliable pharmaceutical intermediate suppliers.
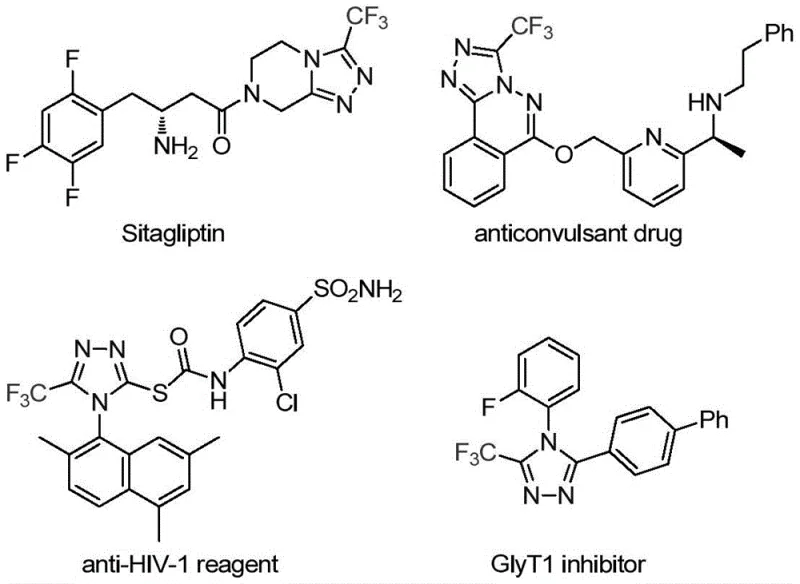
This innovation is particularly relevant for R&D teams focusing on cost reduction in pharmaceutical intermediates manufacturing. By leveraging a dual-catalyst system involving molybdenum and copper, the process achieves high conversion rates under relatively mild thermal conditions. The ability to synthesize diverse derivatives through simple substrate design makes this method a versatile tool for generating libraries of potential drug candidates, ensuring a steady supply of high-purity pharmaceutical intermediates for downstream development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted 1,2,4-triazoles has relied on methodologies that often suffer from significant drawbacks regarding safety and efficiency. Traditional routes frequently involve the cyclization of trifluoroacetyl hydrazine with amidine compounds or the hydrazinolysis of trifluoromethyl-substituted 1,2,4-oxazolinones. These processes can require harsh reaction conditions, specialized reagents that are difficult to source commercially, and multi-step sequences that erode overall yield. Furthermore, alternative copper-catalyzed multi-component reactions utilizing diazonium salts or trifluorodiazoethane often present safety hazards due to the explosive nature of diazo compounds, complicating the commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
In stark contrast, the method described in CN113307778A utilizes a sophisticated yet operationally simple co-catalytic system. By reacting trifluoroethylimidoyl chloride with functionalized isonitrile (NIITP) in the presence of molybdenum hexacarbonyl and cuprous acetate, the synthesis proceeds smoothly. This approach eliminates the need for hazardous diazo reagents and simplifies the reaction setup to a single pot. The use of common organic solvents like THF and mild heating (70-90°C) significantly lowers the barrier for implementation in standard laboratory and pilot plant settings. This shift represents a major step forward in reducing lead time for high-purity pharmaceutical intermediates by streamlining the workflow from raw materials to final product.
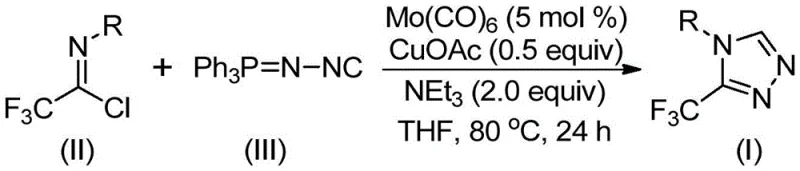
Mechanistic Insights into Mo/Cu Co-Catalyzed Cycloaddition
The core of this technological advancement lies in the synergistic interaction between the molybdenum and copper catalysts. Mechanistically, the molybdenum hexacarbonyl acts as a metal activator for the functionalized isonitrile, forming a reactive metal complex that facilitates the subsequent bond formation. Simultaneously, the cuprous acetate promotes a [3+2] cycloaddition reaction between the activated isonitrile species and the trifluoroethylimidoyl chloride. This concerted effort leads to the formation of a five-membered ring intermediate, which is the precursor to the triazole core. The elegance of this mechanism is further highlighted by the in-situ removal of triphenylphosphine oxide under the action of water within the system, driving the equilibrium towards the final 3-trifluoromethyl-substituted 1,2,4-triazole compound without requiring additional purification steps for phosphine byproducts.
