Advanced Chiral Resolution Strategy for Argatroban Precursors Enhancing Purity and Scalability
Advanced Chiral Resolution Strategy for Argatroban Precursors Enhancing Purity and Scalability
The pharmaceutical industry's relentless pursuit of high-purity anticoagulants has placed significant focus on the synthesis of Argatroban, a potent thrombin inhibitor widely used in clinical settings for the prophylaxis and treatment of thrombosis. As detailed in patent CN109734653B, the quality of the final Active Pharmaceutical Ingredient (API) is intrinsically linked to the stereochemical purity of its starting materials, specifically the piperidine derivatives. The presence of isomeric impurities in the early stages of synthesis can propagate through the entire manufacturing chain, leading to costly downstream purification challenges and potential regulatory hurdles. This technical insight report analyzes a breakthrough resolution method that addresses these critical pain points by utilizing L-(-)-dibenzoyltartaric acid to achieve exceptional optical purity.
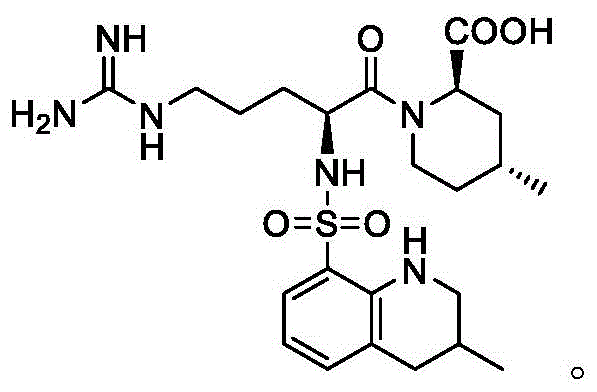
Argatroban, chemically known as (2R,4R)-4-methyl-1-[N-[(3-methyl-1,2,3,4-tetrahydro-8-quinolinyl)sulfonyl]-L-arginyl]-2-piperidinecarboxylic acid, relies heavily on the precise configuration of its piperidine ring. The structural integrity shown in the diagram above underscores why even minor deviations in chirality can render the molecule ineffective or toxic. Consequently, the development of robust methods to isolate specific isomers, such as the (2R,4R) configuration or its precursors, is a paramount concern for R&D directors overseeing API production. The patent data reveals a sophisticated approach to managing these isomeric impurities, moving away from traditional, less efficient resolution techniques toward a more streamlined and high-yielding process.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of key piperidine intermediates like (2R,4R)-4-methylpiperidine-2-carboxylic acid ethyl ester has been fraught with inefficiencies. Prior art, such as the methods described in Chinese patents CN101712645A and CN108047125A, predominantly relied on L-tartaric acid as the resolving agent. While L-tartaric acid is a common chiral pool material, its application in this specific context often results in suboptimal separation factors, leading to lower total yields and optical purities that barely meet the stringent requirements for modern pharmaceutical manufacturing. Furthermore, some conventional routes necessitate the use of benzyl ester protection groups to mask functionality during the resolution process. This introduces additional synthetic steps, including protection and deprotection cycles, which not only increase the overall processing time but also require the use of palladium on carbon for hydrogenolysis. The reliance on precious metal catalysts and high-pressure hydrogenation equipment significantly escalates capital expenditure and operational complexity, creating bottlenecks for supply chain managers aiming for cost-effective scale-up.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN109734653B introduces a paradigm shift by employing L-(-)-dibenzoyltartaric acid (L-DBTA) as the primary resolving agent. This strategic substitution leverages the bulky benzoyl moieties attached to the tartaric acid backbone to create substantial steric differentiation between the diastereomeric salts formed with the racemic amine. This enhanced steric hindrance facilitates a much sharper distinction in solubility properties between the target isomer salt and the unwanted impurity salts. The result is a direct resolution process that bypasses the need for cumbersome protection-deprotection sequences. By operating in a optimized solvent system of isopropanol and acetonitrile, the process achieves a total resolution yield of approximately 45.2% with an impressive optical purity reaching 99.2%. This represents a significant leap forward in process chemistry, offering a direct route to high-quality intermediates that simplifies the overall synthetic tree and reduces the burden on downstream purification units.
