Advanced Synthesis of Abiraterone Acetate: Scalable Solutions for Pharmaceutical Intermediates
The pharmaceutical industry continuously seeks robust and economically viable pathways for the production of critical oncology therapeutics. Patent CN107176965B introduces a transformative methodology for synthesizing Abiraterone Acetate, a pivotal active pharmaceutical ingredient used in the treatment of metastatic castration-resistant prostate cancer. This novel approach addresses significant bottlenecks associated with traditional synthetic routes, specifically targeting the high cost of raw materials and the complexity of purification processes that have historically hindered large-scale manufacturing. By leveraging a unique sequence involving sulfoxide chemistry and hetero-Diels-Alder cycloaddition, this technology offers a streamlined alternative that enhances both yield and operational efficiency. For global supply chain stakeholders, understanding the technical nuances of this patent is essential for securing a reliable pharmaceutical intermediates supplier capable of meeting the growing demand for high-purity Abiraterone Acetate. The following analysis dissects the chemical innovation and its profound implications for commercial production.
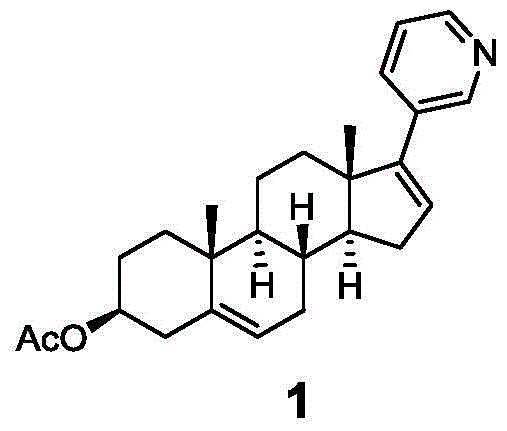
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Abiraterone Acetate has relied heavily on routes starting from dehydroepiandrosterone acetate, which present substantial economic and technical challenges. One prominent method involves the formation of a trifluoromethanesulfonyl derivative followed by coupling with diethyl (3-pyridyl) borane. While this route is conceptually direct, it suffers from the generation of numerous byproducts that are notoriously difficult to separate, complicating the purification workflow and reducing overall throughput. Furthermore, the reliance on expensive reagents such as trifluoromethanesulfonic anhydride and specialized borane compounds drastically inflates the cost of goods sold, making cost reduction in API manufacturing a critical priority for procurement teams. Another existing pathway utilizes hydrazone formation and iodination, which, although it attempts to lower reagent costs, introduces additional synthetic steps that increase the cumulative risk of yield loss and extend the production lead time. These conventional strategies often struggle with scalability, as the accumulation of impurities and the need for rigorous chromatographic purification create bottlenecks that prevent efficient commercial scale-up of complex steroid intermediates.
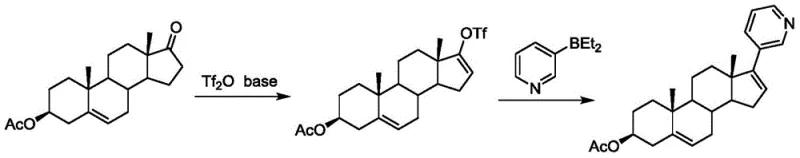
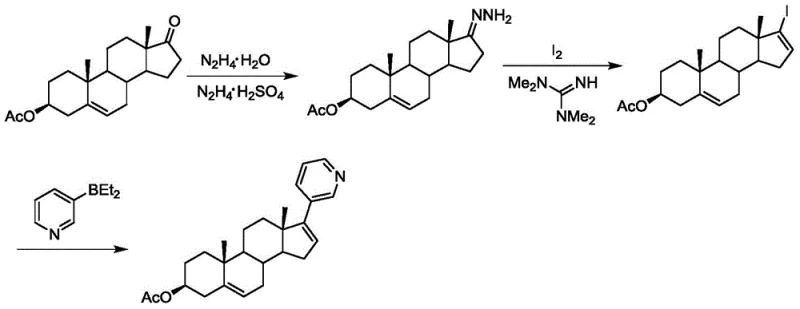
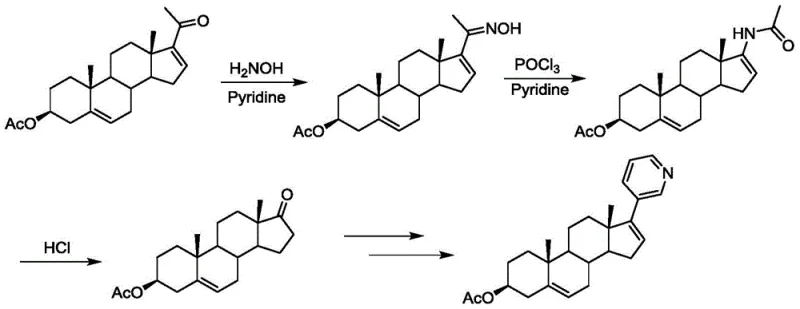
The Novel Approach
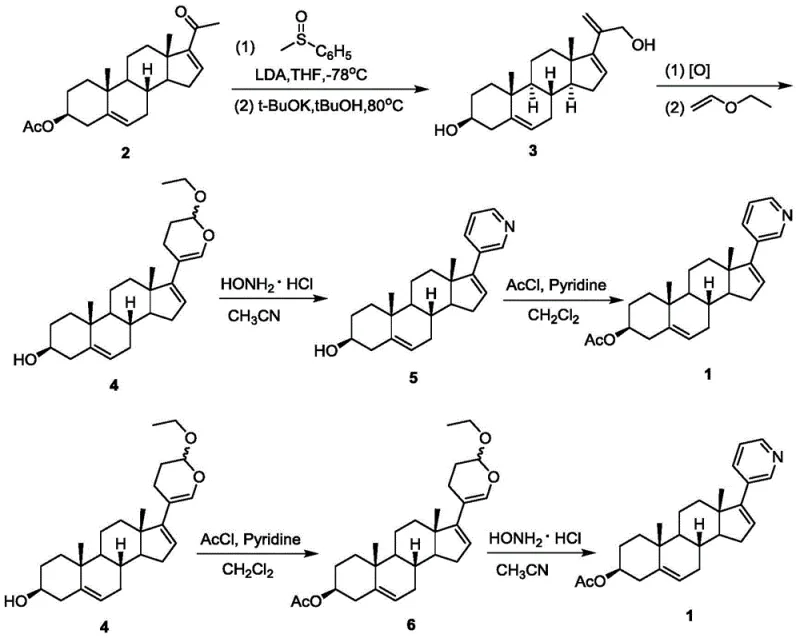
