Advanced Manufacturing of (4R-cis)-6-Substituted-1,3-Dioxane Derivatives for Statin Production
Advanced Manufacturing of (4R-cis)-6-Substituted-1,3-Dioxane Derivatives for Statin Production
The pharmaceutical industry's relentless pursuit of efficient synthetic routes for hypolipidemic agents has brought significant attention to the preparation of key chiral intermediates. Patent CN102180862A introduces a groundbreaking methodology for synthesizing (4R-cis)-6-substituted-2,2-dimethyl-1,3-dioxane-4-tert-butyl acetate, a critical precursor in the production of Rosuvastatin. This novel approach fundamentally shifts the paradigm from traditional, cumbersome condensation reactions to a more elegant Blaise reaction strategy. By leveraging organozinc chemistry and precise hydroboration techniques, the process achieves exceptional stereocontrol and operational simplicity. The target molecule, represented structurally as formula (VIII), serves as a pivotal building block where the integrity of the chiral centers dictates the efficacy of the final API. 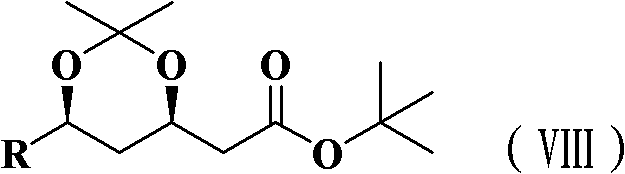
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this dioxane derivative has been plagued by inefficiencies inherent in classical organic transformations. Early routes, such as those reported by G. Wess, relied heavily on (S)-3,4-dihydroxymethyl-butyrate as a starting material but suffered from low overall yields and poor diastereomeric excess (de value). A major bottleneck in these legacy processes was the dependence on lithium diisopropylamide (LDA) for Claisen ester condensation. 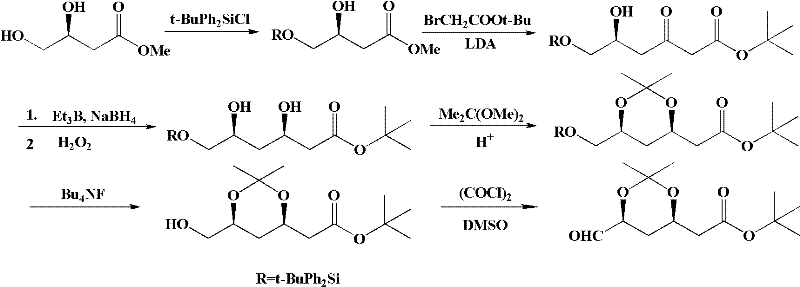
LDA is not only expensive but also requires stringent low-temperature conditions and inert atmospheres, driving up operational expenditures and safety risks. Furthermore, alternative pathways involving Ruthenium-catalyzed hydrogenation followed by Claisen condensation, as seen in US Pat 5399722, introduced complications with by-product formation and insufficient stereocontrol. Other methods attempted to utilize toxic potassium cyanide for chain extension or complex biocatalytic steps that proved difficult to control on a large scale, often resulting in saponification of ester groups or unsatisfactory de values that compromised the purity profile required for pharmaceutical grade intermediates.
The Novel Approach
The methodology disclosed in CN102180862A offers a robust solution by replacing the problematic Claisen condensation with a Blaise reaction. This strategic pivot allows for carbon chain elongation under significantly milder conditions, effectively bypassing the need for cryogenic temperatures and strong, non-nucleophilic bases like LDA. The new route initiates with the protection of the hydroxyl group, followed by the reaction with an organic zinc reagent. 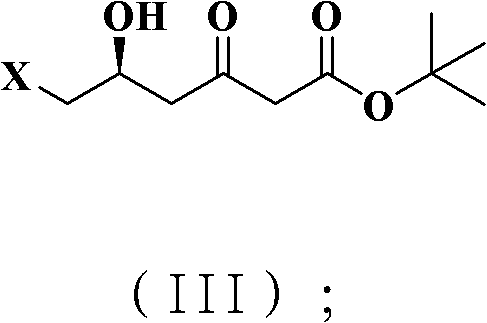
This organozinc-mediated step is far more tolerant of functional groups and proceeds with higher chemoselectivity. Subsequent steps involve a carefully orchestrated hydroboration reduction and acetonide protection, culminating in a streamlined purification process that requires isolation only at the final stage. This reduction in unit operations not only minimizes material loss but also drastically cuts down on solvent usage and waste generation, presenting a clear advantage for green chemistry initiatives in modern pharmaceutical manufacturing.
Mechanistic Insights into Blaise Reaction and Stereoselective Reduction
The core of this technological advancement lies in the mechanistic elegance of the Blaise reaction coupled with stereoselective hydroboration. In the chain extension phase, the organic zinc reagent, generated in situ from activated zinc and tert-butyl bromoacetate, attacks the nitrile functionality of the protected starting material. Unlike Grignard reagents which can be overly reactive and prone to side reactions, the organozinc species offers a balanced nucleophilicity that facilitates the formation of the beta-keto ester intermediate upon acid hydrolysis. This step is crucial as it sets up the carbonyl framework necessary for the subsequent reduction without compromising the existing chiral center at the 4-position.
