Advanced Manufacturing of Clevidipine Butyrate: A Chromatography-Free Route for Global Supply Chains
Introduction to Advanced Clevidipine Butyrate Manufacturing
The global demand for rapid-acting antihypertensive agents has placed significant pressure on supply chains to deliver high-purity active pharmaceutical ingredients (APIs) efficiently. Patent CN101759631A introduces a transformative methodology for the preparation of Clevidipine Butyrate, a fourth-generation dihydropyridine calcium channel blocker widely used for acute blood pressure control. This patent outlines a robust synthetic route that overcomes historical bottlenecks associated with chromatographic purification and harsh reaction conditions. By leveraging a one-pot Hantzsch reaction followed by a streamlined deprotection and esterification sequence, the technology achieves a final product purity exceeding 99.5% without the need for preparative chromatography. This breakthrough is particularly relevant for reliable clevidipine butyrate suppliers aiming to scale production while maintaining rigorous quality standards.
The strategic value of this patent lies in its ability to simplify the manufacturing landscape for cardiovascular intermediates. Traditional methods often rely on nitrogen protection and toxic solvents, creating environmental and operational hurdles. In contrast, this novel approach utilizes mild conditions and accessible reagents, such as 2,3-dichlorobenzaldehyde and methyl 3-amino-2-butenoate. For procurement managers and supply chain heads, this represents a shift towards more sustainable and cost-effective manufacturing paradigms. The elimination of complex purification steps not only reduces processing time but also minimizes solvent consumption, aligning with modern green chemistry principles essential for long-term commercial viability in the pharmaceutical sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Clevidipine Butyrate has been plagued by inefficiencies that hinder large-scale commercialization. Prior art, such as the routes described in patents WO1995/12578 and WO2000/31035, typically involves multi-step sequences that require stringent nitrogen protection and the use of hazardous solvents like acetonitrile. A critical bottleneck in these conventional methods is the reliance on preparative chromatography for purification. Chromatographic separation, while effective for small-scale laboratory synthesis, is notoriously difficult to scale up due to high solvent usage, low throughput, and significant operational costs. Furthermore, the need for nitrogen protection adds complexity to the reactor setup and increases the risk of batch failure due to oxygen sensitivity.
Additionally, existing methods often suffer from impurity profiles that are difficult to manage without extensive downstream processing. The use of expensive or hard-to-source intermediates further exacerbates the cost structure, making the final API less competitive in price-sensitive markets. For example, the synthesis of key intermediates often involves stepwise reactions that accumulate impurities, necessitating repeated purification cycles. These factors collectively contribute to longer lead times and higher production costs, creating a fragile supply chain that struggles to meet the consistent demand required for critical care medications like Clevidipine Butyrate.
The Novel Approach
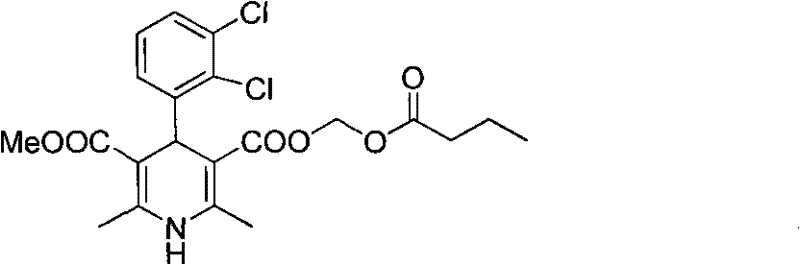
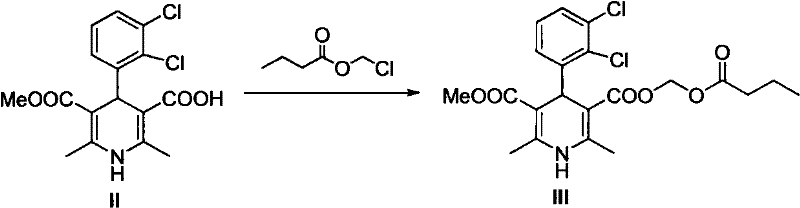
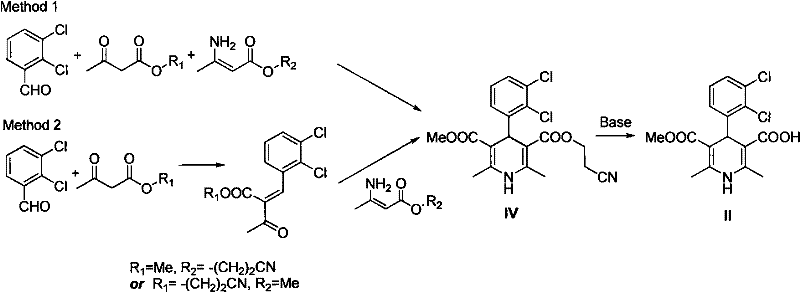