In stark contrast to the limitations of prior art, the methodology disclosed in CN107176965B utilizes dehydropregnenolone acetate as a cost-effective starting material, fundamentally altering the economic landscape of production. This innovative route bypasses the need for expensive coupling reagents by employing a methyl phenyl sulfoxide lithium reagent for nucleophilic addition, followed by a base-mediated rearrangement to generate a key allyl alcohol intermediate. This strategic shift not only reduces the direct material costs but also simplifies the reaction profile, resulting in fewer side reactions and a cleaner crude product profile. The subsequent transformation involves an oxidation step followed immediately by a hetero-Diels-Alder reaction with alkyl vinyl ether, which efficiently constructs the necessary ring system with high stereocontrol. By integrating these steps into a cohesive flow, the process minimizes the need for intermediate isolation and extensive purification, thereby enhancing the overall operational efficiency. This approach represents a significant advancement for any reliable pharmaceutical intermediates supplier aiming to optimize their manufacturing portfolio for high-volume demand.
Mechanistic Insights into Sulfoxide-Mediated Rearrangement and Hetero-Diels-Alder Cyclization
The core chemical innovation of this patent lies in the precise manipulation of steroid functionality through sulfoxide chemistry. The process initiates with the generation of a lithiated methyl phenyl sulfoxide species at cryogenic temperatures, typically around -78°C, using LDA as a strong base. This nucleophile attacks the ketone moiety of the dehydropregnenolone acetate, forming a tertiary alcohol intermediate which subsequently undergoes a syn-elimination and rearrangement under basic conditions to yield the allyl alcohol. This transformation is critical as it installs the necessary carbon framework for the subsequent ring construction while maintaining the integrity of the steroid backbone. The control of reaction parameters, such as temperature and base stoichiometry, is paramount to ensuring high conversion rates and minimizing the formation of regioisomers that could complicate downstream processing. The ability to perform this rearrangement with high fidelity demonstrates the robustness of the chemistry for industrial applications.
Following the formation of the allyl alcohol, the synthesis proceeds through an oxidation step to generate an unstable acrolein intermediate, which is immediately trapped in a hetero-Diels-Alder reaction with ethyl vinyl ether. This cycloaddition is a powerful tool for constructing the dihydropyran ring, which serves as a precursor to the pyridine ring found in the final Abiraterone structure. The use of vinyl ether as a dienophile allows for the introduction of oxygen functionality that is later manipulated during the isomerization step. The subsequent treatment with hydroxylamine hydrochloride facilitates the conversion of the dihydropyran system into the aromatic pyridine ring, completing the construction of the C17 side chain. This sequence exemplifies a high-purity API intermediate strategy where complex molecular architecture is built efficiently without the need for transition metal catalysts, thereby eliminating the risk of heavy metal contamination and the associated costs of scavenging processes.
