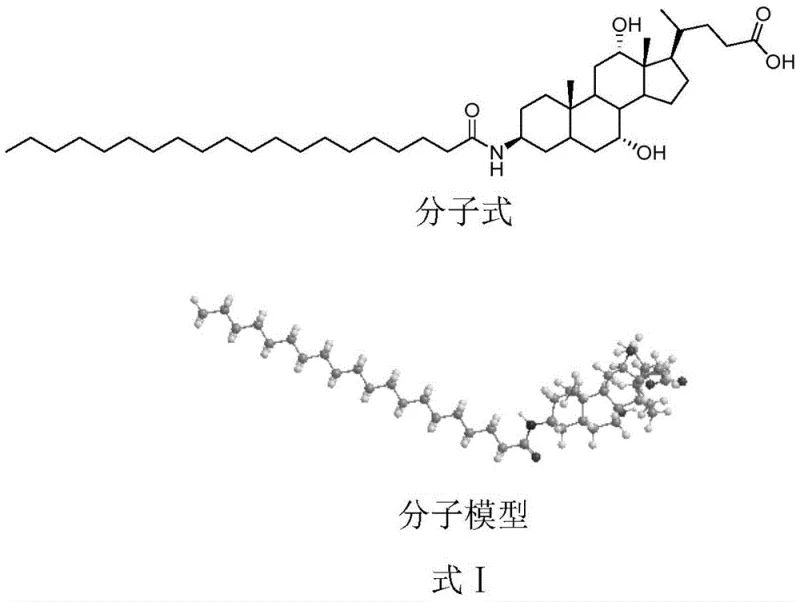
Advanced Manufacturing Strategy for High-Purity Aramchol Intermediates and Commercial Scale-Up
Advanced Manufacturing Strategy for High-Purity Aramchol Intermediates and Commercial Scale-Up
The pharmaceutical industry is constantly seeking robust synthetic routes for complex therapeutic intermediates, particularly for metabolic disorders like Non-Alcoholic Fatty Liver Disease (NAFLD). Patent CN110981933A introduces a groundbreaking methodology for the efficient synthesis of Aramchol, a fatty acid-bile acid conjugate currently in clinical development. This technical disclosure addresses critical bottlenecks in traditional synthesis, specifically focusing on the selective protection of hydroxyl groups to enhance purity and yield. By implementing a strategic protection-deprotection sequence, the process mitigates the severe emulsification phenomena that have historically plagued the purification of cholic acid derivatives. For R&D directors and procurement specialists, this represents a significant opportunity to secure a reliable Aramchol supplier capable of delivering high-purity pharmaceutical intermediates with consistent quality. The following analysis dissects the chemical innovations and their direct implications for commercial manufacturing viability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Aramchol has been hindered by the inherent reactivity of the multiple hydroxyl groups present on the cholic acid scaffold. Conventional routes, such as those involving direct tosylation or multi-step acetylation strategies, often suffer from poor regioselectivity at the 3-position versus the 7 and 12-positions. This lack of selectivity leads to a complex mixture of isomers and byproducts, drastically complicating downstream purification. Furthermore, the presence of free hydrophilic hydroxyl groups during workup stages causes severe emulsification, making phase separation inefficient and leading to significant product loss. Previous methods reported in literature often achieve low overall yields, sometimes below 15%, due to these cumulative inefficiencies. The reliance on hazardous reagents like sodium azide without adequate safety controls in older routes also poses significant operational risks for large-scale manufacturing. Consequently, these limitations have restricted the availability of high-quality Aramchol for clinical and commercial applications.
The Novel Approach
The methodology disclosed in CN110981933A offers a sophisticated solution by introducing a specific hydroxyl protection strategy after the initial 3-position activation. By selectively protecting the 7 and 12-hydroxyl groups as esters or ethers prior to the critical amidation step, the process effectively masks these reactive sites. This modification prevents unwanted side reactions with arachidoyl chloride, ensuring that the acylation occurs exclusively at the intended amino functionality. The introduction of hydrophobic protecting groups significantly alters the physicochemical properties of the intermediates, thereby eliminating the persistent emulsification issues during aqueous workups. This results in a much cleaner reaction profile and simplifies the isolation of intermediates through standard chromatography or crystallization. The strategic design of this route not only improves the chemical yield but also enhances the operational safety and reproducibility required for GMP manufacturing environments.
