Advanced Catalytic Strategy for High-Purity Azoxystrobin Intermediate Manufacturing and Scale-Up
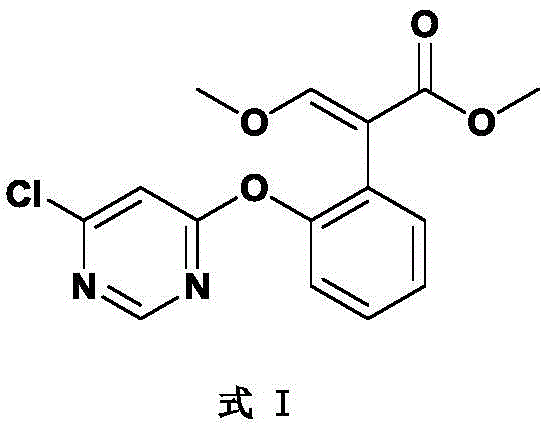
The global demand for high-efficiency fungicides continues to drive innovation in agrochemical intermediate manufacturing, specifically for broad-spectrum agents like azoxystrobin. Patent CN114195723A introduces a transformative preparation method that addresses critical bottlenecks in traditional synthesis routes, utilizing 2-cyanophenol or its salts reacting with a specific pyrimidine derivative known as Formula I. This technical breakthrough shifts the paradigm from cumbersome, high-waste processes to a streamlined, catalytic approach employing N-alkyl cyclic amines in non-polar inert solvents. For R&D directors and process engineers, this represents a significant leap forward in achieving consistent high purity and yield, particularly when transitioning from laboratory discovery to commercial manufacturing. The core innovation lies not just in the catalyst selection, but in the holistic integration of reaction media and post-treatment protocols that facilitate catalyst recovery, thereby aligning synthetic efficiency with stringent environmental standards required by modern regulatory bodies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of azoxystrobin has been plagued by reliance on polar aprotic solvents such as N,N-Dimethylformamide (DMF) coupled with catalysts like triethylenediamine (DABCO). While early literature suggested high theoretical yields, practical industrial application revealed severe drawbacks, including the difficulty of removing high-boiling solvents which necessitates energy-intensive vacuum distillation. Furthermore, the water solubility of DABCO means that the majority of the catalyst is lost to the aqueous waste stream during workup, leading to elevated levels of total nitrogen and Chemical Oxygen Demand (COD) in wastewater, complicating effluent treatment and increasing operational costs. Alternative methods utilizing trimethylamine hydrochloride have shown promise in pilot studies but frequently suffer from significant yield attenuation when amplified to kilogram-scale production, rendering them unreliable for consistent commercial supply chains.
The Novel Approach
The methodology disclosed in CN114195723A fundamentally re-engineers the reaction environment by substituting polar solvents with non-polar inert solvents like toluene or xylene, paired with N-alkyl cyclic amine catalysts such as N-methylpyrrolidine or N-methylpiperidine. This strategic shift ensures that the reaction solvent is compatible with the post-treatment isolation steps, allowing for simple phase separation rather than complex distillation sequences. As illustrated in the reaction scheme below, the coupling of Formula I with 2-cyanophenol proceeds smoothly under these conditions, maintaining yields exceeding 98% even at larger scales. The compatibility of the catalyst with the organic phase enables a novel recovery loop where the catalyst can be extracted, regenerated, and reused, effectively closing the material loop and minimizing raw material consumption.
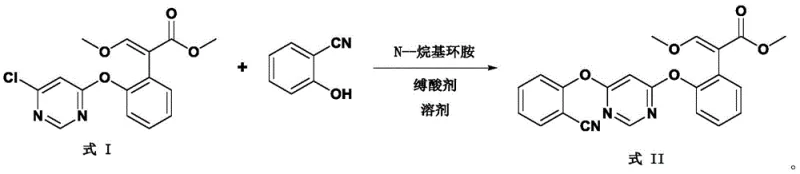
Mechanistic Insights into N-Alkyl Cyclic Amine Catalyzed Coupling
The efficacy of this synthetic route is rooted in the specific nucleophilic properties of the N-alkyl cyclic amine catalyst, which facilitates the displacement of the chloro group on the pyrimidine ring of Formula I by the phenolic oxygen of 2-cyanophenol. Unlike linear amines or bulky bicyclic amines, the cyclic structure provides an optimal balance of steric accessibility and basicity, promoting the formation of the ether linkage without inducing significant side reactions such as hydrolysis or polymerization. The presence of an acid-binding agent, typically potassium carbonate or sodium carbonate, further drives the equilibrium forward by neutralizing the generated hydrogen chloride, ensuring that the reaction proceeds to near-completion within a moderate temperature range of 60-120°C. This mechanistic precision is crucial for maintaining the integrity of the sensitive acrylate moiety present in the substrate.
