Advanced Manufacturing of Rosuvastatin Intermediates Using Novel Blaise Reaction Technology
The pharmaceutical industry continuously seeks robust and scalable pathways for the production of high-value statin intermediates, specifically for blockbuster drugs like Rosuvastatin. Patent CN102180862B, published in August 2013, introduces a groundbreaking method for preparing (4R-cis)-6-substituted-2,2-dimethyl-1,3-dioxane-4-tert-butyl acetate, a critical chiral building block. This technology addresses long-standing inefficiencies in prior art by leveraging a Blaise reaction strategy that fundamentally alters the economic and operational landscape of statin intermediate manufacturing. By replacing traditional condensation methods with organozinc chemistry, the patent outlines a route that operates under significantly milder conditions while achieving superior stereochemical outcomes. For global supply chain leaders and R&D directors, this represents a pivotal shift towards more sustainable and cost-effective production methodologies that do not compromise on the stringent purity standards required for active pharmaceutical ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this specific dioxane scaffold has been plagued by complex multi-step sequences that rely on hazardous reagents and difficult reaction controls. As illustrated in the prior art routes, such as those utilizing Claisen ester condensation, manufacturers often face low overall yields due to competing side reactions and the formation of unwanted diastereomers. 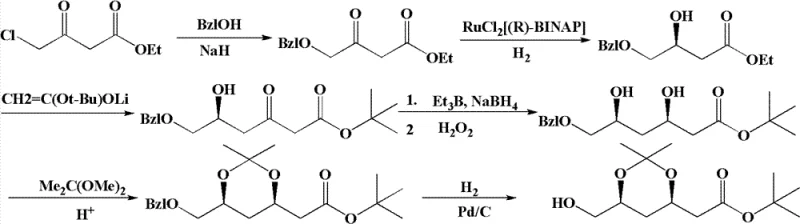 Furthermore, alternative pathways described in the background technology frequently necessitate the use of extremely expensive bases like lithium diisopropylamide (LDA) which require cryogenic temperatures below -70°C to maintain selectivity, driving up energy consumption and capital expenditure for specialized cooling infrastructure. Other reported methods involve the use of highly toxic potassium cyanide for chain extension or rely on unpredictable biocatalytic reductions that are notoriously difficult to scale reproducibly in a large-scale chemical plant environment, creating significant bottlenecks for reliable commercial supply.
Furthermore, alternative pathways described in the background technology frequently necessitate the use of extremely expensive bases like lithium diisopropylamide (LDA) which require cryogenic temperatures below -70°C to maintain selectivity, driving up energy consumption and capital expenditure for specialized cooling infrastructure. Other reported methods involve the use of highly toxic potassium cyanide for chain extension or rely on unpredictable biocatalytic reductions that are notoriously difficult to scale reproducibly in a large-scale chemical plant environment, creating significant bottlenecks for reliable commercial supply.
The Novel Approach
The patented methodology offers a transformative solution by substituting the problematic Claisen condensation with a Blaise reaction, which facilitates carbon chain growth under much gentler and more manageable conditions. This innovative approach completely circumvents the need for expensive LDA and the associated low-temperature constraints, allowing the reaction to proceed efficiently at temperatures ranging from -20°C to 120°C depending on the specific step. By utilizing an organic zinc reagent generated in situ from activated zinc and tert-butyl bromoacetate, the process achieves high conversion rates with minimal byproduct formation. Additionally, the route strategically avoids the cumbersome oxidation of alcohol to aldehyde found in other syntheses, instead employing direct substitution or mild oxidation with DMSO. This simplification results in a streamlined process flow that requires purification only at the final stage, drastically reducing solvent usage and processing time compared to the laborious workups required by conventional techniques.
