Revolutionizing Ciprofloxacin Production: A Deep Dive into Aqueous Lewis Acid Catalysis and Commercial Scalability
Revolutionizing Ciprofloxacin Production: A Deep Dive into Aqueous Lewis Acid Catalysis and Commercial Scalability
The pharmaceutical industry is constantly seeking more efficient, environmentally benign, and cost-effective pathways for the synthesis of critical antibiotics like Ciprofloxacin. Patent CN101481381B presents a transformative approach to this challenge by detailing a method for preparing Ciprofloxacin through a piperazification reaction that utilizes a metal Lewis acid catalyst within an aqueous system. This innovation addresses long-standing technical hurdles associated with traditional synthesis routes, specifically targeting the reduction of solvent usage, optimization of piperazine consumption, and acceleration of reaction kinetics. By shifting from harmful organic solvents to a water-based medium enhanced by aluminum or iron Lewis acids, this technology offers a robust framework for high-purity API intermediate production. The structural integrity and therapeutic efficacy of the final product, as depicted in the chemical architecture below, are maintained while significantly streamlining the manufacturing process.
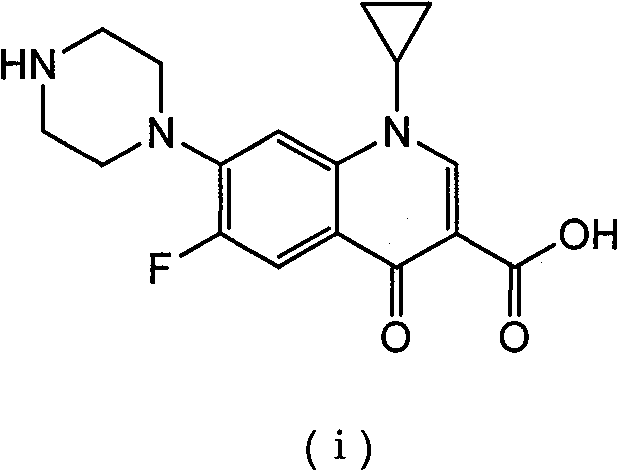
This patent represents a pivotal shift towards green chemistry in the quinolone sector, providing a viable alternative to energy-intensive and waste-generating conventional methods. For R&D directors and process chemists, the introduction of Lewis acid catalysis in water opens new avenues for reaction optimization that were previously unexplored due to solubility concerns. The ability to conduct this nucleophilic substitution efficiently in water not only simplifies the workup procedure but also drastically reduces the environmental footprint of the facility. Furthermore, the method demonstrates remarkable flexibility in terms of catalyst loading and temperature ranges, allowing for fine-tuning based on specific production scale requirements. As we delve deeper into the technical specifics, it becomes evident that this approach is not merely a laboratory curiosity but a commercially viable strategy for modern API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the condensation of fluoroquinolone precursors with piperazine has relied heavily on aprotic organic solvents such as dimethyl sulfoxide (DMSO), dimethylformamide (DMF), or pyridine to facilitate the nucleophilic attack. While these solvents effectively solvate the nucleophile and drive the reaction forward, they introduce severe downstream processing challenges and environmental liabilities. The removal of high-boiling point solvents like DMSO requires significant energy input for distillation or extensive aqueous washing, which generates large volumes of wastewater contaminated with organic residues. Moreover, the use of these solvents often necessitates specialized equipment to handle their toxicity and potential carcinogenicity, increasing capital expenditure and operational safety protocols. In many traditional processes, the reaction times are prolonged, often exceeding several hours at elevated temperatures, which can lead to thermal degradation of sensitive functional groups and the formation of difficult-to-remove impurities. The reliance on excess piperazine to drive conversion further complicates the purification landscape, requiring additional steps to recover and recycle the unreacted amine, thereby inflating the overall cost of goods sold.
The Novel Approach
In stark contrast, the methodology outlined in patent CN101481381B leverages the unique properties of metal Lewis acids to activate the electrophilic center of the quinolone substrate directly within an aqueous environment. This novel approach eliminates the need for toxic aprotic solvents entirely, replacing them with water, which serves as a benign, non-flammable, and inexpensive reaction medium. The addition of aluminum (III) or iron (III) Lewis acids, such as anhydrous aluminum trichloride or ferric chloride hydrate, dramatically enhances the electrophilicity of the carbon atom at the 7-position of the quinolone ring, facilitating a rapid nucleophilic displacement by piperazine even in the presence of water. This catalytic activation allows the reaction to proceed at moderate temperatures ranging from 80°C to 150°C, significantly shortening the reaction cycle compared to uncatalyzed thermal methods. The reaction scheme illustrated below highlights the direct conversion of the chloro-substituted precursor and piperazine into the target Ciprofloxacin molecule, showcasing the elegance and atom economy of this catalytic system.
