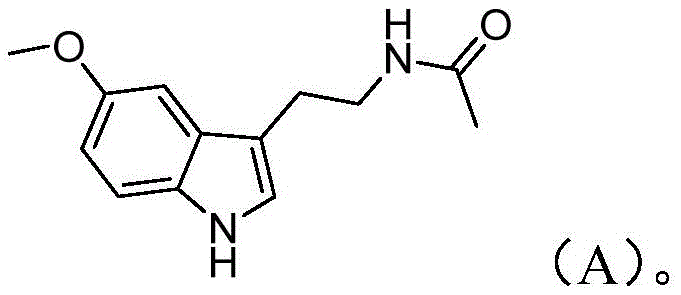
Advanced Melatonin Manufacturing: A Safe, High-Yield Synthetic Route for Global Supply Chains
Advanced Melatonin Manufacturing: A Safe, High-Yield Synthetic Route for Global Supply Chains
The pharmaceutical and nutraceutical industries are constantly seeking robust, scalable, and safe synthetic pathways for high-value active ingredients like Melatonin. A significant breakthrough in this domain is detailed in patent CN110818610B, which outlines a novel preparation method that addresses critical pain points in traditional manufacturing. This innovative approach leverages a strategic sequence of nucleophilic substitution, diazotization ring closure, hydrolysis, and acetylation to achieve exceptional yields while maintaining rigorous safety standards. For R&D directors and procurement specialists, this patent represents a paradigm shift away from hazardous reagents toward a more sustainable and economically viable production model. By utilizing inexpensive and readily available starting materials such as phthalimide and 1,3-dichloropropane, the process not only simplifies the supply chain but also mitigates the risks associated with handling explosive or highly toxic intermediates. The following analysis delves into the technical nuances and commercial implications of this superior synthetic route.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the chemical synthesis of Melatonin has been plagued by significant safety and environmental challenges that hinder efficient commercial scale-up. Traditional methodologies often rely on reagents that pose severe hazards to both personnel and infrastructure. For instance, certain prior art processes utilize borohydride as a reducing agent in conjunction with Jones reagent or Dess-Martin periodinane for oxidation steps. While these methods might offer synthetic utility on a small scale, the involvement of heavy metal reagents like chromium in Jones reagent creates substantial environmental pollution liabilities and necessitates complex waste treatment protocols. Furthermore, other existing routes employ acrylonitrile, a raw material known for its high toxicity and potential explosiveness, which introduces unacceptable safety risks during large-scale hydrogenation steps. These conventional pathways often require harsh reaction conditions, leading to higher energy consumption and increased wear on reactor vessels, ultimately inflating the operational expenditure for manufacturers.
The Novel Approach
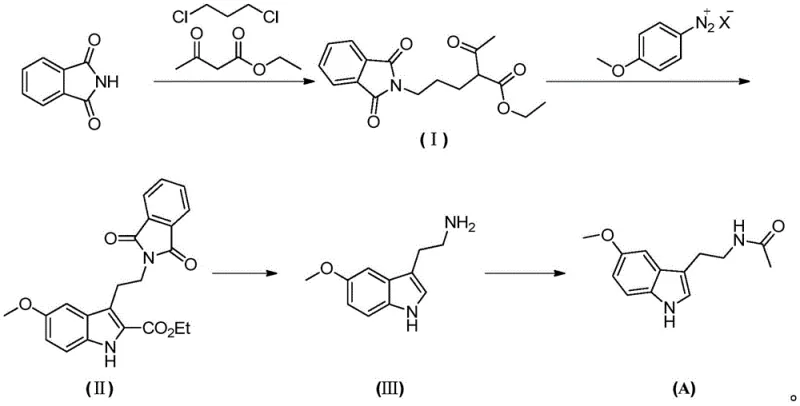
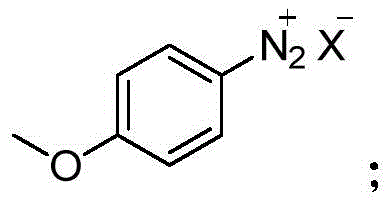
In stark contrast, the methodology disclosed in patent CN110818610B offers a streamlined and safer alternative that fundamentally redesigns the synthetic logic. This novel approach initiates with the alkylation of phthalimide using 1,3-dichloropropane and ethyl acetoacetate, a sequence that proceeds under relatively mild thermal conditions to generate the key intermediate, Compound I. The subsequent ring-closure reaction utilizes a p-methoxyphenyldiazonium salt, facilitating the construction of the indole core without the need for dangerous cyclization agents. This route effectively bypasses the use of explosive cyanides or expensive reducing agents found in older patents. By optimizing reaction parameters such as temperature and solvent systems—utilizing common solvents like acetonitrile and methanol—the process achieves high conversion rates with minimal byproduct formation. This technological advancement not only shortens the overall reaction time but also ensures that the final product meets stringent purity specifications required for pharmaceutical applications.