Mechanistic Insights into Blaise Reaction and Stereoselective Reduction
The core chemical innovation lies in the application of the Blaise reaction for the construction of the beta-keto ester framework, followed by a highly stereoselective hydroboration. In the initial phase, the protected chiral nitrile intermediate reacts with the organozinc species to form a zinc enolate, which subsequently undergoes hydrolysis to yield the desired beta-keto ester with high fidelity. 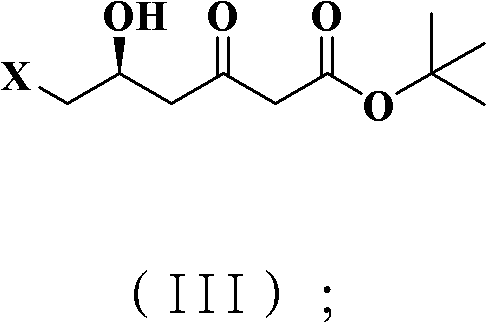 This step is critical as it establishes the carbon skeleton without racemizing the existing chiral center, a common failure point in base-catalyzed condensations. Following this, the ketone functionality is subjected to a hydroboration reduction using triethylboron or similar boron species in the presence of sodium borohydride. This specific reduction protocol is engineered to deliver the hydroxyl group with exceptional diastereoselectivity, ensuring the formation of the syn-diol motif required for the subsequent acetonide protection. The mechanistic precision here ensures that the resulting intermediate possesses the correct (4R-cis) configuration essential for the biological activity of the final statin drug.
This step is critical as it establishes the carbon skeleton without racemizing the existing chiral center, a common failure point in base-catalyzed condensations. Following this, the ketone functionality is subjected to a hydroboration reduction using triethylboron or similar boron species in the presence of sodium borohydride. This specific reduction protocol is engineered to deliver the hydroxyl group with exceptional diastereoselectivity, ensuring the formation of the syn-diol motif required for the subsequent acetonide protection. The mechanistic precision here ensures that the resulting intermediate possesses the correct (4R-cis) configuration essential for the biological activity of the final statin drug.
Impurity control is inherently built into this mechanism through the avoidance of harsh basic conditions that typically trigger epimerization or elimination side reactions. The use of mild Lewis acidic conditions during the acetonide formation step, catalyzed by camphorsulfonic acid, further preserves the stereochemical integrity of the molecule. 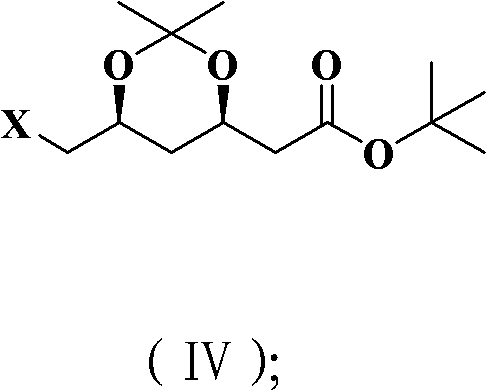 By bypassing the need for strong oxidants or nucleophiles in the final functionalization steps, the process minimizes the generation of structurally related impurities that are difficult to separate. The patent data indicates that the final compounds achieve de values exceeding 99.4%, demonstrating that the mechanistic pathway effectively suppresses the formation of the undesired trans-isomer. This level of purity is achieved without the need for chromatographic purification, relying instead on simple crystallization or extraction, which is a massive advantage for industrial scalability and regulatory compliance regarding residual solvents and heavy metals.
By bypassing the need for strong oxidants or nucleophiles in the final functionalization steps, the process minimizes the generation of structurally related impurities that are difficult to separate. The patent data indicates that the final compounds achieve de values exceeding 99.4%, demonstrating that the mechanistic pathway effectively suppresses the formation of the undesired trans-isomer. This level of purity is achieved without the need for chromatographic purification, relying instead on simple crystallization or extraction, which is a massive advantage for industrial scalability and regulatory compliance regarding residual solvents and heavy metals.
How to Synthesize (4R-cis)-6-substituted-2,2-dimethyl-1,3-dioxane-4-tert-butyl acetate Efficiently
The synthesis protocol outlined in the patent provides a clear, step-by-step guide for producing the target intermediate with high efficiency and reproducibility. The process begins with the protection of the starting hydroxy ester, followed by the key Blaise coupling and stereoselective reduction, culminating in ring closure and functional group adjustment. Each step has been optimized for solvent compatibility and reagent stoichiometry to ensure maximum yield and minimal waste generation. The detailed standardized synthesis steps for implementing this technology in a GMP environment are provided in the guide below.
- Protect the hydroxyl group of the starting chiral ester using a silyl protecting agent to form the nitrile intermediate.
- Perform a Blaise reaction using an activated zinc reagent and tert-butyl bromoacetate to extend the carbon chain under mild conditions.
