Scalable Synthesis of Methyl 2,3-Diaminopropionate from Serine for Peptide Inhibitors
Introduction to Novel Antiviral Intermediate Synthesis
The development of potent cysteine protease inhibitors, particularly for targeting Human Rhinovirus (HRV) and Norwalk Virus (NV), relies heavily on the availability of high-purity non-natural amino acid building blocks. Patent CN109251150B discloses a robust and efficient preparation method for methyl 2,3-diaminopropionate, a critical scaffold found in the P1 section of classical peptidomimetic inhibitors. As illustrated in the structural analysis of existing inhibitors, the (S)-2-amino-3-substituted aminopropionic acid motif is essential for binding to the highly conserved His-Cys-Glu catalytic triad of viral proteases. This patent presents a strategic shift from expensive, specialized starting materials to the abundant chiral pool amino acid, L-serine, thereby addressing key supply chain vulnerabilities in the manufacturing of antiviral pharmaceutical intermediates.
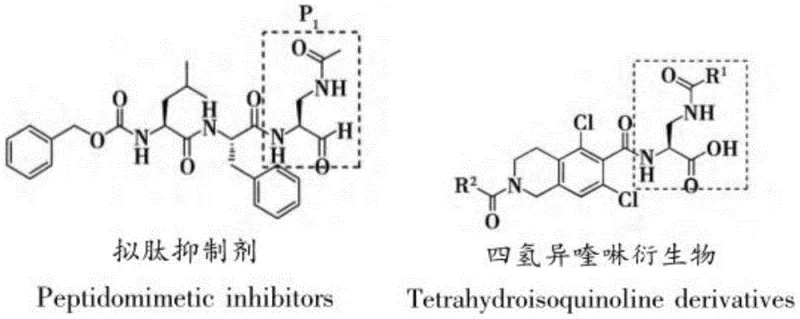
By leveraging a sequence of esterification, orthogonal protection, Mitsunobu coupling, and selective deprotection, this technology enables the production of complex diamino acid derivatives under mild reaction conditions. For R&D directors and procurement specialists, this represents a significant opportunity to optimize the cost of goods sold (COGS) for next-generation antiviral therapies. The methodology not only simplifies the synthetic pathway but also enhances the overall safety profile by eliminating the need for hazardous reagents often associated with traditional diazotization or Schmidt reaction protocols used in earlier generations of amino acid synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2,3-diaminopropionic acid derivatives has been plagued by significant economic and technical hurdles. Traditional routes, such as the one depicted in Synthesis Route 1, often rely on (S)-2-benzyloxycarbonylamino-3-aminopropionic acid as a starting material. While effective, this precursor is a high-value fine chemical with a price point substantially higher than basic bulk amino acids, directly inflating the raw material costs for the final API intermediate. Furthermore, alternative methods involving the Schmidt reaction on aspartic acid or ring-opening of aziridine derivatives frequently require harsh acidic conditions, toxic hydrazoic acid, or complex multi-step protection-deprotection sequences that suffer from moderate yields and difficult purification challenges.
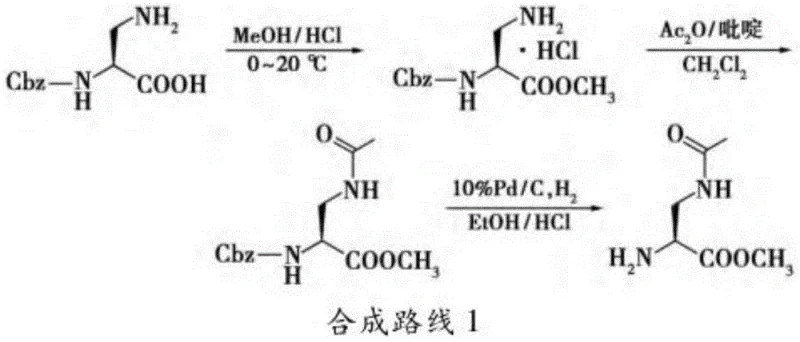
These conventional approaches often result in impurity profiles that are difficult to control, necessitating extensive downstream processing which further erodes profit margins. The reliance on specialized starting materials also introduces supply chain risks, as the availability of these niche intermediates can be limited by the production capacity of a few specialized vendors. Consequently, pharmaceutical manufacturers seeking to scale up production of cysteine protease inhibitors have long sought a more direct, cost-effective, and scalable route that utilizes commoditized feedstocks without compromising stereochemical integrity or product purity.