From an impurity control perspective, this mechanism offers distinct advantages. The specificity of the [3+2] cycloaddition minimizes the formation of regioisomers, a common issue in triazole synthesis. The mild conditions prevent the decomposition of sensitive functional groups on the aromatic rings, such as halogens or nitro groups, which might degrade under more vigorous traditional conditions. This high chemoselectivity ensures that the resulting crude product has a cleaner profile, simplifying the downstream purification process and ensuring that the final material meets the stringent quality standards required for high-purity pharmaceutical intermediates.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for producing these valuable heterocycles. The process involves mixing specific molar ratios of the imidoyl chloride, isonitrile, and catalysts in an aprotic solvent, followed by heating. The simplicity of the workup, involving filtration and standard chromatography, makes it highly accessible. For detailed operational parameters and specific stoichiometric ratios optimized for different substrates, please refer to the standardized synthesis guide below.
- Combine molybdenum hexacarbonyl, cuprous acetate, triethylamine, molecular sieves, trifluoroethylimidoyl chloride, and functionalized isonitrile in an organic solvent such as THF.
- Heat the reaction mixture to a temperature between 70°C and 90°C and maintain stirring for a duration of 18 to 30 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the final 3-trifluoromethyl-substituted 1,2,4-triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible benefits that extend beyond mere chemical yield. The reliance on commercially available and inexpensive starting materials, such as trifluoroethylimidoyl chloride and functionalized isonitriles, stabilizes the supply chain against volatility. Unlike methods requiring custom-synthesized exotic reagents, the inputs for this process are commodity chemicals, ensuring consistent availability and predictable pricing structures for long-term contracts.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the use of earth-abundant metal catalysts like copper and molybdenum, which are significantly cheaper than precious metals like palladium or rhodium often used in cross-coupling reactions. Furthermore, the elimination of hazardous reagents reduces the costs associated with special handling, storage, and waste disposal. The high reaction efficiency means less raw material is wasted, directly contributing to substantial cost savings in the overall manufacturing budget without compromising on quality.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes to a more reliable supply chain. Because the reaction tolerates a wide range of functional groups and proceeds effectively in common solvents like THF, there is less risk of batch failure due to minor variations in raw material quality or environmental conditions. This reliability translates to more consistent delivery schedules for clients, reducing the risk of production delays in their own downstream drug manufacturing processes.
- Scalability and Environmental Compliance: The method is explicitly designed to be scalable, having been demonstrated effectively at the gram level with potential for ton-scale production. The simplified post-treatment process, which avoids complex extraction or neutralization steps typical of acid/base heavy syntheses, reduces the volume of chemical waste generated. This aligns with modern green chemistry principles, making it easier for manufacturers to meet increasingly strict environmental regulations while maintaining high throughput.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis method. The answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on its practical application and advantages over legacy technologies.
Q: What are the key advantages of this Mo/Cu co-catalyzed method over traditional synthesis?
A: This method utilizes cheap and readily available starting materials like functionalized isonitrile and trifluoroethylimidoyl chloride. It operates under mild conditions (70-90°C) compared to harsher traditional cyclization methods, offering higher reaction efficiency and easier post-treatment.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the patent explicitly states that the method can be expanded to gram-level reactions and beyond. The use of common solvents like THF and standard purification techniques like column chromatography facilitates scale-up for commercial manufacturing.
Q: What types of substituents are tolerated in this reaction?
A: The method demonstrates wide substrate tolerance. The R group can be phenethyl or various substituted aryl groups, including those with methyl, methoxy, halogen, or trifluoromethyl substituents at ortho, meta, or para positions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthetic methodologies play in accelerating drug discovery and development. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory results translate seamlessly into industrial reality. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify every batch.
We invite you to collaborate with us to leverage this advanced technology for your specific project needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to help you optimize your supply chain and bring your innovative therapies to market faster.
