Optimizing Abiraterone Acetate Manufacturing: A Technical Breakthrough in Purification Efficiency
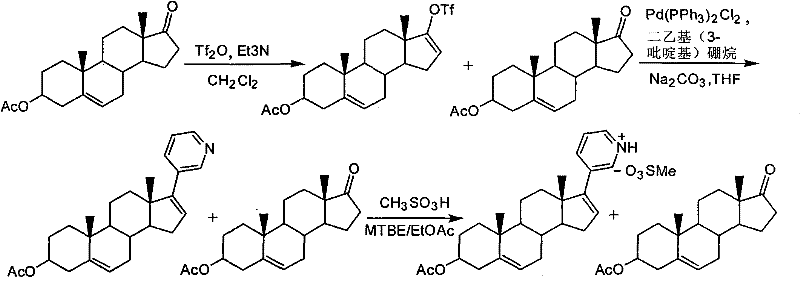
The pharmaceutical industry's relentless pursuit of efficient oncology treatments has placed Abiraterone Acetate at the forefront of prostate cancer therapy manufacturing. As detailed in patent CN102030798A, a significant technological leap has been achieved in the downstream processing of this critical active pharmaceutical ingredient (API). Traditional purification methodologies have long struggled with the physical characteristics of Abiraterone salts, often resulting in operational bottlenecks that hinder large-scale production. This new approach utilizes trifluoromethanesulfonic acid (triflic acid) as a superior salt-forming reagent, fundamentally altering the crystallization behavior of the intermediate. By shifting from conventional mesylate formation to a triflate-based protocol, manufacturers can achieve a dry, granular solid that is exceptionally easy to filter and handle. This innovation not only streamlines the workflow but also ensures that the final product meets stringent purity specifications exceeding 97% without the need for laborious recrystallization cycles. For global supply chains, this represents a pivotal shift towards more robust and scalable manufacturing processes for high-value hormonal inhibitors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the purification of Abiraterone Acetate crude products has relied heavily on the formation of methanesulfonate (mesylate) salts, a technique documented in prior art such as WO2006021777. While chemically feasible, this legacy approach presents severe engineering challenges during the isolation phase. When methanesulfonic acid is introduced to the crude reaction mixture, it tends to generate a stiff, viscous suspension rather than a free-flowing precipitate. This physical state creates a nightmare for filtration operations, as the resulting filter cake is excessively thick and retains significant amounts of mother liquor and impurities. Consequently, the drying process becomes prolonged and inefficient, often leading to the formation of hard, agglomerated blocks that are difficult to process further. Furthermore, the entrapment of impurities within this sticky matrix necessitates additional purification steps, such as recrystallization, to meet pharmaceutical grade standards. These extra unit operations increase solvent consumption, extend batch cycle times, and introduce potential yield losses, making the conventional mesylate route economically and operationally suboptimal for industrial-scale applications.
The Novel Approach
In stark contrast, the methodology disclosed in CN102030798A introduces trifluoromethanesulfonic acid as the salt-forming agent, which dramatically improves the physical properties of the isolated intermediate. Upon reaction with the crude Abiraterone Acetate oil in a mixed solvent system of ethyl acetate and methyl tert-butyl ether (MTBE), the triflic acid induces the rapid precipitation of abiraterone acetate trifluoromethanesulfonate. Unlike its mesylate counterpart, this triflate salt crystallizes as distinct, granular particles that do not adhere to vessel walls or filter media. This granular morphology allows for rapid suction filtration, significantly reducing the time required for solid-liquid separation. The resulting filter cake is loose and dry, facilitating efficient removal of residual solvents and soluble impurities without the need for extensive washing or pressing. This fundamental improvement in solid-state chemistry transforms a previously cumbersome isolation step into a streamlined, high-throughput operation. The ease of handling extends to the subsequent neutralization step, where the high purity of the triflate salt directly translates to a high-purity free base, effectively bypassing the need for complex downstream polishing.
Mechanistic Insights into Triflic Acid-Mediated Salt Formation
The efficacy of this purification strategy lies in the unique physicochemical interactions between the steroid backbone of Abiraterone and the trifluoromethanesulfonate anion. Triflic acid is a superacid with exceptional stability and a large, weakly coordinating anion. When introduced to the basic nitrogen of the pyridine ring in Abiraterone Acetate, it forms a highly stable ionic salt. The bulkiness and charge distribution of the triflate anion appear to disrupt the intermolecular forces that typically lead to the formation of amorphous or gel-like aggregates seen with smaller anions like mesylate. Instead, the steric and electronic properties of the triflate group promote a crystal lattice structure that favors the growth of discrete, well-defined crystals. This crystalline order is crucial for purification, as it inherently excludes impurities from the growing crystal face, a phenomenon known as crystal lattice rejection. The patent data indicates that this mechanism is so effective that the resulting salt achieves a purity of over 97% immediately after filtration and drying. This high level of intrinsic purity suggests that the thermodynamic stability of the triflate crystal form is significantly higher than that of the impurities present in the crude mixture, driving a spontaneous purification effect during the precipitation event.
Furthermore, the control of impurities is enhanced by the specific solvent system employed during the salting-out process. The use of a 1:1 volume ratio of ethyl acetate and MTBE creates a solvent environment with optimized polarity and solubility parameters. At low temperatures, specifically between -5 and 5 degrees Celsius, the solubility of the desired triflate salt is minimized, while the solubility of key impurities, such as unreacted starting materials or side products from the preceding Suzuki coupling reaction, remains high. This differential solubility ensures that upon the addition of triflic acid, only the target molecule precipitates out of the solution. The subsequent neutralization step using aqueous sodium carbonate in dichloromethane is equally critical; it efficiently liberates the free base without degrading the sensitive steroid skeleton. The entire sequence demonstrates a sophisticated understanding of solution chemistry, where pH control, temperature management, and solvent selection are harmonized to maximize both yield and purity. This mechanistic precision allows the process to consistently reduce the number of impurities greater than 1% from over five in the crude material to just one in the final product.
