Advanced Synthesis of Argatroban Intermediates: Overcoming Moisture Barriers for Commercial Scale
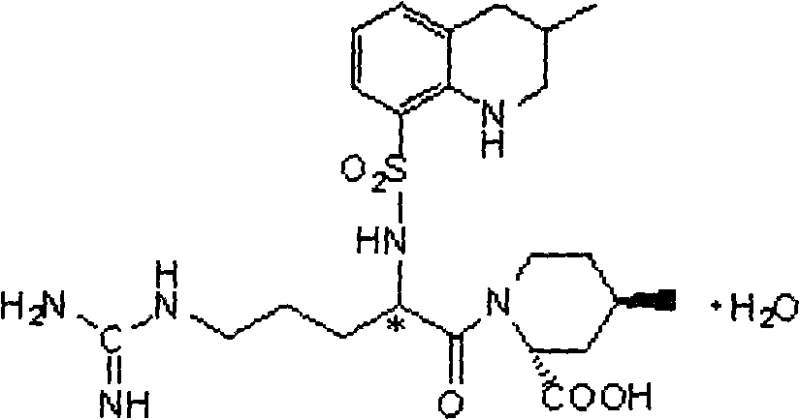
The pharmaceutical landscape for treating acute ischemic cerebral apoplexy has been significantly shaped by the development of direct thrombin inhibitors, among which Argatroban stands out as a critical therapeutic agent. The patent CN101348463A introduces a pivotal advancement in the synthetic methodology for producing N-(3-methyl-8-quinoline sulfonyl)-N-nitryl-L-arginine, a key intermediate in the Argatroban production chain. This technical disclosure addresses a longstanding bottleneck in the industrial manufacturing of this cardiovascular drug: the rigorous control of moisture content in intermediate stages. Traditional synthetic routes often struggle with the physical state of intermediates, frequently resulting in syrups that are difficult to purify and dry without degradation. By implementing a novel workup procedure involving precise pH adjustment, salting-out techniques, and a dual-stage drying protocol, this method ensures water content is reduced to below 0.2 percent, thereby guaranteeing the success of subsequent amidation reactions. This innovation not only enhances the chemical integrity of the product but also aligns with the stringent requirements of large-scale industrial production, offering a reliable pathway for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Argatroban intermediates has been plagued by significant operational challenges that hinder efficient commercial scale-up. Prior art methods, such as those described in earlier patents like EP8746 and US4258192, typically involve the condensation of protected amino acids followed by hydrolysis and hydrogenation. A major drawback in these conventional routes is the physical nature of the intermediate N-(3-methyl-8-quinoline alkylsulfonyl)-N-nitro-L-arginine, which tends to form a viscous syrup rather than a free-flowing solid. To remove residual solvents and moisture from this syrupy mass, manufacturers are forced to employ high-vacuum distillation. This process is not only energy-intensive but also poses severe risks of thermal decomposition due to the prolonged heating times required to achieve anhydrous conditions. Furthermore, the reliance on vacuum tightness introduces a variable that is difficult to control consistently across different batch sizes, leading to potential variability in moisture content that can compromise the yield of the final coupling step.
The Novel Approach
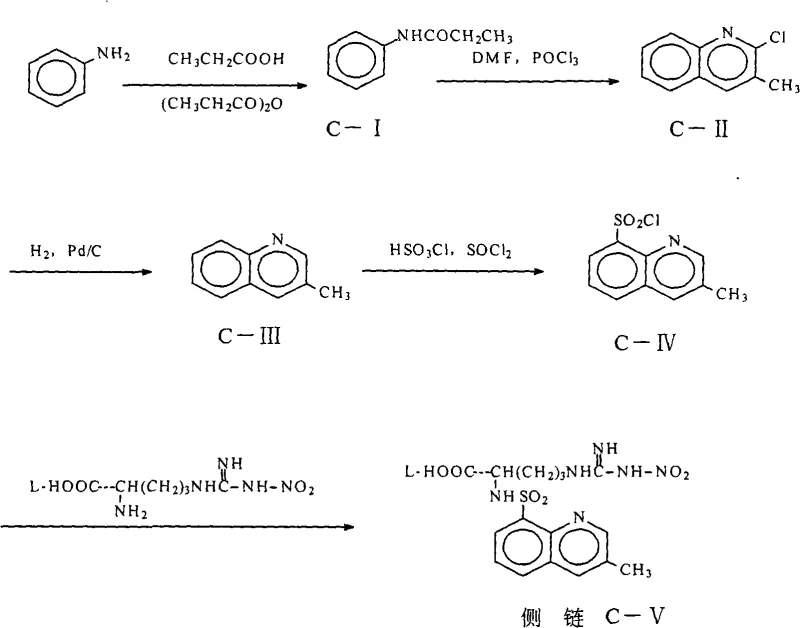
The methodology disclosed in CN101348463A represents a paradigm shift by replacing energy-intensive distillation with a sophisticated extraction and solid-phase drying strategy. Instead of struggling with a syrup, the new process utilizes an aqueous sodium carbonate system for the initial sulfonylation, followed by a strategic acidification to pH 2-3. The addition of saturated inorganic salt solutions, such as sodium chloride or ammonium chloride, induces a 'salting-out' effect that facilitates the separation of the organic phase containing the product. This allows for the isolation of the intermediate in a solution state that is amenable to gentle drying. By employing a two-step drying regimen—first using anhydrous magnesium sulfate or sodium sulfate, followed by treatment with 3A to 5A molecular sieves—the process achieves a water content of less than 0.2 percent without exposing the sensitive molecule to high temperatures. This approach effectively solves the thorny problem of moisture removal, ensuring the intermediate is chemically stable and ready for the critical amidation step.