The methodology disclosed in CN101759631A offers a decisive break from these traditional constraints by introducing a convergent and efficient synthetic strategy. The core innovation involves a one-pot Hantzsch reaction that directly constructs the dihydropyridine skeleton from three readily available components: 2,3-dichlorobenzaldehyde, propionitrile acetoacetate, and methyl 3-amino-2-butenoate. This telescoped reaction significantly reduces the number of unit operations and eliminates the need for isolating unstable intermediates. Following the cyclization, a base-catalyzed hydrolysis step efficiently removes the 2-cyanoethyl protecting group to yield the key carboxylic acid intermediate. This intermediate is then esterified with chloromethyl n-butyrate under controlled alkaline conditions to form the final product.
Crucially, this novel approach replaces the cumbersome chromatographic purification with a simple recrystallization process using common solvent systems such as ethyl acetate and petroleum ether. This shift dramatically enhances the scalability of the process, allowing for the production of metric ton quantities with consistent quality. The reaction conditions are mild, operating effectively in isopropanol without the absolute necessity for inert atmosphere protection in all steps, thereby simplifying equipment requirements. For a reliable clevidipine butyrate supplier, this translates to a more robust manufacturing process that is less susceptible to operational variances and capable of delivering high-purity material at a fraction of the traditional cost.
Mechanistic Insights into One-Pot Hantzsch Cyclization
The heart of this synthetic advancement is the optimized Hantzsch dihydropyridine synthesis, a classic multicomponent reaction that has been refined for industrial application. In this specific embodiment, the reaction proceeds through the condensation of the aldehyde, the beta-keto ester derivative (propionitrile acetoacetate), and the enamine (methyl 3-amino-2-butenoate). The mechanism involves the initial formation of an enamine intermediate from the beta-keto ester and the amine component, followed by Knoevenagel condensation with the aldehyde. Subsequent Michael addition and cyclization steps close the dihydropyridine ring. The use of isopropanol as a solvent facilitates the solubility of all three components while providing a thermal environment conducive to the reaction kinetics, typically ranging from 20°C to 180°C depending on the specific optimization.
Following the cyclization, the removal of the 2-cyanoethyl group is a critical deprotection step that unlocks the carboxylic acid functionality required for the final esterification. This is achieved using alkaline catalysts such as sodium hydroxide or potassium carbonate, which hydrolyze the nitrile or facilitate its elimination depending on the specific structural arrangement. The patent highlights the use of bases like NaOH or organic amines to drive this transformation efficiently. The resulting carboxylic acid intermediate is then isolated via acidification, often using phosphoric acid, which precipitates the product in high purity. This mechanistic clarity allows for precise control over impurity formation, ensuring that side products are minimized before the final coupling step.
How to Synthesize Clevidipine Butyrate Efficiently
Implementing this synthesis route requires careful attention to stoichiometry and temperature control to maximize yield and purity. The process begins with the mixing of the three primary reactants in isopropanol, followed by heating to initiate the Hantzsch cyclization. Once the intermediate is formed, the reaction mixture is treated with a base to effect deprotection, followed by extraction and acidification to isolate the key acid intermediate. The final step involves coupling this acid with chloromethyl n-butyrate in a polar aprotic solvent like DMF, using a base such as sodium carbonate to scavenge the generated acid. Detailed standard operating procedures for each stage, including specific molar ratios and workup protocols, are essential for reproducibility.
