Scalable Synthesis of Methyl 2,3-Diaminopropionate for Advanced Peptidomimetic Inhibitor Manufacturing
Introduction to Novel Peptidomimetic Building Blocks
The pharmaceutical landscape for antiviral therapeutics continues to evolve, driven by the urgent need for effective inhibitors against cysteine proteases found in viruses such as Human Rhinovirus (HRV) and Norwalk Virus (NV). Central to the development of these therapeutics is the availability of high-purity chiral building blocks, specifically methyl 2,3-diaminopropionate and its derivatives. Patent CN109251150B discloses a robust and efficient preparation method for this critical intermediate, addressing long-standing challenges in synthetic accessibility and cost-efficiency. This innovation enables the construction of complex peptidomimetic inhibitors and tetrahydroisoquinoline derivatives, which function by targeting the highly conserved His-Cys-Glu catalytic triad of viral proteases. By securing a reliable supply of this scaffold, manufacturers can accelerate the development of next-generation antiviral agents.
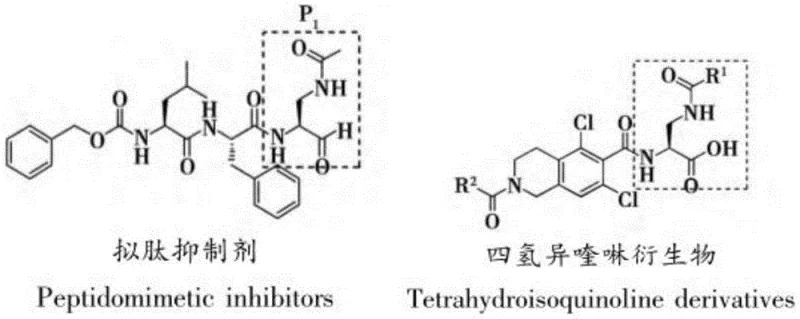
The structural versatility of the (S)-2-amino-3-substituted aminopropionic acid skeleton allows for precise modulation of binding affinity and pharmacokinetic properties in drug candidates. As illustrated in the associated chemical structures, the P1 section of classical peptide-like inhibitors often incorporates this specific aldehyde or derivative structure to mimic the natural substrate of the protease. Furthermore, research into LFA-1/ICAM-1 antagonists for immune regulation also relies heavily on this molecular framework. The ability to synthesize this core structure efficiently is therefore not merely an academic exercise but a strategic imperative for pharmaceutical companies aiming to secure their pipeline against respiratory and intestinal viral pathogens.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
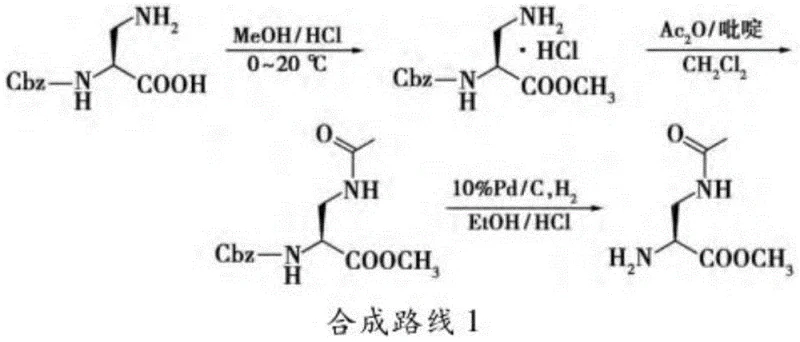
Historically, the synthesis of 2,3-diaminopropionic acid derivatives has been plagued by significant economic and technical hurdles that hinder large-scale adoption. Early approaches, such as the route described by Desolms, relied on (S)-2-benzyloxycarbonylamino-3-aminopropionic acid as a starting material. While chemically feasible, this precursor is prohibitively expensive compared to common bulk chemicals, driving up the overall cost of goods for the final API. Additionally, alternative synthetic strategies involving the Schmidt reaction on aspartic acid require harsh acidic conditions and generate hazardous byproducts, posing severe safety risks in a manufacturing environment.