Mechanistic Insights into Selective Hydroxyl Protection and Amidation
The core innovation lies in the precise manipulation of the cholic acid stereocenters and functional groups. The process begins with the esterification of the carboxylic acid to prevent interference during sulfonylation. Subsequently, a bulky alkylbenzene sulfonyl chloride is employed to selectively target the 3-hydroxyl group, leveraging steric hindrance to avoid reaction at the 7 and 12-positions. Following this activation, the remaining free hydroxyls are protected using acylating agents like acetic anhydride or alkylating agents like methyl iodide under catalytic conditions. This protection step is crucial as it converts polar hydroxyls into less polar esters or ethers, which drastically reduces the molecule's tendency to form stable emulsions at organic-aqueous interfaces. The subsequent nucleophilic substitution with sodium azide and reduction to the amine proceeds with high fidelity due to the stability of the protecting groups under these conditions.
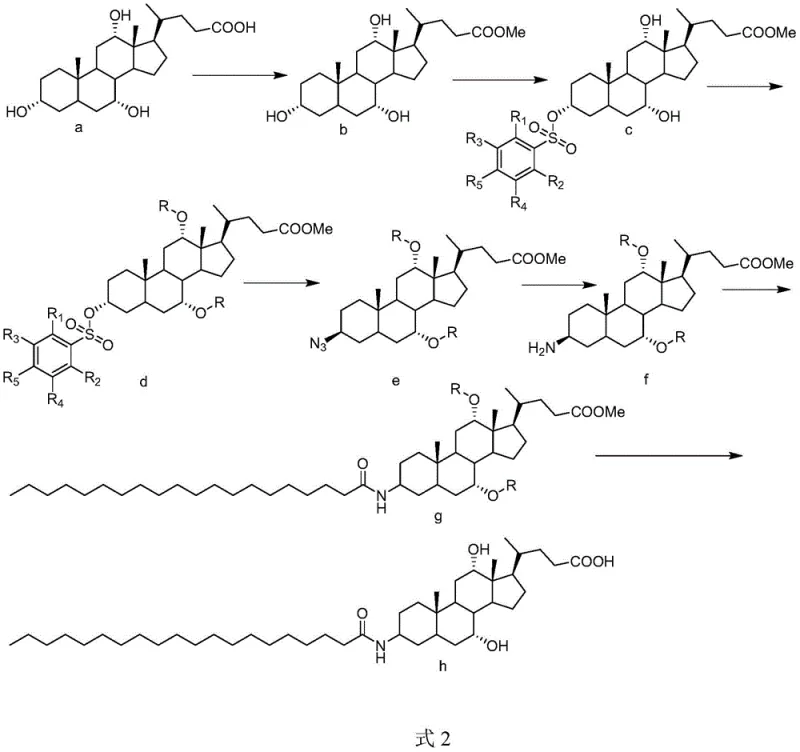
Impurity control is inherently built into this synthetic design by minimizing the formation of regioisomers. In traditional methods, the reaction of arachidoyl chloride with unprotected 7 or 12-hydroxyls generates difficult-to-remove impurities that co-elute with the product. By masking these groups, the new route ensures that the amidation reaction is highly chemoselective for the 3-amino group. The deprotection steps at the end of the synthesis are carefully tuned to remove the protecting groups without hydrolyzing the newly formed amide bond or the steroid backbone. This level of control over the impurity profile is essential for meeting the stringent purity specifications required for pharmaceutical intermediates. The result is a final product with significantly reduced impurity levels, facilitating easier regulatory approval and reducing the burden on quality control laboratories during batch release testing.
How to Synthesize Aramchol Efficiently
The synthesis of Aramchol via this patented route involves a sequence of well-defined chemical transformations that prioritize yield and purity. The process begins with the esterification of cholic acid, followed by selective sulfonylation and subsequent protection of the remaining hydroxyl groups. Detailed standardized synthesis steps see the guide below. This structured approach allows for precise control over reaction parameters such as temperature, pH, and stoichiometry, which are critical for reproducibility. The use of specific catalysts like DMAP and acid-binding agents ensures that each step proceeds to completion with minimal byproduct formation. Understanding these operational details is key for technical teams evaluating the feasibility of technology transfer.