How to Synthesize Abiraterone Acetate Efficiently
Implementing this purification protocol requires precise adherence to the reaction conditions outlined in the patent to ensure reproducibility and safety. The process begins with the dissolution of the crude Abiraterone Acetate oil, typically obtained from a Suzuki coupling reaction involving diethyl(3-pyridyl)borane, into the specified binary solvent system. Temperature control is paramount during the addition of the corrosive triflic acid; maintaining the reaction mixture below 5 degrees Celsius prevents exothermic runaway and ensures the formation of the desired crystal habit. Following the precipitation and filtration of the triflate salt, the material is subjected to a mild basic workup to regenerate the free base. This two-step sequence—salting followed by freeing—is designed to be telescoped or performed in quick succession to minimize hold times and potential degradation. For detailed operational parameters, stoichiometry, and safety precautions, please refer to the standardized synthesis guide below.
- Dissolve crude Abiraterone Acetate oil in a mixed solvent system of ethyl acetate and methyl tert-butyl ether (MTBE) and cool the solution to a low temperature range of -5 to 5 degrees Celsius.
- Slowly add trifluoromethanesulfonic acid (triflic acid) dropwise to the cooled solution to induce the formation of the abiraterone acetate trifluoromethanesulfonate salt, maintaining low temperature during addition.
- Filter the resulting granular solid salt, wash, and dry. Subsequently, neutralize the salt in dichloromethane with aqueous sodium carbonate to liberate the pure free base Abiraterone Acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this triflic acid-based purification method offers substantial strategic advantages beyond mere technical elegance. The primary value driver is the drastic simplification of the manufacturing workflow, which directly correlates to reduced operational expenditures. By eliminating the need for recrystallization and avoiding the use of column chromatography, facilities can significantly lower their consumption of high-grade solvents and silica gel, which are major cost centers in API production. The reduction in unit operations also means less equipment occupancy time, allowing existing manufacturing suites to produce more batches per year without capital expansion. This efficiency gain is critical in a competitive market where speed-to-market for generic versions of blockbuster drugs like Zytiga is essential. Moreover, the robustness of the process reduces the risk of batch failures due to filtration issues, thereby enhancing overall supply reliability.
- Cost Reduction in Manufacturing: The transition to a granular, free-filtering salt eliminates the expensive and time-consuming recrystallization steps often required to purge impurities from sticky mesylate cakes. This reduction in processing intensity leads to significant savings in utility costs, labor hours, and solvent disposal fees. Additionally, the higher yield consistency associated with easier filtration minimizes product loss during isolation, further improving the overall cost-of-goods-sold (COGS) profile for the manufacturer.
- Enhanced Supply Chain Reliability: The physical characteristics of the triflate salt ensure that filtration and drying times are predictable and short, removing a major variable from the production schedule. This predictability allows for tighter planning and shorter lead times, ensuring that downstream formulation teams receive their API intermediates on schedule. The ability to consistently produce high-purity material without rework reduces the risk of supply disruptions caused by out-of-specification batches, fostering a more resilient supply chain.
- Scalability and Environmental Compliance: From an environmental perspective, the process is greener due to the reduced solvent usage and the elimination of silica-based chromatography waste. The granular nature of the solid facilitates safer handling and packaging at scale, reducing dust generation and exposure risks for operators. This alignment with green chemistry principles not only lowers waste treatment costs but also supports corporate sustainability goals, making the supply chain more attractive to environmentally conscious partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this purification technology. They are derived from the specific experimental data and comparative examples provided in the patent documentation, focusing on the practical implications for industrial adoption. Understanding these nuances is vital for technical teams evaluating the feasibility of integrating this route into their existing manufacturing infrastructure.
Q: Why is trifluoromethanesulfonic acid preferred over methanesulfonic acid for Abiraterone purification?
A: Trifluoromethanesulfonic acid forms a granular, non-sticky salt that filters rapidly and dries easily, whereas methanesulfonic acid creates a viscous suspension that clogs filters and traps impurities, requiring additional recrystallization steps.
Q: What purity levels can be achieved with this triflate salt method?
A: The process yields an intermediate triflate salt with purity exceeding 97%, and the final free base Abiraterone Acetate also achieves purity greater than 97% with significantly reduced impurity profiles compared to conventional methods.
Q: Does this method eliminate the need for column chromatography?
A: Yes, the improved crystallization properties of the triflate salt allow for effective purification via simple filtration and washing, removing the need for costly and time-consuming column chromatography typically required for this intermediate.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Abiraterone Acetate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the complexity of steroid synthesis requires a partner with deep technical expertise and a commitment to quality. Our team has extensively analyzed the purification breakthroughs described in CN102030798A and possesses the capability to implement this advanced triflic acid salting protocol at commercial scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits observed in the laboratory are fully realized in the plant. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of Abiraterone Acetate we deliver meets the highest international standards for oncology APIs.
We invite potential partners to engage with our technical procurement team to discuss how this optimized purification route can benefit your specific supply chain needs. By leveraging our expertise, you can access a Customized Cost-Saving Analysis tailored to your production volumes. We encourage you to contact us directly to request specific COA data and route feasibility assessments, allowing you to make informed decisions about securing a reliable, high-quality supply of this critical pharmaceutical intermediate.
