Revolutionizing Abiraterone Acetate Purification: A Scalable Oxalate Salt Strategy for Global API Manufacturing
Introduction to Advanced Purification Technologies
The pharmaceutical landscape for oncology treatments demands rigorous standards for Active Pharmaceutical Ingredients (APIs), particularly for potent agents like Abiraterone acetate, a critical inhibitor used in treating metastatic castration-resistant prostate cancer. As detailed in patent CN103059090A, a significant technological breakthrough has been achieved in the purification of this complex steroid intermediate through the novel application of oxalate salt formation. This innovation addresses long-standing bottlenecks in downstream processing, shifting away from labor-intensive chromatographic separations toward efficient crystallization-based purification. By leveraging the specific physicochemical properties of Abiraterone acetate oxalate, manufacturers can now bypass the formation of intractable oily residues and sticky suspensions that have historically plagued production lines. This report analyzes the technical merits of this oxalate-mediated pathway, offering a robust solution for achieving ultra-high purity while simultaneously optimizing operational expenditure and supply chain reliability for global pharmaceutical partners.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the purification of Abiraterone acetate crude product has relied heavily on column chromatography or alternative salt formation strategies that introduce significant operational inefficiencies. Prior art, such as the mesylate salt method described in CN101044155, suffers from severe physical handling issues; the reaction between Abiraterone acetate and methanesulfonic acid generates a viscous, paste-like suspension that is notoriously difficult to filter. This rheological challenge leads to thick filter cakes that retain substantial amounts of mother liquor and impurities, necessitating repeated recrystallization steps that erode overall yield and extend cycle times. Furthermore, the resulting free base obtained after alkali dissociation often remains as an oily substance with purity levels hovering around 89%, contaminated by stubborn pigment impurities and hydrolysis byproducts that are resistant to removal. Alternatively, the trifluoromethanesulfonate (triflate) approach outlined in CN102030798, while producing a filterable solid, relies on triflic acid, a reagent that is prohibitively expensive, highly corrosive to standard stainless steel equipment, and requires strict low-temperature control below 5°C to prevent acid mist generation, thereby increasing energy consumption and safety risks.
The Novel Approach
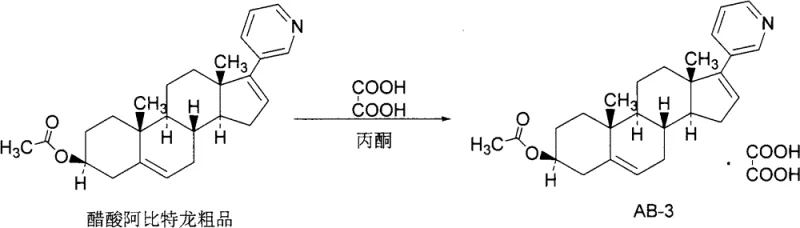
In stark contrast to these legacy methodologies, the oxalate salt strategy introduced in patent CN103059090A presents a paradigm shift towards greener and more manageable processing. By reacting the crude Abiraterone acetate with oxalic acid in solvents such as acetone or acetonitrile, the process yields Abiraterone acetate oxalate as a free-flowing, off-white granular solid. This physical form allows for rapid suction filtration and efficient washing, effectively trapping impurities like unreacted starting materials and colored byproducts within the mother liquor rather than the crystal lattice. The mild reaction conditions, operating optimally between 30°C and 50°C, eliminate the need for cryogenic cooling and specialized corrosion-resistant reactors. Subsequent dissociation of the oxalate salt using mild bases like sodium bicarbonate regenerates the free base in a highly pure crystalline form, consistently achieving purity specifications greater than 99.5% without the need for silica gel column chromatography. This streamlined workflow not only simplifies the unit operations but also drastically reduces the environmental footprint associated with solvent usage and hazardous waste disposal.