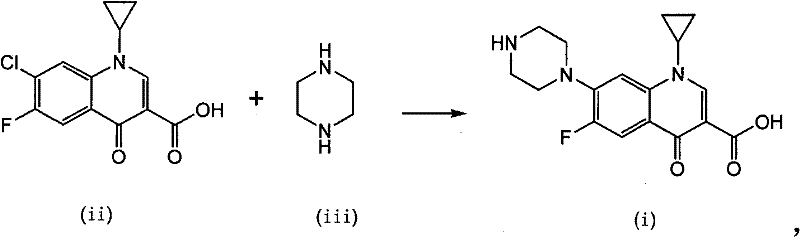
Furthermore, this aqueous Lewis acid system inherently simplifies the isolation process; since the product often precipitates or can be easily induced to crystallize upon pH adjustment, the need for complex solvent exchange or extraction protocols is minimized. The ability to tune the molar ratio of piperazine to the substrate between 1:1 and 10:1, with optimal results often found at lower ratios like 3:1 or 4:1, represents a substantial material saving over traditional methods that might require vast excesses of the amine. By integrating a hot filtration step followed by precise pH-controlled recrystallization, the process effectively segregates the desired product from metal catalyst residues and inorganic salts, yielding a crude product with high purity suitable for subsequent salt formation. This streamlined workflow not only reduces the physical footprint of the manufacturing plant but also aligns perfectly with increasingly stringent global regulations regarding volatile organic compound (VOC) emissions and hazardous waste disposal.
Mechanistic Insights into Lewis Acid-Catalyzed Piperazification
The core of this technological advancement lies in the mechanistic role played by the metal Lewis acid catalyst within the aqueous phase. In a typical bimolecular nucleophilic substitution (SNAr) reaction involving fluoroquinolones, the rate-determining step is often the formation of the Meisenheimer complex, which is highly dependent on the electron-withdrawing nature of the substituents and the solvent's ability to stabilize the transition state. Aluminum (III) and Iron (III) species act as potent Lewis acids by coordinating with the carbonyl oxygen at the 4-position and potentially the leaving group at the 7-position, thereby withdrawing electron density from the aromatic ring. This coordination significantly lowers the energy barrier for the nucleophilic attack by piperazine, making the reaction feasible in water, a solvent that would typically inhibit such reactions through competitive hydrogen bonding or hydrolysis of the substrate. The water molecules in the system likely participate in a proton-shuttle mechanism, facilitating the deprotonation of the attacking piperazine nitrogen and the subsequent expulsion of the leaving group, thus maintaining the catalytic cycle without the need for strong, stoichiometric bases during the initial reaction phase.
Impurity control is another critical aspect where this mechanism offers distinct advantages over thermal-only methods. One of the persistent challenges in Ciprofloxacin synthesis is the competitive substitution at the 6-position fluorine, which leads to structurally similar impurities that are notoriously difficult to separate. The Lewis acid catalyst appears to exhibit a degree of regioselectivity, preferentially activating the 7-position chlorine (or other leaving group) over the 6-position fluorine, likely due to steric and electronic factors enhanced by the metal coordination geometry. Additionally, the post-reaction treatment involving the addition of auxiliary agents such as EDTA, phosphates, or organic acids plays a vital role in sequestering residual metal ions. These auxiliaries form water-soluble complexes with the aluminum or iron catalysts, preventing them from co-precipitating with the product or catalyzing degradation pathways during the workup. The subsequent pH adjustment to the 9-12 range ensures that any acidic byproducts remain in the aqueous phase while the zwitterionic Ciprofloxacin product is prepared for precipitation, followed by a final pH adjustment to 7-8 for optimal crystallization, resulting in a product profile that meets rigorous pharmacopoeial standards with minimal decarboxylation or ethylenediamine analog formation.
How to Synthesize Ciprofloxacin Efficiently
The practical implementation of this synthesis route involves a carefully orchestrated sequence of mixing, heating, and pH manipulation designed to maximize yield while minimizing impurity carryover. The process begins with the precise weighing of the cyclopropanecarboxylic acid derivative and piperazine, which are combined in a reactor equipped with heating and agitation capabilities. The addition of the Lewis acid catalyst, typically at a loading of 1% to 30% molar equivalent relative to the substrate, is a critical step that initiates the catalytic cycle; this can be done prior to or concurrently with the introduction of the aqueous medium depending on the specific solubility characteristics of the catalyst salt chosen. Once the reaction mixture reaches the target temperature window of 80°C to 150°C, the system is maintained under reflux conditions to ensure thorough mixing and consistent thermal energy distribution, with reaction progress monitored via thin-layer chromatography (TLC) to determine the endpoint accurately. Following the completion of the substitution reaction, the mixture undergoes a controlled basification and hot filtration to remove insoluble particulates and catalyst complexes, setting the stage for the final isolation of the high-purity active pharmaceutical ingredient.
- Mix piperazine and cyclopropanecarboxylic acid derivative in a molar ratio of 1-10: 1 with a metal Lewis acid catalyst (1%-30% equivalent) in an aqueous system.