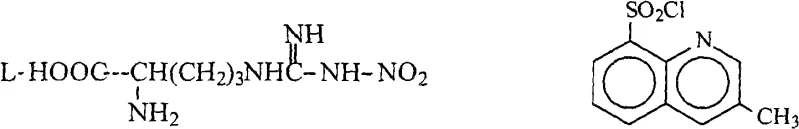
Mechanistic Insights into Sulfonylation and Moisture Control
The core chemical transformation in this synthesis involves the nucleophilic attack of the guanidine nitrogen of N-nitro-L-arginine on the sulfur atom of 3-methyl-8-quinoline sulfonyl chloride. This reaction is conducted in a biphasic system utilizing tetrahydrofuran (THF) and water, buffered by sodium carbonate to neutralize the hydrochloric acid byproduct. The mechanistic elegance of this patent lies not just in the bond formation but in the management of the reaction environment post-synthesis. The presence of water is initially necessary for the solubility of the inorganic base and the amino acid derivative, but it becomes a detrimental impurity in the subsequent step. The downstream reaction involves the activation of the carboxylic acid group using phosphorus oxychloride (POCl3) to couple with (2R,4R)-4-methyl-pipecolic acid ethyl ester. POCl3 is highly moisture-sensitive; even trace amounts of water can hydrolyze the activated acyl chloride species or the POCl3 itself, generating phosphoric acid derivatives that act as impurities and reduce the coupling efficiency. Therefore, the rigorous drying protocol described is not merely a purification step but a critical mechanistic requirement to preserve the electrophilicity of the activated intermediate.
Furthermore, the control of impurities is intrinsically linked to the physical handling of the intermediate. In conventional vacuum distillation, the heat stress can lead to the racemization of the chiral centers or the decomposition of the nitro-guanidine moiety. By maintaining the temperature below 50°C during solvent removal and utilizing molecular sieves for final drying, the novel method preserves the stereochemical integrity of the L-arginine backbone. The use of specific molecular sieves, particularly 4A, provides a thermodynamic sink for water molecules, driving the equilibrium towards an anhydrous state more effectively than simple hygroscopic salts. This ensures that when the intermediate is introduced into the amidation reactor, the stoichiometry of the activating agent remains predictable, leading to consistent yields ranging from 46 percent to 55 percent as demonstrated in the patent examples. This level of control is essential for maintaining the high enantiomeric purity required for the biological activity of the final Argatroban drug substance.
How to Synthesize N-(3-methyl-8-quinoline sulfonyl)-N-nitryl-L-arginine Efficiently
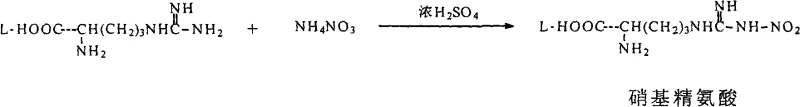
The synthesis of this critical intermediate requires precise adherence to the patented workup procedures to ensure the moisture specifications are met for downstream processing. The process begins with the reaction of N-nitro-L-arginine and 3-methyl-8-quinoline sulfonyl chloride in a THF-water mixture, followed by a series of extraction and drying steps designed to eliminate water without thermal stress. The detailed standardized synthetic steps, including specific molar ratios, temperature controls, and drying durations, are outlined in the guide below to assist process chemists in replicating this robust methodology.