Mechanistic Insights into L-DBTA Mediated Diastereomeric Salt Formation
The core of this technological advancement lies in the thermodynamics and kinetics of diastereomeric salt crystallization. When the racemic 4-methyl-2-piperidinecarboxylic acid ethyl ester interacts with L-(-)-dibenzoyltartaric acid, two distinct diastereomeric salts are formed in situ. The molecular recognition between the chiral acid and the chiral base is highly specific; the spatial arrangement of the (2R,4S) isomer allows for a more stable crystal lattice packing with the L-DBTA anion compared to other isomeric combinations. This stability difference is the driving force behind the selective precipitation. The patent highlights the critical role of the solvent mixture, where the ratio of isopropanol to acetonitrile (approximately 2.5:1) is tuned to maximize the solubility gap between the desired salt and the mother liquor components. This precise solvent engineering ensures that the target salt crystallizes out efficiently while keeping the impurities dissolved, a delicate balance that is often the differentiator between a lab-scale curiosity and a commercially viable process.
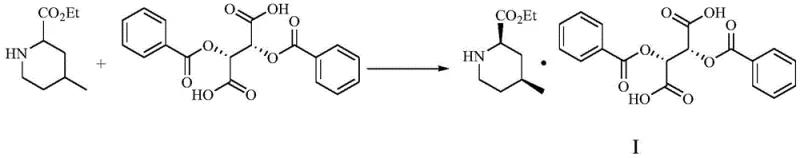
Furthermore, the inclusion of specific additives such as 1-butylpyridinium bromide and L-(-)-dibenzoyl tartaric acid pyridine salt serves a mechanistic purpose beyond simple stoichiometry. These additives function as crystal habit modifiers and nucleation seeds. In crystallization processes, the rate of nucleation versus crystal growth determines the purity of the final solid. Rapid, uncontrolled nucleation can trap impurities within the crystal lattice (occlusion). By introducing these tailored additives, the process controls the supersaturation levels and guides the crystal growth towards a more ordered, pure morphology. Following the isolation of the high-purity salt (Compound I), the final step involves a mild alkaline treatment to liberate the free base. As illustrated in the reaction scheme below, treating the salt with aqueous potassium carbonate in methanol effectively breaks the ionic bond without compromising the sensitive ester functionality, yielding the target (2R,4S)-4-methyl-2-piperidinecarboxylic acid ethyl ester (Compound II) with exceptional chemical and optical integrity.
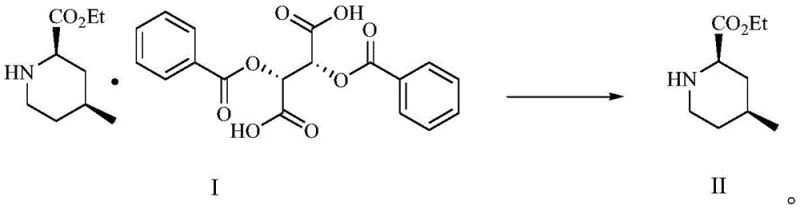
How to Synthesize (2R,4S)-4-methyl-2-piperidinecarboxylic acid ethyl ester Efficiently
The synthesis protocol outlined in the patent provides a robust framework for producing this critical intermediate with high reproducibility. The process is designed to be operationally simple, avoiding extreme temperatures or hazardous reagents that complicate safety protocols in large-scale reactors. The initial salification step is conducted at a moderate temperature of 65-68°C, which ensures complete dissolution of reactants before controlled cooling induces crystallization. This thermal profile is easy to maintain in standard jacketed reactors, making the technology highly transferable from pilot plant to commercial manufacturing scales. The subsequent workup involves standard filtration and washing techniques, followed by a straightforward basification and extraction sequence. For R&D teams looking to implement this route, the key to success lies in strict adherence to the solvent ratios and the timing of additive introduction, as these parameters dictate the crystallization kinetics and final purity profile. The detailed standardized synthesis steps are provided in the guide below.
- React 4-methyl-2-piperidinecarboxylate ethyl ester racemate with L-(-)-dibenzoyltartaric acid in an isopropanol and acetonitrile solvent system at 65-68°C to form the diastereomeric salt.
