Advanced Suzuki Coupling Strategy for Commercial Scale-up of Abiraterone Acetate Intermediates
Advanced Suzuki Coupling Strategy for Commercial Scale-up of Abiraterone Acetate Intermediates
The pharmaceutical landscape for oncology treatments has been significantly transformed by the introduction of Abiraterone Acetate, a potent inhibitor of CYP17A1 used in the management of metastatic castration-resistant prostate cancer. As global demand for this critical active pharmaceutical ingredient continues to rise, the industry faces increasing pressure to optimize manufacturing processes that are not only cost-effective but also environmentally sustainable and scalable. Patent CN103864877A, published in June 2014, introduces a groundbreaking synthetic methodology that addresses these challenges by leveraging a highly efficient Suzuki-Miyaura coupling reaction. This technical insight report analyzes the proprietary advancements detailed in the patent, focusing on the transition from hazardous, low-yield conventional methods to a streamlined, aqueous-phase catalytic system. By integrating a specialized palladium catalyst system with a polyoxyethanyl-tocopheryl sebacate (PTS) surfactant, this novel approach achieves yields exceeding 90% while operating under remarkably mild conditions, positioning it as a superior choice for reliable pharmaceutical intermediates supplier networks seeking to enhance their production capabilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Abiraterone Acetate has been plagued by complex multi-step sequences that introduce significant operational risks and economic inefficiencies. One prominent conventional pathway, disclosed in earlier literature, relies on the formation of a triflate intermediate from Dehydroepiandrosterone Acetate using trifluoromethanesulfonic anhydride. As illustrated in the reaction scheme below, this route is fundamentally limited by the poor conversion efficiency of the initial esterification step, which often caps at approximately 58%, leaving a substantial portion of the valuable starting material unreacted and complicating downstream purification efforts.
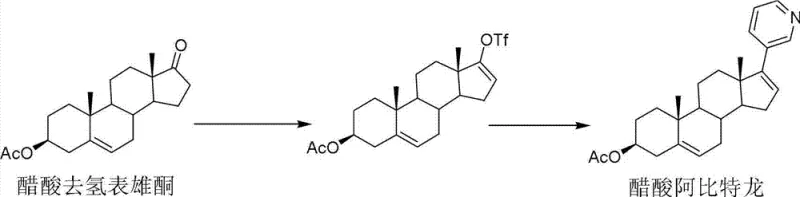
Furthermore, alternative historical methods have depended on the use of hydrazine hydrate and hydrazonium sulfate to generate hydrazone intermediates, followed by iodination and subsequent coupling. These processes necessitate the use of diethyl ether, a solvent with well-documented explosion hazards, and involve reagents with irritating odors and high toxicity profiles. The cumulative effect of these lengthy synthetic sequences is a total recovery rate that struggles to exceed 48.7%, resulting in excessive waste generation and inflated production costs that are unsustainable for modern cost reduction in pharmaceutical intermediates manufacturing initiatives.
The Novel Approach
In stark contrast to these cumbersome legacy processes, the methodology outlined in Patent CN103864877A offers a direct and robust solution by utilizing 17-iodoandrosta-5,16-dien-3beta-ol acetate as the key starting material. This innovative route eliminates the need for hazardous hydrazine derivatives and explosive ether solvents, replacing them with a sophisticated palladium-catalyzed cross-coupling reaction performed in the presence of an aqueous PTS catalyst solution. The reaction scheme depicted below highlights the elegance of this single-step coupling strategy, where the iodo-intermediate reacts directly with 3-pyridylboronic acid to form the target biaryl structure with exceptional precision.
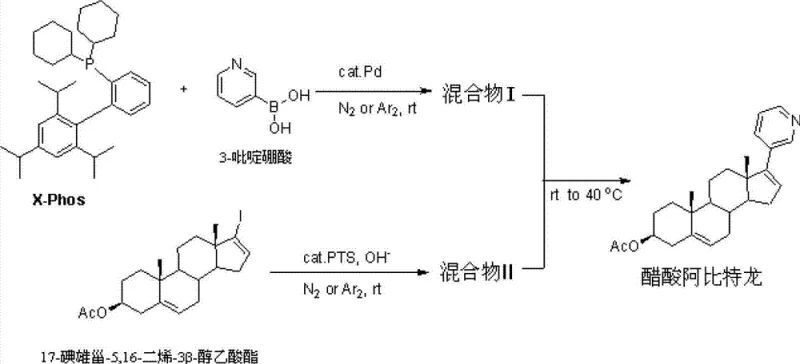
This novel approach operates under mild thermal conditions, typically ranging from room temperature to 40°C, which drastically reduces energy consumption compared to high-temperature reflux methods required by older technologies. By streamlining the synthesis into fewer operational units and utilizing a water-miscible catalyst system, the process not only simplifies the workflow but also significantly enhances the safety profile of the manufacturing environment, making it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into PTS-Assisted Suzuki-Miyaura Coupling
The core of this technological breakthrough lies in the unique catalytic system that facilitates the cross-coupling reaction within a biphasic or micellar environment. The process employs a palladium catalyst, such as PdCl2(dtbpf) or Pd(OAc)2, in conjunction with the bulky phosphine ligand X-Phos (2-dicyclohexylphosphino-2',4',6'-triisopropylbiphenyl). This specific ligand choice is critical for stabilizing the active palladium species and promoting the oxidative addition of the sterically hindered steroid iodide. The inclusion of Polyoxyethanyl-alpha-tocopheryl Sebacate (PTS) serves a dual function: it acts as a phase transfer agent that solubilizes the organic substrates in the aqueous medium and potentially stabilizes the palladium nanoparticles, thereby maintaining high catalytic activity throughout the reaction cycle without the need for expensive anhydrous organic solvents.
