Scalable Manufacturing of Melatonin via Novel 4-Aminobutyric Acid Cyclization Strategy
Scalable Manufacturing of Melatonin via Novel 4-Aminobutyric Acid Cyclization Strategy
The global demand for high-purity neurohormones and sleep-regulating agents continues to surge, driving the need for robust and economically viable synthetic pathways. Patent CN101575311B introduces a groundbreaking method for preparing epiphysin, commonly known as melatonin, utilizing low-cost 4-propalanine (4-aminobutyric acid) as the foundational raw material. This innovative approach circumvents the traditional reliance on expensive indole derivatives, offering a streamlined sequence involving carboxylic esterification, amino acylation, ester reduction, hydroxyl oxidation, and finally, Fischer indole cyclization. By reconstructing the indole core from a linear aliphatic precursor, this technology addresses critical pain points in both process chemistry and supply chain economics. For R&D directors and procurement specialists seeking a reliable melatonin supplier, this patent represents a pivotal shift towards more sustainable and cost-efficient manufacturing paradigms.
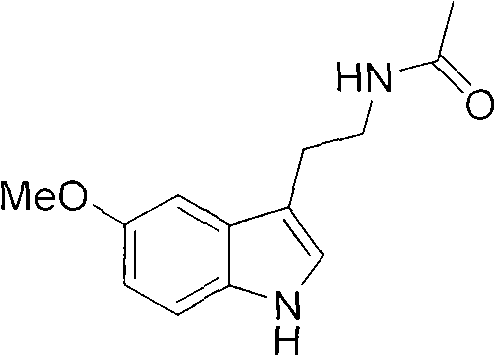
The strategic value of this synthesis lies in its ability to decouple production from volatile indole markets. Conventional literature often cites the use of pre-formed indole rings, which not only commands a premium price but also introduces significant purification challenges. In contrast, the methodology disclosed in CN101575311B constructs the heterocyclic system in the final stages, allowing for rigorous quality control of the linear intermediates. This structural assembly strategy ensures that the final high-purity melatonin meets stringent pharmacopeial standards while minimizing the formation of difficult-to-remove regioisomers often associated with direct substitution on pre-existing indole scaffolds.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of melatonin has been constrained by the necessity of using sophisticated and costly indole derivatives as starting materials. Existing bibliographical information, such as EP 0330625A2 and various Journal of the American Chemical Society publications, predominantly relies on these expensive precursors. Furthermore, traditional routes frequently necessitate the use of lithium aluminum hydride (LiAlH4) as a reductive agent. While effective in laboratory settings, LiAlH4 poses severe safety hazards and operational complexities on a commercial scale, including strict moisture sensitivity and difficult quenching procedures. These factors collectively limit the suitability for industrialized production, inflating operational expenditures and complicating waste management protocols. The reliance on such苛刻 (harsh) reagents often results in lower overall yields due to side reactions and necessitates extensive downstream processing to ensure product safety and purity.
The Novel Approach
The novel approach detailed in the patent fundamentally reimagines the synthetic trajectory by employing 4-aminobutyric acid, a commodity chemical with wide sources and low cost. This method ingeniously bypasses the need for expensive indole derivatives entirely. Instead, it builds the molecular complexity stepwise, starting with simple esterification and acylation reactions that are inherently safer and easier to control. The process avoids the pitfalls of previous patents by eliminating the mandatory use of hazardous reductive agents in favor of more manageable alternatives or optimized conditions. By simplifying the reaction processes and conditions, this route achieves a high total yield, providing a new and superior choice for preparing and producing melatonin. This shift not only reduces the direct cost of goods sold (COGS) but also significantly lowers the barrier to entry for scaling production capacities to meet global demand.
Mechanistic Insights into Fischer Indole Cyclization and Side-Chain Manipulation
The core of this synthetic innovation lies in the precise manipulation of the aliphatic side chain prior to the cyclization event. The process begins with the conversion of 4-aminobutyric acid into its methyl ester hydrochloride using thionyl chloride in methanol, a standard yet highly efficient activation step. Subsequent acylation protects the amine functionality, setting the stage for the critical reduction phase. In this phase, the ester group is reduced to a primary alcohol. As illustrated in the reaction schemes, this transformation is pivotal for generating the necessary oxidation state for the subsequent step. The use of borane reducing agents or optimized hydride systems allows for the selective reduction of the ester without compromising the acetamide protecting group, ensuring high fidelity in the intermediate structure.
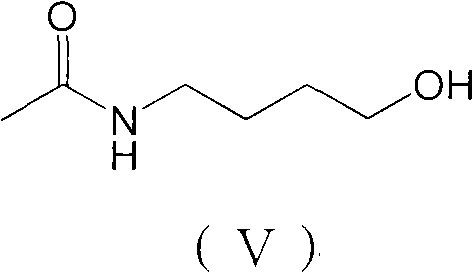
Following the reduction, the resulting alcohol undergoes oxidation to form the corresponding aldehyde, a key electrophile for the final cyclization. This oxidation can be achieved using Jones reagent, Sarrett reagent, or Dess-Martin periodinane, offering flexibility depending on the desired scale and environmental constraints. The generated aldehyde, N-acetyl butyraldehyde, then reacts with p-methoxyphenylhydrazine under acidic conditions. This triggers the Fischer indole synthesis, a classic organic transformation that constructs the indole ring system with high regioselectivity. The mechanism involves the formation of a phenylhydrazone intermediate, which undergoes a [3,3]-sigmatropic rearrangement followed by aromatization and loss of ammonia. This elegant cascade effectively closes the ring, delivering the target melatonin scaffold with the correct substitution pattern inherently built-in, thereby minimizing isomeric impurities.