Mechanistic Insights into Oxalate Salt Formation and Dissociation
The core of this purification technology lies in the precise acid-base chemistry that governs the selective crystallization of the target molecule. Abiraterone acetate, possessing a basic pyridine nitrogen moiety within its steroidal framework, acts as a proton acceptor when introduced to oxalic acid, a dicarboxylic acid with suitable pKa values for stable salt formation. Upon dissolution of the crude material in a polar aprotic solvent like acetone, the addition of oxalic acid initiates a proton transfer reaction, converting the lipophilic free base into a more polar ionic salt species. This transformation significantly alters the solubility profile of the molecule, driving it out of the solution phase as a defined crystalline lattice. The crystallization kinetics are carefully managed by controlling the temperature and stirring time, ensuring that the growing crystals exclude structurally similar impurities and amorphous pigment materials which remain solvated. This selective precipitation is the fundamental mechanism that enables the dramatic increase in purity observed in the intermediate salt form.
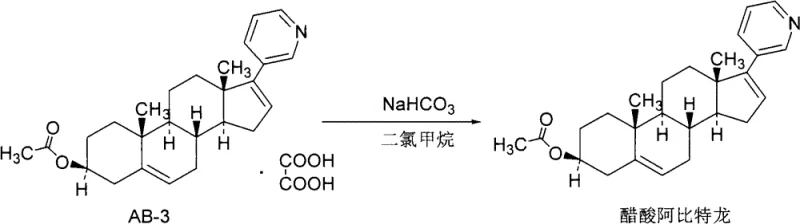
Following the isolation of the high-purity oxalate salt, the final step involves a controlled dissociation reaction to regenerate the pharmacologically active free base. The solid oxalate salt is suspended in a biphasic system typically comprising dichloromethane and water, where a mild alkaline agent such as sodium bicarbonate is introduced. The bicarbonate ions neutralize the oxalic acid component of the salt, shifting the equilibrium back towards the neutral free base form of Abiraterone acetate. Due to the lipophilic nature of the steroid backbone, the regenerated free base partitions preferentially into the organic phase, leaving the water-soluble oxalate salts and any remaining inorganic impurities in the aqueous layer. This liquid-liquid extraction serves as a secondary purification barrier, further polishing the product before final solvent removal and recrystallization. The result is a chemically pristine API intermediate ready for formulation, demonstrating how simple stoichiometric adjustments can replace complex physical separation techniques.
How to Synthesize Abiraterone Acetate Efficiently
The implementation of this oxalate-based purification route requires careful attention to solvent selection and stoichiometric ratios to maximize recovery and crystal quality. The process begins with the dissolution of the crude Abiraterone acetate, typically obtained from upstream coupling reactions involving dehydroepiandrosterone acetate derivatives, in a volume of acetone ranging from 5 to 15 times the weight of the substrate. Oxalic acid is then added in a molar ratio of 1:1 to 2:1 relative to the crude drug, ensuring complete conversion to the salt form without excessive acid residue. The mixture is heated to approximately 50°C to facilitate reaction kinetics and then slowly cooled to room temperature to promote orderly crystal growth over a period of 1 to 3 hours. For detailed operational parameters, safety guidelines, and specific workup procedures, please refer to the standardized synthesis guide provided below.
- Dissolve Abiraterone acetate crude product in a suitable solvent such as acetone or acetonitrile.
- Add oxalic acid to the solution at a temperature between 30°C and 50°C to induce salt formation and crystallization.
- Filter the resulting Abiraterone acetate oxalate solid, wash, and dissociate with sodium bicarbonate to obtain high-purity free base.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this oxalate purification methodology translates directly into tangible operational efficiencies and risk mitigation. The transition away from corrosive triflic acid and sticky mesylate intermediates removes several critical failure points from the manufacturing value chain. By utilizing commodity-grade oxalic acid and common solvents like acetone, the process decouples production from the volatility of specialized reagent markets, ensuring a more stable and predictable cost structure. Furthermore, the physical characteristics of the oxalate salt—specifically its free-flowing granular nature—dramatically improve throughput in the filtration and drying stages, reducing the batch cycle time compared to the prolonged handling required for paste-like mesylate filters. This enhancement in processing speed allows for better utilization of existing reactor capacity, effectively increasing plant throughput without the need for capital-intensive infrastructure upgrades.