The Novel Approach
The methodology described in CN109251150B fundamentally disrupts this status quo by establishing L-serine as the primary feedstock. This novel approach initiates with a straightforward esterification using thionyl chloride in methanol, followed by the strategic introduction of a trityl (Trt) group to protect the alpha-amino functionality. The core innovation lies in the subsequent Mitsunobu reaction, which efficiently installs the beta-amino precursor via phthalimide displacement. This sequence allows for the construction of the 1,2-diamine skeleton with high regioselectivity, bypassing the need for pre-functionalized diamino acid starting materials. The final steps involve the orthogonal removal of the phthalimide and trityl groups, yielding the target methyl 2,3-diaminopropionate hydrochloride salt with excellent purity.
Mechanistic Insights into Mitsunobu Coupling and Orthogonal Deprotection
The heart of this synthetic strategy is the Mitsunobu reaction employed to convert the protected serine derivative into the phthalimido intermediate. In this transformation, triphenylphosphine (PPh3) and diisopropyl azodicarboxylate (DIAD) activate the hydroxyl group of the serine side chain, facilitating a nucleophilic attack by phthalimide. This mechanism proceeds with inversion of configuration, although since the beta-carbon in serine is achiral prior to this step, the stereochemistry at the alpha-carbon (derived from L-serine) is preserved throughout the process. The use of anhydrous tetrahydrofuran (THF) as the solvent ensures optimal solubility of the bulky trityl-protected intermediate while maintaining the stability of the reactive phosphonium species generated in situ.
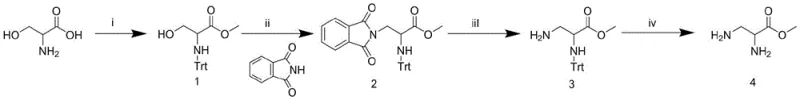
Impurity control is meticulously managed through the orthogonal nature of the protecting groups. The phthalimide (Pht) group is base-labile and can be selectively cleaved using hydrazine hydrate in ethanol without affecting the acid-labile trityl (Trt) ether/amine protection. Conversely, the final deprotection step utilizes ethanolic HCl to remove the robust trityl group, which would otherwise require harsh hydrogenolysis or strong acid conditions that might degrade the sensitive ester moiety. This sequential deprotection strategy minimizes the formation of side products such as diketopiperazines or elimination byproducts, ensuring that the final crude product meets stringent quality specifications suitable for direct use in peptide coupling reactions.
How to Synthesize Methyl 2,3-Diaminopropionate Efficiently
The operational protocol for this synthesis is designed for reproducibility and scalability, utilizing standard laboratory and pilot plant equipment. The process begins with the in situ generation of methyl ester hydrochloride, followed by protection and coupling steps that can be monitored via TLC or HPLC. The detailed standardized synthetic steps, including precise molar ratios, temperature controls, and workup procedures for each of the four key transformations, are outlined in the guide below to ensure consistent batch-to-batch quality.
- Esterify L-Serine with thionyl chloride in methanol, followed by protection of the alpha-amino group with triphenylmethyl chloride (Trt-Cl) to form Intermediate 1.
- Perform a Mitsunobu reaction on Intermediate 1 using phthalimide, triphenylphosphine, and DIAD in anhydrous THF to introduce the beta-amino precursor (Intermediate 2).