Impurity control is another critical aspect where this mechanism excels, as the mild reaction conditions prevent the degradation of the methoxyacrylate pharmacophore which is essential for biological activity. In conventional high-temperature or harsh solvent systems, there is a risk of isomerization or decomposition of the double bond, leading to difficult-to-remove impurities that compromise the final pesticide efficacy. By operating in a non-polar medium with a tailored cyclic amine catalyst, the process minimizes these degradation pathways, resulting in a crude product with a content exceeding 98% prior to final crystallization. This high initial purity simplifies the downstream purification steps, reducing the need for extensive chromatography or multiple recrystallizations, which directly translates to higher overall throughput and reduced solvent usage in the manufacturing plant.
How to Synthesize Azoxystrobin Efficiently
The operational protocol for this synthesis is designed for robustness and ease of execution in standard stainless steel reactors commonly found in fine chemical facilities. The process begins with the sequential charging of toluene, the Formula I intermediate, 2-cyanophenol, the acid-binding agent, and the N-alkyl cyclic amine catalyst into the reaction vessel. Following the reaction period, the workup involves a straightforward aqueous wash to remove inorganic salts, followed by solvent removal and crystallization from methanol to isolate the final white solid product.
- Charge toluene, Formula I compound, 2-cyanophenol, potassium carbonate, and N-alkyl cyclic amine catalyst into a reactor.
- Heat the mixture to 60-120°C and maintain for 5-15 hours to complete the nucleophilic substitution reaction.
- Perform aqueous workup to separate layers, recover catalyst from the organic phase, and crystallize the product from methanol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this N-alkyl cyclic amine catalyzed process offers substantial strategic benefits beyond mere technical performance. The elimination of high-boiling polar solvents like DMF removes the need for specialized, energy-intensive vacuum distillation equipment, thereby lowering capital expenditure and utility costs associated with steam and cooling water. Additionally, the ability to recover and reuse the catalyst creates a circular economy within the production line, insulating the manufacturing cost from fluctuations in raw material pricing and reducing the volume of hazardous waste requiring disposal. These factors collectively contribute to a more resilient and cost-effective supply chain capable of meeting high-volume demands without compromising on margin.
- Cost Reduction in Manufacturing: The replacement of expensive and difficult-to-remove solvents with common non-polar solvents like toluene significantly simplifies the downstream processing workflow. By avoiding the complex vacuum distillation required for DMF removal, the process reduces energy consumption and equipment wear, while the recyclability of the N-alkyl cyclic amine catalyst eliminates the recurring cost of purchasing fresh catalyst for every batch. This qualitative shift in process design leads to a drastic simplification of the operational workflow, allowing for faster batch turnover times and lower overall production costs per kilogram of active ingredient.
- Enhanced Supply Chain Reliability: The raw materials required for this method, including toluene, potassium carbonate, and simple cyclic amines, are commodity chemicals with stable global availability, reducing the risk of supply disruptions compared to specialized catalysts or solvents. Furthermore, the robustness of the reaction at the kilogram scale ensures that production schedules can be met consistently without the yield variability often encountered when scaling up sensitive chemical transformations. This reliability is critical for maintaining continuous supply to formulation plants and ensuring that market demand for the final fungicide product is met without interruption.
- Scalability and Environmental Compliance: From an environmental perspective, the reduction of total nitrogen and COD in wastewater simplifies the effluent treatment process, making it easier for facilities to comply with increasingly strict environmental regulations. The use of non-polar solvents also facilitates easier solvent recovery and recycling, further minimizing the environmental footprint of the manufacturing site. This alignment with green chemistry principles not only mitigates regulatory risk but also enhances the corporate sustainability profile, which is becoming an increasingly important factor in vendor selection for major agrochemical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, highlighting the practical advantages over prior art methods.
Q: Why is the N-alkyl cyclic amine catalyst superior to DABCO for azoxystrobin synthesis?
A: Unlike DABCO, which is water-soluble and difficult to recover from high-boiling DMF, N-alkyl cyclic amines allow for efficient recovery via acid-base extraction in non-polar solvents like toluene, significantly reducing waste and cost.
Q: How does this process improve environmental compliance?
A: By replacing DMF with toluene and enabling catalyst recycling, the process drastically lowers the Chemical Oxygen Demand (COD) and total nitrogen content in wastewater, simplifying effluent treatment.
Q: Is this method suitable for large-scale industrial production?
A: Yes, experimental data confirms that yields remain above 96% even when scaled from gram-level to kilogram-level batches, overcoming the yield drop often seen with previous methods like trimethylamine hydrochloride catalysis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Azoxystrobin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global agrochemical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to full-scale manufacturing is seamless and efficient. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs which utilize state-of-the-art analytical instrumentation to verify every batch against the highest industry standards.
We invite potential partners to engage with our technical procurement team to discuss how this innovative catalytic process can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits specific to your operation. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and efficiency in your azoxystrobin manufacturing operations.