How to Synthesize Abiraterone Acetate Efficiently
Implementing this novel synthesis route requires careful attention to reaction conditions and reagent quality to maximize yield and purity. The process is designed to be operationally simple, with several steps amenable to telescoping, which reduces solvent usage and processing time. For R&D teams looking to adopt this technology, the key lies in optimizing the addition of the sulfoxide reagent and controlling the exotherm during the rearrangement phase. The subsequent oxidation and cycloaddition steps must be monitored closely to ensure complete conversion before proceeding to isomerization. Detailed standardized synthesis steps see the guide below.
- Perform nucleophilic addition of methyl phenyl sulfoxide to dehydropregnenolone acetate using LDA at -78°C, followed by base-mediated rearrangement to form the allyl alcohol intermediate.
- Oxidize the allyl alcohol to an acrolein compound and immediately conduct a hetero-Diels-Alder reaction with ethyl vinyl ether to generate the dihydropyran intermediate.
- Execute isomerization using hydroxylamine hydrochloride to form the pyridine ring, followed by acetylation to yield the final Abiraterone Acetate product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis route offers compelling advantages for procurement managers and supply chain heads focused on cost efficiency and reliability. The primary driver of value is the substitution of expensive starting materials and reagents with readily available and affordable alternatives, which directly impacts the bottom line. By eliminating the need for precious metal catalysts or specialized borane reagents, the process significantly reduces the raw material expenditure and simplifies the supply chain logistics. Furthermore, the reduction in purification steps translates to lower solvent consumption and waste generation, aligning with environmental compliance goals while reducing disposal costs. These factors collectively contribute to a more resilient supply chain capable of withstanding market fluctuations in reagent pricing.
- Cost Reduction in Manufacturing: The utilization of dehydropregnenolone acetate as a starting material represents a strategic shift away from high-cost precursors like dehydroepiandrosterone acetate derivatives. This change, combined with the avoidance of expensive coupling agents, results in substantial cost savings per kilogram of produced intermediate. The simplified workflow also reduces labor and utility costs associated with extended reaction times and complex work-up procedures, making the overall manufacturing process more economically attractive for large-scale production.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals rather than specialized reagents enhances the security of supply. Procurement teams can source raw materials from multiple vendors, reducing the risk of shortages that often plague specialized chemical supply chains. Additionally, the robustness of the reaction conditions ensures consistent output quality, minimizing the risk of batch failures and ensuring reducing lead time for high-purity oncology intermediates. This reliability is crucial for maintaining continuous production schedules in the pharmaceutical sector.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, with reaction conditions that are easy to control and monitor in large reactors. The reduction in side products and the elimination of heavy metal catalysts simplify waste treatment and reduce the environmental footprint of the manufacturing process. This aligns with increasingly stringent regulatory requirements and supports sustainable manufacturing practices, making the facility more attractive for audits and long-term partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived from the specific technical disclosures and advantageous effects outlined in the patent documentation, providing clarity for stakeholders evaluating this route for potential adoption.
Q: What are the primary cost advantages of this new synthesis route compared to traditional methods?
A: The novel route utilizes dehydropregnenolone acetate, a significantly cheaper starting material compared to the expensive trifluoromethanesulfonic anhydride and diethyl (3-pyridyl) borane required in conventional Suzuki coupling methods. Additionally, the process eliminates complex purification steps, reducing overall operational expenditure.
Q: How does the hetero-Diels-Alder reaction impact the scalability of Abiraterone Acetate production?
A: The hetero-Diels-Alder reaction allows for the direct construction of the pyridine ring system with high regioselectivity. This reduces the number of synthetic steps and minimizes byproduct formation, making the process more robust and easier to scale from kilogram to metric ton quantities without compromising yield.
Q: Is the purity profile of this method suitable for GMP pharmaceutical manufacturing?
A: Yes, the method is designed to minimize side reactions and impurities. The simplified purification process, combined with high-yield steps such as the acetylation reaction, ensures that the final product meets stringent purity specifications required for API intermediate supply chains.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Abiraterone Acetate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes for life-saving medications like Abiraterone Acetate. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. We are committed to delivering high-purity API intermediates that meet stringent purity specifications through our rigorous QC labs and advanced analytical capabilities. Our infrastructure is designed to support the complex chemistry required for steroid synthesis, providing our partners with a secure and reliable source of supply.
We invite pharmaceutical companies and procurement leaders to collaborate with us to optimize their supply chains. By leveraging our expertise in process development, we can help you achieve significant operational efficiencies. Please contact our technical procurement team to request a Customized Cost-Saving Analysis specific to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your commercial goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