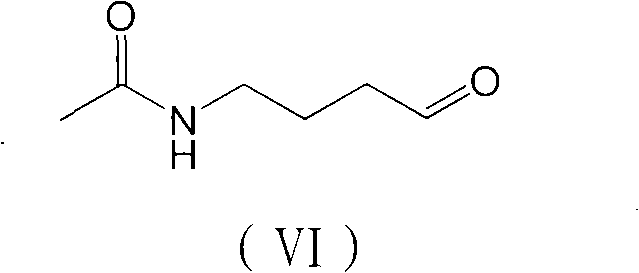
Impurity control is intrinsically managed through this linear construction strategy. By establishing the side-chain functionality before ring closure, the process avoids the random substitution patterns that can occur when modifying a pre-existing indole ring. The intermediates, such as the N-acetyl methyl butyrate and the aldehyde, are chemically distinct and can be purified using standard crystallization or distillation techniques before the final cyclization. This "build-then-cyclize" approach ensures that any impurities carried forward are structurally unrelated to the indole core, making them easier to separate from the final product. Consequently, the final melatonin exhibits a clean impurity profile, which is critical for regulatory approval and patient safety in pharmaceutical applications.
How to Synthesize Melatonin Efficiently
The synthesis of melatonin via this patented route offers a clear pathway for process chemists aiming to optimize production efficiency. The procedure leverages common reagents and straightforward unit operations, making it accessible for implementation in standard multipurpose chemical plants. The initial steps involve simple solution-phase reactions that can be easily monitored by TLC or HPLC, ensuring precise endpoint determination. For a detailed breakdown of the specific reaction parameters, stoichiometry, and workup procedures, please refer to the standardized synthesis guide below.
- Perform carboxylic esterification of 4-aminobutyric acid using methanol and thionyl chloride to obtain the methyl ester hydrochloride.
- Conduct amino acylation using an acetylation reagent and alkaline agents to form N-acetyl methyl butyrate.
- Execute ester reduction followed by hydroxyl oxidation to generate the key aldehyde intermediate, then finalize with Fischer indole cyclization using p-methoxyphenylhydrazine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route translates into tangible strategic benefits beyond mere technical elegance. The primary driver is the substantial reduction in raw material costs achieved by substituting expensive indole derivatives with 4-aminobutyric acid. This commodity chemical is produced in vast quantities for other industries, ensuring a stable and competitive pricing structure that is less susceptible to the supply shocks often seen in niche fine chemical markets. Furthermore, the simplified reaction conditions reduce the dependency on specialized equipment required for handling pyrophoric reagents, thereby lowering capital expenditure and maintenance costs associated with production lines.
- Cost Reduction in Manufacturing: The elimination of expensive starting materials directly impacts the bottom line, allowing for a more competitive pricing strategy in the global marketplace. By avoiding the use of costly indole derivatives and optimizing the reductive steps to potentially avoid hazardous lithium aluminum hydride, the process reduces both material and safety compliance costs. The high total yield reported in the patent further amplifies these savings, as less raw material is wasted per kilogram of final product. This efficiency gain is crucial for maintaining margins in a high-volume, low-margin commodity environment.
- Enhanced Supply Chain Reliability: Sourcing 4-aminobutyric acid is significantly more reliable than procuring specialized indole intermediates, which may have limited suppliers. This diversification of the supply base mitigates the risk of production stoppages due to raw material shortages. Additionally, the use of common solvents and reagents like methanol, thionyl chloride, and acetyl chloride ensures that the supply chain remains resilient even during regional disruptions. The robustness of the synthesis route means that production schedules can be maintained with greater certainty, reducing lead times for high-purity neurohormones and ensuring consistent delivery to downstream customers.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction conditions that are easily transferable from pilot plant to commercial scale. The avoidance of heavy metal catalysts and the potential to use milder oxidants contributes to a greener manufacturing footprint. Simplified purification steps mean less solvent consumption and reduced waste generation, aligning with increasingly stringent environmental regulations. This environmental compliance not only avoids potential fines but also enhances the brand reputation of the manufacturer as a sustainable partner in the pharmaceutical supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this melatonin synthesis technology. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits and chemical feasibility of the route for potential partners and licensees.
Q: What is the primary cost advantage of this melatonin synthesis method?
A: The primary advantage lies in the use of 4-aminobutyric acid (GABA) as the starting material, which is widely sourced and significantly cheaper than the expensive indole derivatives required in conventional methods.
Q: How does this process improve supply chain reliability?
A: By avoiding reliance on scarce or costly indole ring derivatives and hazardous reagents like lithium aluminum hydride where possible, the process utilizes common bulk chemicals, ensuring consistent raw material availability and reduced procurement risk.
Q: Is this method suitable for large-scale commercial production?
A: Yes, the patent highlights simple reaction conditions and high total yield, making it highly suitable for commercial scale-up. The avoidance of complex purification steps associated with traditional routes further enhances its industrial feasibility.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Melatonin Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic route disclosed in CN101575311B for the global melatonin market. As a premier CDMO partner, we possess the technical expertise and infrastructure to translate this patented methodology into commercial reality. Our facilities are equipped to handle the diverse chemical transformations required, from esterification to Fischer indole cyclization, ensuring that every batch meets the highest standards of quality. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, guaranteeing that your supply needs are met with precision and consistency. Our rigorous QC labs and adherence to stringent purity specifications ensure that the melatonin we produce is suitable for the most demanding pharmaceutical and nutraceutical applications.
We invite you to collaborate with us to leverage this cost-effective and scalable technology for your product portfolio. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this route can optimize your procurement budget. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a stable, high-quality supply of melatonin that drives your business forward while minimizing risk and maximizing value.