- Heat the reaction mixture to 80-150°C to facilitate the nucleophilic substitution reaction, monitoring progress via TLC.
- Adjust the pH to 9-12 with inorganic base, filter while hot to remove impurities, then adjust filtrate pH to 7-8 to crystallize the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement and supply chain perspective, the adoption of this aqueous Lewis acid catalyzed process offers profound economic and operational benefits that extend far beyond simple yield improvements. The most immediate impact is the drastic reduction in raw material costs associated with solvent procurement; by eliminating the need for expensive, high-purity aprotic solvents like DMSO or DMF, manufacturers can reallocate capital towards higher-value reagents or capacity expansion. The removal of these solvents also obviates the need for complex solvent recovery distillation columns and incineration units, leading to substantial savings in utility costs (steam and electricity) and maintenance overheads. Furthermore, the simplified workup procedure, which relies on aqueous washes and pH adjustments rather than multi-stage extractions, reduces the cycle time per batch, thereby increasing the overall throughput of existing manufacturing assets without the need for significant capital investment in new reactors. This efficiency gain translates directly into a more responsive supply chain capable of meeting fluctuating market demands for this essential antibiotic with greater agility and reliability.
- Cost Reduction in Manufacturing: The elimination of toxic organic solvents removes a major cost center associated with both purchase price and hazardous waste disposal fees. By utilizing water as the primary medium and employing catalytic amounts of inexpensive metal salts like aluminum chloride, the variable cost per kilogram of Ciprofloxacin produced is significantly lowered. Additionally, the optimized stoichiometry allows for a reduction in piperazine usage, a relatively expensive reagent, further driving down the bill of materials. The process avoids the energy-intensive steps of solvent stripping and drying, resulting in lower utility consumption and a smaller carbon footprint, which is increasingly valued in global supply chains.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals such as water, inorganic bases, and common Lewis acids reduces the risk of supply disruptions often associated with specialty solvents that may have limited suppliers or long lead times. The robustness of the aqueous system allows for greater flexibility in sourcing raw materials, as the tolerance for water content and minor impurities is generally higher than in anhydrous organic systems. This resilience ensures continuous production schedules and minimizes the risk of stockouts, providing a stable supply of high-quality API intermediates to downstream formulation partners. The simplified logistics of handling non-hazardous aqueous waste streams also streamline regulatory compliance and transportation arrangements.
- Scalability and Environmental Compliance: The transition to a solvent-free or low-solvent aqueous process inherently scales well from pilot plant to commercial tonnage production, as heat transfer and mixing dynamics in water are well-understood and easier to manage than in viscous organic media. The significant reduction in VOC emissions and hazardous waste generation aligns perfectly with modern environmental, social, and governance (ESG) goals, reducing the regulatory burden and potential liability for the manufacturing site. This green chemistry approach future-proofs the production facility against tightening environmental regulations, ensuring long-term operational viability and enhancing the brand reputation of the supplier as a responsible manufacturer.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method, drawing directly from the experimental data and claims presented in the source documentation. Understanding these nuances is crucial for technical teams evaluating the feasibility of technology transfer or process optimization projects. The answers provided reflect the specific advantages of the Lewis acid catalyzed aqueous system, particularly regarding impurity profiles, catalyst selection, and operational parameters that define the success of this manufacturing route.
Q: What are the primary advantages of using a metal Lewis acid catalyst in Ciprofloxacin synthesis?
A: The use of aluminum (III) or iron (III) Lewis acids significantly accelerates the reaction rate in an aqueous medium, allowing for lower temperatures and shorter reaction times compared to traditional aprotic solvent methods, while simultaneously improving overall yield.
Q: How does this aqueous method address environmental concerns in API manufacturing?
A: By replacing toxic aprotic solvents like DMSO and DMF with water, this method eliminates the need for complex solvent recovery systems and reduces hazardous waste generation, aligning with green chemistry principles and reducing disposal costs.
Q: Can this process effectively control the formation of 6-position fluorine substituted impurities?
A: Yes, the process includes specific pH adjustment steps and the use of auxiliary agents like EDTA or phosphates which help complex metal ions and facilitate the removal of byproducts during the hot filtration and recrystallization stages, ensuring high purity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ciprofloxacin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our team of expert process chemists has extensively evaluated the aqueous Lewis acid catalysis route described in patent CN101481381B and possesses the technical capability to implement this green chemistry approach at scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs and stringent purity specifications, guaranteeing that every batch of Ciprofloxacin or its intermediates meets the highest international standards, including EP, USP, and ChP requirements, regardless of the synthesis route employed.
We invite procurement leaders and R&D directors to engage with our technical procurement team to discuss how this innovative manufacturing process can be tailored to your specific supply chain needs. By partnering with us, you gain access to a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this aqueous catalytic method for your specific volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to delivering high-quality, cost-effective, and sustainable pharmaceutical solutions that drive value for your organization.