Mechanistic Insights into Diazotization and Cyclization
The core of this synthetic innovation lies in the precise execution of the diazotization and subsequent cyclization steps, which are critical for forming the indole skeleton of Melatonin. The process begins with the generation of the p-methoxyphenyldiazonium salt, a highly reactive electrophile that drives the ring-closure reaction. In the presence of a base and a mixed solvent system typically comprising methanol and water, Compound I undergoes a nucleophilic attack that triggers the cyclization to form Compound II. The control of pH and temperature during this phase is paramount; the patent specifies a temperature range of -10°C to 80°C, with preferred embodiments operating between 45°C and 60°C to maximize yield while minimizing decomposition. This careful modulation of reaction kinetics ensures that the diazonium species reacts selectively with the enolate equivalent of Compound I, preventing side reactions that could lead to impurities difficult to remove in downstream processing.
Following the formation of the indole ester (Compound II), the mechanism shifts to hydrolysis and decarboxylation to reveal the primary amine functionality. Under alkaline conditions, typically using aqueous sodium hydroxide at elevated temperatures (80-90°C), the ester group is hydrolyzed to a carboxylic acid. Subsequent acidification with concentrated hydrochloric acid induces decarboxylation, a thermal elimination of carbon dioxide that yields the tryptamine derivative, Compound III. This two-step transformation is elegantly simple yet highly effective, avoiding the need for harsh reducing agents often required to convert nitro groups or other precursors to amines in alternative routes. The final acetylation step involves reacting Compound III with acetyl chloride in a solvent like dichloromethane. This nucleophilic acyl substitution is rapid and high-yielding, capping the amine to form the final N-acetyl group characteristic of Melatonin. The entire sequence demonstrates a masterful balance of reactivity and selectivity, ensuring a clean impurity profile.
How to Synthesize Melatonin Efficiently
Implementing this synthesis requires strict adherence to the optimized conditions outlined in the patent to ensure reproducibility and high quality. The process is divided into four distinct stages, each with specific stoichiometric ratios and thermal profiles that have been empirically validated to deliver yields exceeding 90% in individual steps. For R&D teams looking to replicate or scale this process, it is crucial to monitor reaction progress using thin-layer chromatography (TLC) to determine precise endpoints, particularly during the alkylation and cyclization phases. The detailed standardized synthetic steps, including specific molar ratios, solvent volumes, and workup procedures, are provided in the guide below to facilitate immediate technology transfer.
- React phthalimide with 1,3-dichloropropane and ethyl acetoacetate in the presence of a base and sodium iodide to form Compound I.
- Perform a ring-closure reaction between Compound I and p-methoxyphenyldiazonium salt under alkaline conditions to generate the indole core (Compound II).
- Hydrolyze Compound II under basic conditions followed by acidic decarboxylation to yield the amine intermediate, Compound III.