- Esterify cholic acid with methanol to form methyl cholate, protecting the carboxyl group.
- Selectively sulfonylate the 3-hydroxyl group using alkylbenzene sulfonyl chloride.
- Protect remaining 7 and 12-hydroxyl groups via esterification or etherification before amidation.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this optimized synthesis route offers substantial strategic benefits. The elimination of emulsification issues translates directly into reduced processing time and lower solvent consumption during workup phases. This efficiency gain allows for faster batch turnover and higher throughput in existing manufacturing facilities without the need for significant capital investment in new equipment. Furthermore, the improved selectivity reduces the consumption of expensive starting materials like arachidic acid derivatives, as less material is lost to side reactions. For supply chain heads, this means a more predictable production schedule and reduced risk of batch failures that could disrupt the supply of critical intermediates. The robustness of the process also supports a more stable supply chain, ensuring continuity of supply for downstream drug formulation.
- Cost Reduction in Manufacturing: The streamlined process significantly lowers manufacturing costs by reducing the number of purification steps required. By avoiding the formation of complex impurity profiles, the need for extensive chromatographic purification is minimized, which is often the most costly part of fine chemical synthesis. Additionally, the ability to use simpler aqueous workups instead of complex extraction procedures reduces solvent waste disposal costs. The higher overall yield means that less raw material is needed to produce the same amount of final product, directly impacting the cost of goods sold. These factors combine to create a more economically viable production model that can withstand market price fluctuations.
- Enhanced Supply Chain Reliability: The robustness of the chemical route ensures consistent batch-to-batch quality, which is critical for maintaining regulatory compliance. The use of readily available reagents and standard reaction conditions reduces the risk of supply disruptions for specialized raw materials. This reliability allows procurement managers to negotiate better long-term contracts with confidence, knowing that the supplier can meet volume commitments. The simplified process also reduces the dependency on highly specialized operational expertise, making it easier to scale production across different manufacturing sites if necessary. This flexibility is a key asset in managing global supply chain risks.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction conditions that are safe and controllable at large volumes. The reduction in solvent usage and waste generation aligns with increasingly strict environmental regulations, reducing the environmental footprint of the manufacturing process. The avoidance of hazardous emulsification issues also improves workplace safety by reducing the need for manual intervention during phase separations. This compliance with safety and environmental standards facilitates smoother regulatory audits and approvals. Ultimately, this makes the technology a sustainable choice for long-term commercial production of high-purity pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Aramchol intermediates. These answers are derived directly from the patent data and practical manufacturing experience. They provide clarity on the specific advantages of this synthetic route compared to traditional methods. Understanding these details helps stakeholders make informed decisions about sourcing and process adoption. The information below reflects the current state of the art in Aramchol synthesis technology.
Q: How does this patent solve the emulsification problem in Aramchol synthesis?
A: By protecting the free hydroxyl groups at the 7 and 12 positions, the molecule becomes more hydrophobic during intermediate stages, allowing for easier phase separation and washing without severe emulsification.
Q: What is the overall yield advantage of this new method?
A: The patent reports an overall yield of 56.7% or more, significantly higher than previous routes which suffered from low selectivity and difficult purification steps.
Q: Is this process suitable for industrial scale-up?
A: Yes, the method uses mild conditions, avoids hazardous azide accumulation risks where possible, and simplifies post-treatment, making it viable for kilogram to ton-scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aramchol Supplier
At NINGBO INNO PHARMCHEM, we understand the critical importance of robust synthetic routes for complex molecules like Aramchol. Our team of experts possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch meets the highest industry standards. Our commitment to technical excellence allows us to navigate the complexities of hydroxyl protection and amidation chemistry effectively. Partnering with us means gaining access to a supply chain that is both resilient and capable of adapting to your specific project requirements.
We invite you to discuss your specific needs with our technical procurement team to explore how we can support your development goals. Request a Customized Cost-Saving Analysis to understand the economic benefits of switching to this optimized route. Our team is ready to provide specific COA data and route feasibility assessments tailored to your project timeline. By collaborating closely, we can ensure a seamless transition from laboratory scale to commercial manufacturing. Contact us today to secure a reliable supply of high-quality Aramchol intermediates for your next phase of development.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