Following the chain extension, the establishment of the second chiral center is achieved through a highly specific hydroboration reduction. The process utilizes trialkylboranes, such as triethylboron or methoxy-diethylboron, in combination with sodium borohydride at temperatures ranging from -100°C to 20°C. 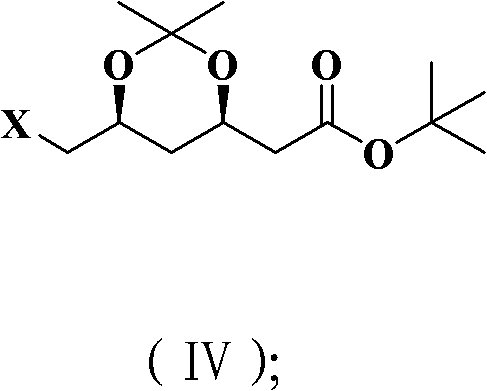
This reagent system creates a cyclic transition state that directs the hydride delivery to the carbonyl face opposite to the existing substituent, thereby ensuring the formation of the desired syn-diol (cis) configuration with exceptional fidelity. The resulting diol is then immediately protected as an acetonide using 2,2-dimethoxypropane under acidic catalysis. This tandem sequence of reduction and protection locks the stereochemistry in place, preventing epimerization during downstream processing and ensuring that the final product maintains a de value exceeding 99%, a critical specification for regulatory compliance in statin production.
How to Synthesize (4R-cis)-6-Substituted-1,3-Dioxane Derivatives Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale execution, emphasizing reproducibility and safety. The process begins with the silylation of the starting hydroxy ester, followed by the critical Blaise coupling and stereoselective reduction steps described above. The final transformation involves either a nucleophilic substitution to install the hydroxymethyl group or an oxidation to yield the aldehyde derivative, depending on the specific downstream requirements for the statin synthesis. For detailed operational parameters, stoichiometry, and workup procedures, please refer to the standardized guide below.
- Protect the hydroxyl group of the starting chloro-hydroxy ester using a silyl protecting agent and catalyst to form the protected nitrile intermediate.
- Perform a Blaise reaction by reacting the protected nitrile with an organic zinc reagent (prepared from activated zinc and tert-butyl bromoacetate), followed by acid hydrolysis to extend the carbon chain.
- Execute a stereoselective hydroboration reduction using triethylboron or similar agents and sodium borohydride at low temperatures (-78°C) to establish the required cis-diol configuration.
- Protect the resulting diol system using 2,2-dimethoxypropane under acidic catalysis to form the 1,3-dioxane ring structure.
- Complete the synthesis by either substituting the terminal halogen with an acetate group followed by hydrolysis to yield the alcohol, or oxidizing directly to the aldehyde derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift to this novel synthetic route represents a significant opportunity for cost optimization and risk mitigation. The elimination of lithium diisopropylamide (LDA) removes a major cost driver associated with specialized reagents and the energy-intensive cooling infrastructure required for its handling. By operating under milder thermal conditions, the process reduces the load on HVAC and refrigeration systems, leading to substantial utility savings over the lifecycle of the product. Furthermore, the avoidance of toxic reagents like potassium cyanide simplifies environmental compliance and waste disposal protocols, reducing the administrative and financial burden associated with hazardous material management.
- Cost Reduction in Manufacturing: The replacement of expensive noble metal catalysts and strong bases with readily available zinc and boron reagents drastically lowers the raw material cost base. The streamlined purification strategy, which consolidates isolation to the final step, minimizes solvent consumption and reduces the number of processing vessels required, thereby lowering both capital expenditure (CapEx) and operating expenditure (OpEx). This efficiency translates directly into a more competitive pricing structure for the finished intermediate without compromising on quality standards.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as zinc powder, tert-butyl bromoacetate, and common solvents like tetrahydrofuran and ethyl acetate ensures a robust and resilient supply chain. Unlike proprietary enzymes or scarce transition metal catalysts which can be subject to geopolitical supply shocks, these raw materials are globally sourced and widely available. This diversification of the supply base mitigates the risk of production stoppages due to raw material shortages, ensuring consistent delivery schedules for downstream API manufacturers.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, avoiding the pitfalls of biocatalytic methods that often face mass transfer limitations in large reactors. The high atom economy of the Blaise reaction and the high stereoselectivity of the reduction step result in minimal by-product formation, simplifying effluent treatment. This aligns with increasingly stringent global environmental regulations, allowing manufacturers to maintain continuous production without the risk of regulatory shutdowns due to waste discharge violations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the comparative data and experimental results presented in the patent documentation, focusing on yield improvements, safety profiles, and scalability factors. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer.
Q: Why is the Blaise reaction preferred over traditional Claisen condensation for this intermediate?
A: Traditional Claisen condensation often requires expensive and hazardous reagents like lithium diisopropylamide (LDA) and strict low-temperature environments, leading to significant side reactions and lower yields. The Blaise reaction utilizes milder organozinc chemistry, avoiding these costly reagents and simplifying the carbon chain extension process while maintaining high stereochemical integrity.
Q: How does this method improve the diastereomeric excess (de value) compared to prior art?
A: Prior art methods, such as those relying on biocatalysis or non-selective reductions, often struggle to achieve high de values, necessitating difficult purification steps. This patented process employs a specific hydroboration reduction protocol using trialkylboranes and borohydrides at controlled low temperatures, which ensures excellent stereoselectivity, achieving de values exceeding 99% without complex chromatographic separation.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the process is specifically designed for industrial scalability. It eliminates the need for toxic reagents like potassium cyanide found in older routes and avoids the operational difficulties of enzymatic processes. Furthermore, the purification strategy is streamlined, requiring purification only at the final step, which significantly reduces solvent consumption and processing time, making it highly viable for metric-ton scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (4R-cis)-6-Substituted-1,3-Dioxane Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to a more efficient synthetic route requires a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a commercial setting. Our facilities are equipped with state-of-the-art cryogenic reactors and rigorous QC labs capable of meeting stringent purity specifications, including the high de values required for next-generation statin intermediates.
We invite global pharmaceutical partners to collaborate with us to leverage this advanced technology for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out for specific COA data and route feasibility assessments to determine how this optimized process can enhance your production efficiency and reduce your overall cost of goods sold.
