Scalable Purification of Clevidipine Butyrate Intermediates for Global Pharmaceutical Supply Chains
Scalable Purification of Clevidipine Butyrate Intermediates for Global Pharmaceutical Supply Chains
The global demand for rapid-acting intravenous antihypertensive agents has placed significant scrutiny on the supply chain reliability of key pharmaceutical intermediates. Specifically, the synthesis of Clevidipine Butyrate, a dihydropyridine calcium channel antagonist approved for perioperative blood pressure management, relies heavily on the quality of its precursor, 4-(2,3-dichlorophenyl)-2,6-dimethyl-1,4-dihydropyridine-3-methyl carboxylate-5-carboxylic acid. Patent CN103086956A introduces a transformative purification methodology that addresses the longstanding bottleneck of dicarboxylic acid impurity removal. This technical breakthrough shifts the paradigm from low-yield chromatographic separation to a highly efficient, scalable salification and precipitation protocol. For R&D directors and procurement strategists, understanding this shift is critical, as it directly impacts the cost of goods sold (COGS) and the ability to meet stringent regulatory purity specifications without compromising production timelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Clevidipine Butyrate has been plagued by the presence of a stubborn structural analog known as the dicarboxylic acid impurity. This byproduct, 4-(2,3-dichlorophenyl)-2,6-dimethyl-1,4-dihydro-3,5-pyridine-dicarboxylic acid, is generated during the initial Hantzsch condensation reaction and possesses physicochemical properties nearly identical to the desired monocarboxylic acid intermediate. Conventional purification strategies described in prior art, such as WO1995/12578 and CN101759631A, often resort to repeated recrystallization using ethyl acetate and petroleum ether or preparative chromatography. These methods are inherently inefficient; recrystallization often fails to reduce impurity levels below critical thresholds, while preparative chromatography is prohibitively expensive and difficult to scale for multi-ton production. Furthermore, literature indicates that traditional routes may suffer from overall yields as low as 45%, representing a massive loss of valuable raw materials and creating significant waste disposal challenges for environmental compliance teams.
The Novel Approach
The innovative strategy outlined in patent CN103086956A circumvents these limitations by exploiting subtle solubility differences between the target monocarboxylic acid and the dicarboxylic acid impurity upon salt formation. Instead of attempting to purify the free acid directly, the process converts the crude mixture into an alkali metal salt using bases such as sodium hydroxide or potassium carbonate in a lower alkanol solvent. This step selectively precipitates the salt of the monocarboxylic acid, leaving the dicarboxylic acid impurities largely in the mother liquor or altering their solubility profile sufficiently for separation. Following filtration, the purified salt is redissolved in water and carefully acidified to a specific pH range to regenerate the high-purity free acid. This approach not only simplifies the operational workflow but also dramatically enhances the purity profile of the intermediate before it enters the final esterification stage, as illustrated in the reaction scheme below.
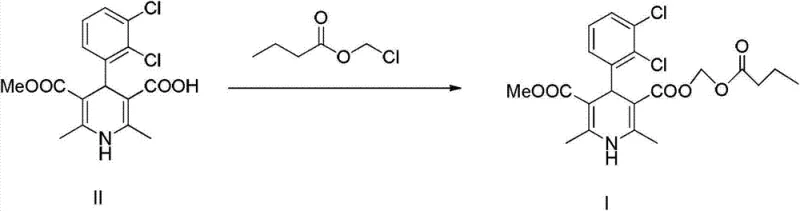
Mechanistic Insights into Selective Salification and Precipitation
The core of this purification technology lies in the differential behavior of the mono-acid and di-acid species under basic conditions in alcoholic media. When the crude intermediate is treated with an alkali metal hydroxide, such as sodium hydroxide, in a solvent like isopropanol or ethanol, the monocarboxylic acid moiety reacts to form a mono-salt. The patent data suggests that the dicarboxylic acid impurity, possessing two acidic protons, may form a di-salt or exhibit different solvation characteristics that prevent it from co-precipitating with the desired mono-salt under controlled temperature conditions (5°C to 30°C). This selective crystallization is the primary purification driver, effectively scrubbing the bulk of the structurally related impurity from the solid phase. The subsequent dissolution of this isolated salt in water creates a homogeneous aqueous phase where the final purification occurs through precise pH control.
The final regeneration step involves adjusting the pH of the aqueous salt solution to a slightly acidic range, specifically between pH 5 and pH 6, using mineral acids like hydrochloric acid. This specific pH window is critical; it is sufficiently acidic to protonate the carboxylate anion of the target molecule, causing it to precipitate out of the aqueous solution as a solid, yet it avoids conditions that might promote the precipitation of remaining impurities or cause degradation of the sensitive dihydropyridine ring. The result is a highly purified solid with HPLC purity exceeding 99.3% and dicarboxylic acid impurity levels reduced to approximately 0.41%. This level of purity is essential because any residual dicarboxylic acid carried forward would react with chloromethyl butyrate to form a di-ester impurity in the final API, which is notoriously difficult to remove later. The mechanistic pathway for this purification is detailed in the following diagram.