Other reported methods, including those utilizing aziridine derivatives, introduce further complexities regarding stereocontrol and step count. These routes often necessitate ring-opening reactions followed by multiple protection and deprotection sequences to differentiate the two amino groups. The cumulative yield losses across these numerous steps, combined with the difficulty in sourcing specialized aziridine starting materials in bulk quantities, create a bottleneck for supply chain reliability. Consequently, the industry has lacked a streamlined, cost-effective pathway that balances high stereochemical fidelity with operational simplicity.
The Novel Approach
The methodology outlined in patent CN109251150B represents a paradigm shift by utilizing L-serine, a ubiquitous and inexpensive natural amino acid, as the foundational raw material. This approach bypasses the need for costly protected diamino acid precursors entirely. The synthetic strategy elegantly employs a sequence of esterification, selective trityl (Trt) protection, Mitsunobu substitution, and orthogonal deprotection to construct the target molecule. By leveraging the inherent chirality of serine, the process ensures the correct stereochemistry at the alpha-carbon without the need for resolution, while the beta-position is functionalized efficiently through nucleophilic substitution.
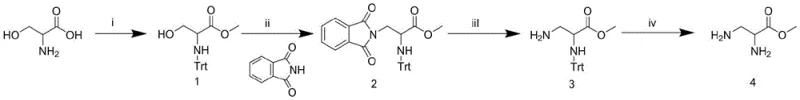
This new route drastically simplifies the manufacturing workflow, reducing the number of unit operations and minimizing waste generation. The use of thionyl chloride for esterification and triphenylchloromethane for amine protection are well-established industrial processes that can be easily scaled. Furthermore, the introduction of the phthalimide group via the Mitsunobu reaction provides a robust handle for the beta-amine that is stable under the conditions required for subsequent transformations. This strategic design results in a process that is not only chemically superior in terms of yield and purity but also economically viable for commercial-scale production.
Mechanistic Insights into Orthogonal Protection and Mitsunobu Substitution
The core of this synthetic success lies in the meticulous application of orthogonal protecting group chemistry coupled with the stereospecific Mitsunobu reaction. Initially, the alpha-amino group of serine methyl ester is protected with a trityl (Trt) group. The steric bulk of the trityl group serves a dual purpose: it prevents unwanted side reactions at the alpha-nitrogen during the subsequent substitution step and enhances the solubility of the intermediate in organic solvents. Following this, the free hydroxyl group at the beta-position is activated by the triphenylphosphine (PPh3) and diisopropyl azodicarboxylate (DIAD) system. This activation converts the poor leaving group (hydroxyl) into a reactive oxyphosphonium intermediate, which is immediately displaced by the phthalimide anion.
This substitution step is critical as it installs the nitrogen functionality at the beta-position with high efficiency, evidenced by the reported yield of 91.89% for Intermediate 2. The mechanism proceeds with inversion of configuration at the beta-carbon; however, since the beta-carbon in serine is achiral (a methylene group), stereochemical issues at this specific center are moot, preserving the integrity of the chiral alpha-center derived from the natural L-serine. The resulting phthalimide group acts as a masked primary amine that is inert to many reaction conditions but can be cleanly removed later. This orthogonality is key to impurity control, as it prevents the formation of polymeric byproducts or cyclic impurities that often plague diamine syntheses.
Impurity control is further refined in the final deprotection stages. The phthalimide group is selectively cleaved using hydrazine hydrate in ethanol, a mild nucleophilic attack that releases the free beta-amine without disturbing the acid-labile trityl group. Finally, the trityl group is removed using ethanolic hydrochloric acid. This sequential deprotection strategy ensures that the final product, methyl 2,3-diaminopropionate hydrochloride, is obtained with minimal contamination from partially deprotected species. The rigorous purification protocols described, including column chromatography and recrystallization steps at intermediate stages, guarantee a final impurity profile that meets the stringent requirements for pharmaceutical intermediates.
How to Synthesize Methyl 2,3-Diaminopropionate Efficiently
The synthesis of this valuable building block follows a logical four-step progression designed for maximum yield and ease of handling. The process begins with the conversion of serine to its methyl ester, followed by the installation of the trityl protecting group. The pivotal third step involves the Mitsunobu coupling with phthalimide, which constructs the carbon-nitrogen bond at the beta-position. The sequence concludes with the sequential removal of the phthalimide and trityl groups to reveal the free diamine functionality. For process chemists looking to implement this route, attention to detail in the stoichiometry of the Mitsunobu reagents and the control of pH during workup is essential to maximize recovery.