- Cost Reduction in Manufacturing: The economic advantages of this route are driven primarily by the substitution of high-cost, hazardous reagents with inexpensive, widely available commodities. Eliminating the need for trifluoromethanesulfonic acid removes a significant line item from the bill of materials, while the avoidance of column chromatography reduces the consumption of tons of silica gel and associated solvent volumes. Additionally, the simplified workup procedure reduces labor hours and utility consumption related to extended drying and recycling processes. These factors combine to lower the overall cost of goods sold (COGS), providing a competitive margin advantage in the generic API market.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the robustness of the chemical process, which tolerates minor variations in crude feedstock quality better than sensitive chromatographic methods. The ability to produce a stable, storable intermediate salt form allows manufacturers to decouple the synthesis and purification stages, creating strategic inventory buffers that protect against upstream disruptions. Moreover, the use of non-hazardous, non-corrosive reagents simplifies logistics and storage requirements, reducing the regulatory burden and transportation costs associated with moving dangerous goods across international borders.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process offers a cleaner profile that aligns with increasingly stringent global regulations. The absence of heavy metal catalysts in the purification step and the reduction of halogenated solvent usage during workup minimize the generation of hazardous waste streams. The high efficiency of the crystallization process also means less solvent is required per kilogram of product, lowering the facility's overall carbon footprint. This sustainability advantage is increasingly becoming a key differentiator for suppliers seeking to qualify with top-tier multinational pharmaceutical companies that prioritize green chemistry principles in their vendor selection criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and validation of the Abiraterone acetate oxalate purification process. These insights are derived directly from the experimental data and comparative studies presented in the underlying patent literature, providing a factual basis for technology transfer discussions. Understanding these nuances is essential for R&D teams evaluating the feasibility of integrating this route into their existing manufacturing platforms.
Q: Why is the oxalate salt method superior to mesylate or triflate purification for Abiraterone acetate?
A: Unlike mesylate salts which form sticky, difficult-to-filter suspensions, or triflate salts which require expensive and corrosive reagents, the oxalate salt forms a dry, granular solid that filters rapidly. This eliminates the need for column chromatography and significantly reduces equipment corrosion risks.
Q: What purity levels can be achieved using this oxalate purification route?
A: The process effectively removes pigments, unreacted raw materials, and hydrolysis byproducts. Following dissociation and recrystallization, the final Abiraterone acetate product consistently achieves purity levels exceeding 99.5%, meeting stringent pharmacopoeial standards.
Q: Is this purification method suitable for large-scale industrial production?
A: Yes, the method is highly scalable. It operates at mild temperatures (30-50°C), uses commodity chemicals like oxalic acid and acetone, and avoids complex separation techniques, making it ideal for multi-ton commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Abiraterone Acetate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and proven scalability. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in patent literature are faithfully reproduced at an industrial level. We maintain stringent purity specifications through our rigorous QC labs, employing advanced analytical techniques to verify that every batch of Abiraterone acetate meets the exacting standards required for oncology therapeutics. Our commitment to quality assurance extends beyond mere compliance; we actively optimize every unit operation to ensure consistency, safety, and efficiency throughout the supply chain.
We invite potential partners to engage with our technical procurement team to discuss how this advanced oxalate purification technology can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a clearer understanding of the economic benefits specific to your volume requirements. We encourage you to reach out for specific COA data and route feasibility assessments, allowing us to demonstrate concretely how our manufacturing capabilities can support your long-term supply goals for high-purity Abiraterone acetate and related intermediates.