- Execute a stereoselective hydroboration reduction followed by acetonide protection and final functional group transformation to yield the target dioxane derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers compelling economic and operational benefits that directly impact the bottom line. The elimination of cryogenic conditions and expensive reagents like LDA translates into a substantial reduction in utility costs and raw material expenditures, making the manufacturing process significantly more cost-competitive. The simplified workflow, which reduces the number of isolation and purification steps, leads to shorter cycle times and higher throughput capacity, allowing suppliers to respond more agilely to market demand fluctuations without compromising on quality standards. Furthermore, the avoidance of toxic cyanide reagents enhances the environmental profile of the manufacturing site, reducing the burden on waste treatment facilities and ensuring compliance with increasingly stringent global environmental regulations.
- Cost Reduction in Manufacturing: The replacement of the Claisen condensation with the Blaise reaction eliminates the dependency on costly lithium bases and the energy-intensive infrastructure required for maintaining ultra-low temperatures. This shift allows the reaction to be run in standard stainless steel reactors without specialized cryogenic jackets, leading to drastic capital expenditure savings and lower operating costs per kilogram of product. Additionally, the high yields reported in the patent examples, often exceeding 85% for key steps, mean that less starting material is wasted, further optimizing the cost of goods sold and improving overall margin potential for the final API.
- Enhanced Supply Chain Reliability: By utilizing readily available reagents such as zinc powder and tert-butyl bromoacetate, the supply chain becomes more resilient against shortages of specialized or controlled chemicals. The robustness of the reaction conditions, which tolerate a wider range of temperatures and solvent systems compared to sensitive biocatalytic or organolithium methods, ensures consistent batch-to-batch quality and reduces the risk of production delays caused by failed runs. This reliability is crucial for maintaining continuous supply to downstream API manufacturers who operate on tight just-in-time inventory schedules.
- Scalability and Environmental Compliance: The process is designed with scale-up in mind, avoiding exothermic hazards associated with large-scale LDA additions and minimizing the use of halogenated solvents where possible. The final purification strategy, which relies on simple hydrolysis and extraction rather than complex chromatography, is easily transferable from pilot plant to multi-ton commercial production scales. This scalability, combined with the reduced generation of hazardous waste streams, positions this technology as a sustainable choice for long-term manufacturing partnerships focused on green chemistry principles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation to ensure accuracy and relevance for potential partners. Understanding these details is essential for evaluating the feasibility of integrating this route into existing production portfolios.
Q: How does the Blaise reaction improve upon traditional Claisen condensation for this intermediate?
A: The Blaise reaction replaces the complicated Claisen ester condensation which typically suffers from low yields and numerous side reactions. Crucially, it avoids the need for expensive lithium diisopropylamide (LDA) and cryogenic low-temperature environments, significantly simplifying the operational requirements and reducing raw material costs.
Q: What represents the key stereochemical advantage of this patented process?
A: The process achieves exceptionally high diastereomeric excess (de value), reported up to 99.9% for the hydroxymethyl derivative and 99.4% for the aldehyde derivative. This high stereocontrol is maintained through the specific hydroboration reduction step, eliminating the need for difficult downstream purification to remove diastereomers.
Q: Does this method avoid toxic reagents found in other synthetic routes?
A: Yes, unlike alternative routes described in the background art which utilize highly toxic potassium cyanide (KCN) or require biological enzymatic steps that are difficult to control, this method utilizes safer zinc-mediated chemistry and standard chemical oxidants like DMSO, enhancing workplace safety and environmental compliance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Rosuvastatin Intermediate Supplier
NINGBO INNO PHARMCHEM stands at the forefront of fine chemical manufacturing, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is uniquely qualified to adapt the innovative Blaise reaction methodology described in CN102180862B to meet your specific volume requirements while adhering to stringent purity specifications. We operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to ensure that every batch of (4R-cis)-6-substituted-2,2-dimethyl-1,3-dioxane-4-tert-butyl acetate meets the highest international standards for chiral purity and chemical identity, providing you with a secure and high-quality supply source.
We invite you to contact our technical procurement team to discuss how this advanced synthesis route can optimize your supply chain and reduce costs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this superior manufacturing process. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your R&D and procurement decision-making processes, ensuring a seamless transition to a more efficient and reliable supply partnership.