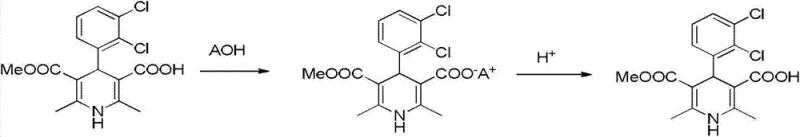
How to Synthesize 4-(2,3-dichlorophenyl)-2,6-dimethyl-1,4-dihydropyridine-3-methyl carboxylate-5-carboxylic acid Efficiently
The implementation of this purification protocol requires careful attention to solvent selection and stoichiometry to maximize recovery and purity. The process begins with the suspension of the crude acid in a lower alkanol, followed by the controlled addition of the base to ensure complete salt formation without excessive exothermicity. Once the salt is isolated and washed, the aqueous workup must be managed to maintain the optimal concentration for precipitation. For a comprehensive breakdown of the specific molar ratios, solvent volumes, and temperature profiles required to replicate this high-yield process, please refer to the standardized synthesis guide provided below.
- React the crude monocarboxylic acid intermediate with an alkali metal hydroxide or carbonate in a lower alkanol solvent to form the corresponding alkali metal salt.
- Separate the precipitated alkali metal salt from the solvent to isolate it from soluble impurities, then dissolve the salt in water.
- Adjust the aqueous solution pH to slightly acidic conditions (pH 5-6) to precipitate the high-purity monocarboxylic acid intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this purification method translates into tangible operational efficiencies and risk mitigation. By eliminating the reliance on preparative chromatography, manufacturers can significantly reduce capital expenditure on specialized separation equipment and lower the ongoing costs associated with chromatographic resins and high-purity solvents. The shift to a crystallization-based purification allows for the use of standard stainless steel reactors and filtration units, which are ubiquitous in fine chemical manufacturing facilities, thereby enhancing supply chain flexibility and reducing lead times for capacity booking.
- Cost Reduction in Manufacturing: The elimination of chromatographic steps and the reduction in recrystallization cycles lead to a substantial decrease in processing time and solvent consumption. Since the process achieves high recovery rates during the purification step itself, there is less need to over-produce crude intermediate to compensate for downstream losses. This efficiency directly lowers the variable cost per kilogram of the intermediate, providing a competitive pricing advantage for the final API without sacrificing margin.
- Enhanced Supply Chain Reliability: Traditional methods that depend on repeated recrystallization often suffer from unpredictable yields and batch-to-batch variability, which can disrupt production schedules. The robust nature of the salification and precipitation method ensures consistent output quality and quantity. This reliability allows supply chain planners to forecast inventory levels with greater accuracy and reduces the risk of stockouts for critical antihypertensive medications, ensuring continuity of supply for hospital formularies.
- Scalability and Environmental Compliance: Scaling a chromatographic process from the lab to multi-ton production is technically challenging and often requires disproportionate increases in facility footprint. In contrast, this precipitation-based method scales linearly with reactor volume, facilitating a smoother transition from pilot plant to commercial manufacturing. Additionally, the reduction in organic solvent usage and the avoidance of complex waste streams associated with chromatography simplify wastewater treatment protocols, aligning with increasingly stringent environmental regulations and sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this purification technology. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering a clear perspective on why this method represents a superior alternative to legacy processes for producing high-value pharmaceutical intermediates.
Q: Why is the removal of dicarboxylic acid impurities critical in Clevidipine Butyrate synthesis?
A: The dicarboxylic acid impurity is structurally similar to the target monocarboxylic acid intermediate, making it extremely difficult to separate via standard recrystallization. If not removed prior to the final esterification step, it forms a related substance in the final API that exceeds strict pharmacopoeial limits, necessitating complex and costly preparative chromatography.
Q: How does the salification purification method improve overall yield compared to conventional routes?
A: Conventional routes often rely on repeated recrystallization or chromatography, which result in significant material loss, with some reports indicating yields as low as 45%. The novel salification method leverages solubility differences to achieve purification yields around 90%, significantly boosting the total throughput of the manufacturing process.
Q: Is this purification process suitable for large-scale industrial production?
A: Yes, the process is designed for scalability. It replaces equipment-intensive chromatography with standard unit operations like filtration, dissolution, and pH-adjusted precipitation. This simplifies the workflow, reduces solvent consumption, and eliminates the need for expensive transition metal catalysts or complex separation columns.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Clevidipine Butyrate Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the quality of the final Active Pharmaceutical Ingredient is inextricably linked to the purity of its precursors. Our technical team has extensively analyzed the purification pathway described in CN103086956A and has integrated similar high-efficiency crystallization technologies into our own manufacturing platforms. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to industrial reality is seamless. Our facilities are equipped with rigorous QC labs capable of detecting trace impurities at ppm levels, guaranteeing that every batch of intermediate meets stringent purity specifications required by global regulatory bodies.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging our optimized synthesis and purification capabilities, we can help you reduce the total cost of ownership for your Clevidipine Butyrate supply chain. Contact our technical procurement team today to request specific COA data, route feasibility assessments, and samples for your validation studies, ensuring your project moves forward with the highest degree of confidence and efficiency.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