From an impurity control perspective, this mechanistic pathway offers distinct advantages over traditional methods. The high selectivity of the Suzuki coupling under these specific basic conditions minimizes the formation of homocoupling byproducts or dehalogenated side products that often plague steroid functionalization reactions. The use of a mild base, such as triethylamine or potassium carbonate, in conjunction with the PTS surfactant ensures that the sensitive steroid backbone remains intact while facilitating the rapid transmetallation step with the boronic acid. This precise control over the reaction environment results in a crude product profile that is exceptionally clean, reducing the burden on downstream purification processes and ensuring that the final high-purity pharmaceutical intermediates meet stringent regulatory specifications for residual metals and organic impurities.
How to Synthesize Abiraterone Acetate Efficiently
The implementation of this synthesis route requires careful attention to the preparation of the catalytic mixture and the control of reaction parameters to maximize yield and purity. The process begins with the activation of the palladium catalyst under an inert atmosphere, followed by the sequential addition of the boronic acid and the steroid substrate in the presence of the aqueous PTS solution. Operators must maintain strict control over the temperature, keeping it within the 22°C to 40°C range, and ensure adequate mixing to facilitate mass transfer between the phases. For a comprehensive understanding of the specific reagent ratios, mixing times, and workup procedures required to replicate this high-efficiency process, please refer to the standardized synthesis guide provided below.
- Prepare Mixture I by mixing a palladium catalyst, X-Phos ligand, and 3-pyridylboronic acid under inert gas protection at room temperature.
- Prepare Mixture II by mixing 17-iodoandrosta-5,16-dien-3beta-ol acetate with an aqueous PTS catalyst solution under basic conditions.
- Combine Mixture II into Mixture I, react at 22-40°C, then quench, extract, and recrystallize to obtain high-purity Abiraterone Acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route translates into tangible strategic benefits that extend beyond simple chemical yield improvements. The elimination of hazardous reagents like hydrazine and explosive solvents like ether removes significant regulatory hurdles and safety compliance costs associated with storage and handling. Furthermore, the ability to conduct the reaction in an aqueous-organic system reduces the volume of volatile organic compounds (VOCs) emitted during production, aligning manufacturing operations with increasingly rigorous environmental standards and reducing the financial liability associated with waste disposal and emissions trading schemes.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the dramatic improvement in overall yield, which exceeds 90% compared to the sub-50% recovery of older methods. This efficiency gain means that less raw material is required to produce the same amount of finished goods, directly lowering the cost of goods sold. Additionally, the removal of the triflation step eliminates the need for expensive triflic anhydride and the associated neutralization waste streams, while the use of readily available industrial intermediates ensures stable pricing and reduces exposure to volatile specialty chemical markets.
- Enhanced Supply Chain Reliability: By simplifying the synthetic sequence and utilizing robust, commercially available starting materials such as 17-iodoandrosta-5,16-dien-3beta-ol acetate, manufacturers can significantly reduce lead time for high-purity pharmaceutical intermediates. The process is less susceptible to supply disruptions caused by the scarcity of exotic reagents, and the mild reaction conditions allow for flexible scheduling without the need for specialized high-temperature or cryogenic equipment, thereby improving overall plant throughput and delivery reliability.
- Scalability and Environmental Compliance: The green chemistry principles embedded in this route, particularly the use of water-miscible catalysts and the avoidance of toxic hydrazines, make it inherently easier to scale from pilot batches to multi-ton commercial production. The reduced toxicity profile simplifies the permitting process for new manufacturing lines and lowers the operational risk profile, ensuring continuous supply continuity even in regions with strict environmental enforcement, thus securing the long-term viability of the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for evaluating the feasibility of this route for your specific production needs. Understanding these details is crucial for R&D teams planning technology transfers and for procurement specialists negotiating supply agreements.
Q: What are the primary advantages of the PTS-catalyzed Suzuki coupling route for Abiraterone Acetate?
A: The primary advantages include significantly higher yields exceeding 90%, the elimination of hazardous reagents like hydrazine and ether, and the ability to operate under mild temperatures between 22°C and 40°C, which enhances process safety and environmental compliance.
Q: How does this method improve upon conventional triflation-based synthesis routes?
A: Conventional triflation routes often suffer from low conversion rates in the esterification step, typically around 58%, and require expensive triflic anhydride. The novel Suzuki coupling method bypasses these inefficient steps, utilizing readily available iodo-intermediates to achieve superior overall recovery and purity.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the process is designed for industrial scalability. It utilizes stable starting materials, avoids explosive solvents, and employs a robust catalytic system that functions effectively in aqueous-organic biphasic systems, making it ideal for commercial scale-up from pilot plants to multi-ton manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Abiraterone Acetate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of life-saving oncology drugs depends on a supply chain that is both resilient and technically sophisticated. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volumetric demands of global pharmaceutical partners without compromising on quality. We are committed to maintaining stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify that every batch of Abiraterone Acetate meets the highest international standards for potency and impurity profiles.
We invite you to collaborate with us to leverage this cutting-edge synthesis technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this efficient route can optimize your budget. Please contact us today to request specific COA data and route feasibility assessments, and let us help you secure a competitive advantage in the rapidly evolving market for prostate cancer therapeutics.