- Acetylate Compound III using acetyl chloride in a suitable solvent to finalize the synthesis of Melatonin.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented route offers compelling advantages that directly address the concerns of procurement managers and supply chain heads regarding cost stability and operational continuity. The shift towards using commodity chemicals as starting materials fundamentally alters the cost structure of Melatonin manufacturing, making it less susceptible to the price volatility associated with specialized fine chemicals. Moreover, the elimination of hazardous reagents simplifies regulatory compliance and reduces the overhead costs related to safety management and waste disposal. These factors combine to create a more resilient supply chain capable of meeting global demand without the bottlenecks often caused by complex or dangerous synthesis requirements.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the utilization of low-cost, widely available raw materials such as phthalimide, 1,3-dichloropropane, and ethyl acetoacetate. Unlike traditional methods that rely on expensive catalysts or oxidants, this route avoids the procurement of high-value reagents that can erode profit margins. Furthermore, the high yields reported in the patent examples indicate a highly efficient atom economy, meaning less raw material is wasted as byproducts. This efficiency translates directly into a lower Cost of Goods Sold (COGS), allowing manufacturers to offer competitive pricing in the global market while maintaining healthy margins. The avoidance of heavy metal catalysts also removes the costly requirement for metal scavenging and purification steps, further streamlining the production budget.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the choice of starting materials, which are bulk commodities with established, multi-vendor supply networks. This diversification reduces the risk of production stoppages due to the shortage of a single specialized precursor. Additionally, the mild reaction conditions—operating primarily at atmospheric pressure and moderate temperatures—mean that the process can be executed in standard glass-lined or stainless steel reactors available in most multipurpose chemical plants. There is no need for specialized high-pressure hydrogenation equipment or cryogenic facilities, which are often capacity-constrained assets in the fine chemical industry. This accessibility ensures that production can be scaled up rapidly to meet surges in demand without lengthy lead times for equipment fabrication or installation.
- Scalability and Environmental Compliance: As environmental regulations become increasingly stringent globally, the "green" credentials of a synthetic route are a major asset. This method avoids the generation of heavy metal waste streams associated with chromium-based oxidants, thereby simplifying wastewater treatment and reducing the environmental footprint of the facility. The solvents used, such as acetonitrile and methanol, are easily recoverable and recyclable through standard distillation processes, promoting a circular economy within the plant. The inherent safety of the process, devoid of explosive intermediates like cyanides, lowers insurance premiums and reduces the administrative burden of safety audits. These factors collectively enhance the long-term sustainability of the manufacturing operation, ensuring uninterrupted supply even in regions with strict environmental enforcement.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Melatonin synthesis route. These answers are derived directly from the technical disclosures and experimental data provided in the patent documentation, offering clarity on feasibility and performance. Understanding these details is essential for stakeholders evaluating the potential for technology adoption or partnership.
Q: What are the primary safety advantages of this Melatonin synthesis route compared to traditional methods?
A: Unlike prior art methods that utilize explosive reagents like cyanide or hazardous oxidants such as Jones reagent, this patented route employs mild reaction conditions and avoids high-risk substances, significantly enhancing operational safety and reducing environmental compliance burdens.
Q: How does the use of phthalimide impact the cost structure of Melatonin production?
A: Phthalimide and 1,3-dichloropropane are commodity chemicals with stable supply chains and low market prices. Utilizing these readily available starting materials eliminates the need for expensive, specialized precursors, thereby driving down the overall Cost of Goods Sold (COGS) for large-scale manufacturing.
Q: Is this synthetic pathway suitable for commercial scale-up?
A: Yes, the process is designed for scalability. The reaction steps operate at moderate temperatures (ranging from 0°C to 110°C) and utilize standard solvents like acetonitrile and methanol, making the transition from laboratory benchtop to multi-ton industrial production feasible without requiring exotic high-pressure or cryogenic equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Melatonin Supplier
The technical robustness of the synthesis route described in patent CN110818610B underscores the immense potential for producing high-quality Melatonin at a commercial scale. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory successes are seamlessly translated into industrial reality. Our state-of-the-art facilities are equipped to handle the specific solvent systems and thermal requirements of this process, while our rigorous QC labs guarantee that every batch meets stringent purity specifications required by global pharmacopeias. We understand that consistency is key in the pharmaceutical supply chain, and our dedicated process engineering team is committed to maintaining the highest standards of quality and reliability.
We invite procurement leaders and R&D directors to explore how this optimized synthesis can enhance your product portfolio. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. Let us demonstrate how our expertise in advanced organic synthesis can drive efficiency and value for your organization, securing a stable supply of this critical bioactive compound for years to come.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