- React N-nitro-L-arginine with 3-methyl-8-quinoline sulfonyl chloride in aqueous sodium carbonate and THF at 25°C.
- Adjust pH to 2-3, add saturated inorganic salt solution to induce phase separation, and extract the organic layer.
- Dry the THF solution first with anhydrous salts, then treat with 4A molecular sieves to achieve water content below 0.2%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers substantial strategic benefits beyond mere chemical yield. The elimination of high-vacuum distillation equipment reduces the capital expenditure required for setting up production lines, as standard reactors and filtration units are sufficient for the salting-out and drying process. This simplification of the unit operations directly translates to cost reduction in pharmaceutical intermediates manufacturing by lowering energy consumption and minimizing the maintenance overhead associated with complex vacuum systems. Moreover, the robustness of the method against moisture variability reduces the risk of batch failures, which is a critical factor in maintaining a steady supply of active pharmaceutical ingredients. By avoiding the thermal degradation associated with distilling syrups, the process also minimizes the generation of hazardous waste and difficult-to-remove impurities, streamlining the quality control workflow and accelerating the release of batches for further processing.
- Cost Reduction in Manufacturing: The shift from vacuum distillation to a salting-out and molecular sieve drying protocol eliminates the need for high-energy evaporation steps and specialized vacuum equipment. This qualitative shift in processing significantly lowers the operational expenditure per kilogram of produced intermediate. Additionally, the reduced risk of thermal decomposition means that raw material utilization is optimized, as fewer batches are lost to degradation, thereby enhancing the overall economic viability of the production campaign without compromising on the purity standards required for regulatory compliance.
- Enhanced Supply Chain Reliability: The simplified process flow, which relies on common reagents like sodium carbonate and sodium chloride rather than exotic catalysts or fragile vacuum setups, ensures that the supply chain is less vulnerable to equipment downtime or specialized part shortages. The ability to consistently achieve water content below 0.2 percent guarantees that the downstream coupling reactions proceed with high reliability, preventing bottlenecks in the final API synthesis. This predictability is crucial for long-term supply agreements, allowing partners to plan their inventory and production schedules with greater confidence and reduced safety stock requirements.
- Scalability and Environmental Compliance: The method is inherently scalable because it avoids the engineering challenges of scaling up high-vacuum distillation for viscous materials, which often suffer from poor heat transfer at larger volumes. The use of aqueous workups and solid desiccants generates waste streams that are easier to treat and dispose of compared to the complex residues from thermal decomposition. This alignment with greener chemistry principles facilitates smoother regulatory approvals and reduces the environmental footprint of the manufacturing site, making it a sustainable choice for long-term commercial production of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, aiming to clarify the operational advantages and chemical rationale behind the process improvements. Understanding these details is vital for technical teams evaluating the feasibility of adopting this route for their own manufacturing pipelines.
Q: Why is moisture control critical in Argatroban intermediate synthesis?
A: Moisture levels exceeding 0.2% can severely inhibit the subsequent amidation reaction with (2R,4R)-4-methyl-pipecolic acid ethyl ester, leading to hydrolysis of activated species and reduced yields.
Q: How does this patent improve upon conventional vacuum distillation methods?
A: Conventional methods rely on vacuum distilling syrupy intermediates, which risks thermal decomposition and requires high-energy equipment. This method uses salting-out and solid-phase drying, which is safer and more scalable.
Q: What represent the key quality indicators for this intermediate?
A: Key indicators include a water content of less than 0.2%, HPLC purity above 83%, and specific concentration metrics to ensure consistent reactivity in downstream coupling steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Argatroban Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global supply of life-saving cardiovascular medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate moisture control and stereoselective requirements of the Argatroban synthesis are met with precision. We operate stringent purity specifications and utilize rigorous QC labs to verify that every batch of N-(3-methyl-8-quinoline sulfonyl)-N-nitryl-L-arginine meets the < 0.2 percent water content benchmark essential for successful downstream coupling. Our commitment to technical excellence ensures that our clients receive intermediates that are not only chemically pure but also physically consistent, facilitating smooth integration into their existing API manufacturing processes.
We invite pharmaceutical partners to engage with our technical procurement team to discuss how this optimized synthetic route can enhance your production efficiency. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your operational scale. We encourage you to contact us to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and reliability in your supply chain for this vital thrombin inhibitor.