- Add 1-butylpyridinium bromide and L-(-)-dibenzoyl tartaric acid pyridine salt as additives to induce selective crystallization of the (2R,4S) isomer salt.
- Treat the isolated salt with 10% potassium carbonate solution in methanol to liberate the free base, followed by extraction and purification to obtain the final high-purity intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel resolution method offers tangible strategic benefits that extend beyond mere technical metrics. The primary advantage lies in the drastic simplification of the supply chain and the reduction of dependency on specialized processing equipment. By eliminating the need for benzyl protection groups, the process removes the requirement for palladium on carbon catalysts and high-pressure hydrogenation reactors. This not only lowers the capital expenditure required for facility setup but also mitigates the risks associated with handling pyrophoric catalysts and high-pressure gases, thereby enhancing overall plant safety and reducing insurance and compliance costs.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts and the reduction in synthetic steps directly translate to significant cost savings. Without the need for expensive palladium catalysts and the associated recovery or disposal processes, the raw material cost per kilogram of the intermediate is substantially reduced. Additionally, the higher yield and purity mean less material is lost to waste streams or requires reprocessing, optimizing the overall material balance and improving the economic efficiency of the production line.
- Enhanced Supply Chain Reliability: The raw materials required for this process, such as L-(-)-dibenzoyltartaric acid and common solvents like isopropanol and acetonitrile, are commodity chemicals with stable global supply chains. Unlike specialized chiral ligands or protected intermediates that may have single-source suppliers, these inputs are readily available from multiple vendors. This diversification of the supply base reduces the risk of production stoppages due to raw material shortages, ensuring a more reliable and continuous supply of the critical Argatroban intermediate to downstream API manufacturers.
- Scalability and Environmental Compliance: The process is inherently scalable due to its reliance on crystallization, a unit operation that scales linearly and predictably from grams to tons. The absence of heavy metal catalysts simplifies the environmental profile of the waste stream, making it easier to meet increasingly stringent environmental regulations regarding heavy metal discharge. This "greener" profile not only reduces waste treatment costs but also aligns with the sustainability goals of major pharmaceutical companies, making the supplier a more attractive partner for long-term contracts focused on eco-friendly manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this resolution technology. These answers are derived directly from the experimental data and process descriptions found in the patent literature, providing a clear understanding of the method's capabilities and limitations. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this route for their specific production needs.
Q: Why is L-dibenzoyltartaric acid preferred over L-tartaric acid for this resolution?
A: L-dibenzoyltartaric acid provides superior steric differentiation due to its bulky benzoyl groups, resulting in significantly higher optical purity (up to 99.2%) and improved total yield compared to the conventional L-tartaric acid method which often suffers from low efficiency.
Q: What role do additives like 1-butylpyridinium bromide play in the crystallization process?
A: These additives act as crystal growth modifiers and nucleation agents, helping to control the crystallization kinetics and ensure the selective precipitation of the desired diastereomeric salt while suppressing the co-crystallization of unwanted isomers.
Q: How does this method impact the environmental profile of Argatroban manufacturing?
A: By eliminating the need for benzyl protection groups and subsequent palladium-catalyzed hydrogenation steps required in older methods, this process reduces the use of heavy metals and high-pressure equipment, leading to a greener and safer production workflow.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Argatroban Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex anticoagulants like Argatroban depends on the unwavering quality of every building block in the synthesis chain. Our technical team has thoroughly analyzed the resolution methodology described in CN109734653B and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We understand that achieving the reported 99.2% optical purity at a multi-ton scale requires precise control over crystallization parameters and rigorous quality assurance. Our state-of-the-art facilities are equipped with advanced analytical instrumentation and stringent purity specifications protocols to ensure that every batch of (2R,4S)-4-methyl-2-piperidinecarboxylic acid ethyl ester meets the exacting standards required for GMP API synthesis.
We invite pharmaceutical partners to leverage our expertise in chiral resolution and process optimization to secure a stable supply of high-quality intermediates. By collaborating with us, you gain access to a Customized Cost-Saving Analysis that evaluates how implementing this specific resolution route can optimize your overall manufacturing budget. We encourage you to contact our technical procurement team today to request specific COA data from our pilot batches and discuss detailed route feasibility assessments tailored to your project timelines and volume requirements.