- Conduct a one-pot Hantzsch reaction by mixing 2,3-dichlorobenzaldehyde, propionitrile acetoacetate, and methyl 3-amino-2-butenoate in isopropanol at 20-180°C.
- Add an alkaline catalyst to remove the 2-cyanoethyl protecting group, followed by acidification to isolate the key intermediate carboxylic acid.
- React the key intermediate with chloromethyl n-butyrate under alkaline conditions, then purify the crude product via recrystallization to achieve >99.5% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented process offers substantial strategic advantages beyond mere technical feasibility. The primary benefit is the drastic simplification of the purification workflow. By eliminating preparative chromatography, manufacturers can significantly reduce solvent consumption and waste generation, leading to direct cost savings in raw materials and waste disposal. This efficiency gain is compounded by the use of inexpensive starting materials; for instance, methyl 3-amino-2-butenoate can be derived from cheap methyl acetoacetate, and propionitrile acetoacetate from diketene. These economic factors contribute to a lower cost of goods sold (COGS), enabling more competitive pricing in the global API market.
- Cost Reduction in Manufacturing: The removal of chromatographic purification steps results in significant operational savings. Chromatography is capital-intensive and consumes vast amounts of high-grade solvents; replacing it with recrystallization reduces both equipment depreciation and consumable costs. Furthermore, the use of commodity chemicals as starting materials ensures that the supply chain is not vulnerable to the price volatility of specialized reagents. This structural cost advantage allows for better margin management and the ability to offer cost reduction in API manufacturing to downstream partners without compromising quality.
- Enhanced Supply Chain Reliability: The robustness of the one-pot reaction and the simplicity of the workup procedures enhance the overall reliability of the supply chain. Fewer unit operations mean fewer points of failure and reduced batch cycle times. The ability to produce the intermediate and final product without nitrogen protection in certain stages further simplifies the infrastructure requirements, allowing for production in a wider range of facilities. This flexibility ensures reducing lead time for high-purity pharmaceutical intermediates, as batches can be turned around more quickly to meet urgent demand fluctuations.
- Scalability and Environmental Compliance: From a sustainability perspective, this process aligns well with environmental regulations. The reduction in solvent usage and the avoidance of toxic acetonitrile in the final steps lower the environmental footprint of the manufacturing process. The scalability is inherently superior because recrystallization is a standard unit operation that scales linearly from kilograms to tons, unlike chromatography which faces diminishing returns at larger scales. This makes the commercial scale-up of complex pharmaceutical intermediates feasible and economically viable, ensuring a continuous supply of this critical antihypertensive agent.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its implementation. The following questions address common concerns regarding purity, scalability, and regulatory compliance associated with the new process. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making.
Q: Does this synthesis method require preparative chromatography?
A: No, a key advantage of this patented process is that it eliminates the need for preparative chromatographic separation. The final product is purified solely through recrystallization using solvent systems like ethyl acetate/petroleum ether, making it highly suitable for large-scale industrial production.
Q: What is the achievable purity of Clevidipine Butyrate using this method?
A: The process yields Clevidipine Butyrate with a purity greater than 99.5%. This high level of purity is achieved through optimized reaction conditions and effective recrystallization steps, meeting stringent pharmaceutical standards without complex purification techniques.
Q: Are the raw materials for this synthesis cost-effective?
A: Yes, the process utilizes readily available and inexpensive starting materials. For instance, methyl 3-amino-2-butenoate can be prepared from cheap methyl acetoacetate, and propionitrile acetoacetate can be derived from industrial chemicals like diketene, significantly lowering the overall cost of goods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Clevidipine Butyrate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes for life-saving medications like Clevidipine Butyrate. Our team of expert chemists has extensively analyzed the technological advancements presented in patent CN101759631A and possesses the capability to implement this chromatography-free process at an industrial scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of high-quality intermediates. Our state-of-the-art facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch meets the >99.5% purity benchmark required for pharmaceutical applications.
We invite global pharmaceutical partners to collaborate with us to leverage this advanced manufacturing technology. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and supply chain needs. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. Together, we can optimize the production of Clevidipine Butyrate, ensuring a stable supply of this essential antihypertensive agent for patients worldwide while maximizing operational efficiency and cost-effectiveness.