- Esterify L-Serine using thionyl chloride in methanol to form methyl serinate hydrochloride.
- Protect the alpha-amino group with Triphenylmethyl chloride (Trt-Cl) to generate Intermediate 1.
- Perform Mitsunobu reaction with phthalimide, PPh3, and DIAD to substitute the beta-hydroxyl group, forming Intermediate 2.
- Remove the phthalimide group with hydrazine hydrate, followed by Trt removal with ethanolic HCl to yield the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this serine-based synthesis offers tangible benefits that extend beyond simple chemistry. The primary advantage is the drastic reduction in raw material costs. By shifting from specialized, protected diamino acid starting materials to commodity-grade serine, the bill of materials is significantly optimized. Serine is produced in massive quantities globally for the food and feed industries, ensuring a stable and competitive pricing structure that is less susceptible to the volatility seen in niche fine chemical markets. This stability allows for more accurate long-term budgeting and cost forecasting for API manufacturing projects.
- Cost Reduction in Manufacturing: The elimination of expensive chiral precursors directly lowers the entry cost for production. Furthermore, the high yields observed in the key coupling steps (exceeding 90% in the Mitsunobu step) minimize material waste and reduce the burden on waste treatment facilities. The avoidance of cryogenic conditions and the use of standard solvents like methanol, ethanol, and dichloromethane mean that existing reactor infrastructure can be utilized without the need for capital-intensive retrofitting, leading to substantial operational expenditure savings.
- Enhanced Supply Chain Reliability: Relying on a synthesis route anchored by serine mitigates the risk of supply disruptions associated with single-source specialty reagents. The reagents used, such as triphenylphosphine, DIAD, and trityl chloride, are widely available from multiple global suppliers, creating a resilient supply network. This diversification ensures that production schedules remain uninterrupted even if one supplier faces logistical challenges, thereby securing the continuity of supply for downstream drug substance manufacturing.
- Scalability and Environmental Compliance: The process operates under mild conditions, largely avoiding the extreme temperatures and pressures that complicate scale-up. The use of hydrazine hydrate and thionyl chloride requires standard safety protocols but does not introduce exotic hazards that would delay regulatory approval for new manufacturing sites. Additionally, the streamlined nature of the synthesis reduces the overall solvent consumption per kilogram of product, aligning with modern green chemistry initiatives and reducing the environmental footprint of the manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of methyl 2,3-diaminopropionate. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a clear picture of the technology's capabilities. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this intermediate into their own synthetic pipelines.
Q: What are the key advantages of using serine as a starting material for this synthesis?
A: Using serine significantly reduces raw material costs compared to traditional protected diamino acid starting materials. It is a naturally abundant amino acid, ensuring supply chain stability and eliminating the need for complex chiral resolution steps often required in racemic syntheses.
Q: How does the orthogonal protection strategy improve purity?
A: The method employs Trt (trityl) for the alpha-amine and Pht (phthalimide) for the beta-amine. These groups can be removed independently under specific conditions (acid for Trt, hydrazine for Pht), minimizing cross-reactivity and side products, thereby ensuring high chemical purity suitable for API production.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the reaction conditions are mild, primarily operating at room temperature or under gentle reflux without requiring cryogenic conditions or hazardous reagents like hydrazoic acid. This operational simplicity facilitates safe scale-up from kilogram to multi-ton manufacturing capacities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Methyl 2,3-Diaminopropionate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of antiviral therapeutics. Our technical team has thoroughly analyzed the serine-based route disclosed in CN109251150B and is fully prepared to execute this chemistry at scale. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from clinical trials to market launch. Our facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of methyl 2,3-diaminopropionate meets the exacting standards required for GMP API synthesis.
We invite you to collaborate with us to optimize your supply chain for peptidomimetic inhibitor production. By leveraging our expertise in orthogonal protection strategies and large-scale Mitsunobu reactions, we can deliver a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can support your drug development goals.