- Deprotect the phthalimide group using hydrazine hydrate to yield Intermediate 3, followed by final removal of the Trt group with ethanolic HCl to obtain the target diamine salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this serine-based route offers compelling economic and logistical benefits. By shifting the raw material base from expensive, specialized diamino acid derivatives to L-serine, a commodity chemical produced on a massive global scale, manufacturers can drastically reduce their exposure to raw material price volatility. The elimination of complex, multi-step precursor synthesis translates directly into a simplified supply chain with fewer touchpoints and reduced lead times for raw material acquisition. Furthermore, the use of common organic solvents such as methanol, dichloromethane, and ethyl acetate ensures that the process can be implemented in existing multipurpose facilities without requiring specialized corrosion-resistant reactors or hazardous waste handling infrastructure.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the substitution of high-cost starting materials with low-cost L-serine. Unlike previous routes that required purchasing pre-protected diamino acid scaffolds at a premium, this method builds the complexity from a basic amino acid. Additionally, the high yields observed in the Mitsunobu coupling step (reported over 90% in examples) and the final deprotection step (nearly quantitative) minimize material loss. The avoidance of precious metal catalysts, such as palladium on carbon often used for Cbz removal in older routes, further eliminates the cost associated with metal scavenging and recovery, leading to substantial overall cost savings in the manufacturing of these peptide mimetic intermediates.
- Enhanced Supply Chain Reliability: Sourcing L-serine is significantly more reliable than sourcing niche protected amino acids, as it is a standard fermentation product with multiple global suppliers. This diversification of the supply base mitigates the risk of single-source bottlenecks that can halt production lines. The robustness of the chemical steps, which tolerate mild conditions and do not require cryogenic temperatures or ultra-high vacuum, ensures that production schedules are less susceptible to equipment failures or utility fluctuations. This reliability is crucial for maintaining continuous supply contracts with downstream pharmaceutical clients who demand just-in-time delivery of critical intermediates for clinical and commercial programs.
- Scalability and Environmental Compliance: The synthetic route is inherently scalable, relying on solution-phase chemistry that translates well from gram to kilogram and ton scales. The waste streams generated are primarily organic salts and solvents which can be managed through standard distillation and wastewater treatment protocols, avoiding the generation of heavy metal waste or highly toxic azide byproducts associated with alternative Schmidt reaction pathways. This alignment with green chemistry principles facilitates easier regulatory approval for the manufacturing process and reduces the environmental compliance burden, making it a sustainable choice for long-term commercial production of antiviral drug substances.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this serine-based synthesis route. These answers are derived directly from the experimental data and technical disclosures within the patent documentation, providing clarity on reaction mechanisms, purification strategies, and scalability potential for industrial partners evaluating this technology for their supply chains.
Q: Why is the serine-based route preferred over the Schmidt reaction for 2,3-diaminopropionic acid derivatives?
A: The serine-based route avoids the use of hazardous reagents like hydrazoic acid required in Schmidt reactions and utilizes inexpensive, commercially available L-serine as a chiral pool starting material, significantly improving safety and cost-efficiency.
Q: What is the role of the Trt and Pht protecting groups in this synthesis?
A: The Trt (triphenylmethyl) and Pht (phthalimide) groups provide orthogonal protection, allowing for selective deprotection. The Pht group is removed first under mild hydrazine conditions, while the acid-labile Trt group is removed last, ensuring high regioselectivity and purity.
Q: Can this process be scaled for commercial production of antiviral intermediates?
A: Yes, the process utilizes standard unit operations such as reflux, filtration, and column chromatography with common solvents like methanol, DCM, and ethyl acetate, making it highly amenable to scale-up from kilogram to multi-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Methyl 2,3-Diaminopropionate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving antiviral therapies. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless. We adhere to stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of methyl 2,3-diaminopropionate meets the exacting standards required for GMP peptide synthesis.
We invite you to collaborate with us to leverage this innovative synthetic route for your specific project needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing capabilities can enhance the efficiency and profitability of your supply chain.
