Scaling High-Purity Brominated Pyridine Derivatives for Commercial API Manufacturing
Introduction to Advanced Pyridine Functionalization
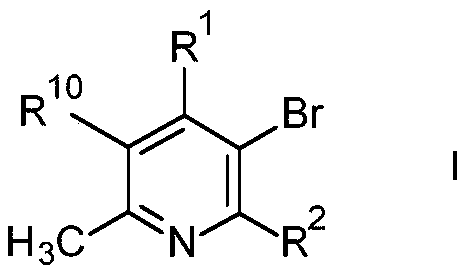
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for more efficient and selective synthetic routes that can withstand the rigors of commercial production. A significant advancement in this domain is detailed in patent CN111630034A, which outlines a novel process for the preparation of specific pyridine derivatives of formula I. This technology addresses long-standing challenges in the halogenation of electron-deficient heterocycles, offering a pathway that balances high selectivity with operational safety. For R&D directors and procurement specialists, understanding the nuances of this bromination methodology is crucial, as it directly impacts the purity profile of downstream active pharmaceutical ingredients. The ability to introduce a bromine atom at a specific position on the pyridine ring without generating excessive poly-halogenated impurities is a key differentiator in modern process chemistry. This report analyzes the technical merits and commercial implications of adopting this refined synthetic strategy for large-scale supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the bromination of pyridine derivatives has relied heavily on the use of elemental bromine in the presence of strong acids such as oleum or fuming sulfuric acid. While effective in terms of reactivity, these conventional methods suffer from significant drawbacks that complicate commercial manufacturing. The use of liquid bromine poses severe safety hazards due to its volatility and corrosivity, requiring specialized containment and handling equipment that increases capital expenditure. Furthermore, reactions involving elemental bromine often proceed with poor regioselectivity, leading to complex mixtures of mono- and di-bromo isomers that are notoriously difficult to separate. This lack of selectivity not only reduces the overall yield of the desired intermediate but also necessitates extensive purification steps, such as multiple recrystallizations or chromatographic separations, which are impractical at multi-ton scales. The slow reaction kinetics observed in some traditional protocols also demand prolonged heating at elevated temperatures, increasing energy consumption and the risk of thermal degradation of sensitive substrates.
The Novel Approach
In contrast, the novel approach described in the patent data utilizes solid brominating agents such as N-bromosuccinimide (NBS) or 1,3-dibromo-5,5-dimethylhydantoin (DBDMH) within a 65% oleum medium. This shift from liquid to solid reagents fundamentally changes the safety and efficiency profile of the operation. The reaction proceeds with remarkable selectivity, favoring the formation of the desired mono-brominated product while suppressing the formation of unwanted di-bromo byproducts. This inherent selectivity simplifies the downstream workup, often allowing for direct extraction and distillation without the need for complex purification trains. The process operates effectively within a moderate temperature range, typically between 80°C and 125°C, which optimizes reaction rates while minimizing thermal stress on the molecular framework. By eliminating the need for handling hazardous liquid bromine, this method aligns better with modern environmental, health, and safety (EHS) standards, making it a more attractive option for sustainable chemical manufacturing.
Mechanistic Insights into DBDMH-Catalyzed Bromination
The mechanistic advantage of using DBDMH or NBS lies in the controlled release of electrophilic bromine species within the acidic medium. In the presence of 65% oleum, these solid reagents generate a steady concentration of reactive brominating agents that interact selectively with the electron-rich positions of the pyridine ring. Unlike the brute-force attack of elemental bromine, which can overwhelm the substrate and lead to over-halogenation, this controlled generation allows for precise functionalization. The reaction mechanism likely involves the protonation of the pyridine nitrogen, which activates the ring towards electrophilic substitution at the specific position dictated by the existing substituents. The steric and electronic properties of the DBDMH molecule further contribute to this selectivity, ensuring that the bromine atom is delivered to the intended site with minimal deviation. This precision is critical for maintaining the integrity of the molecular scaffold, especially when dealing with complex substrates that contain other sensitive functional groups susceptible to oxidation or halogenation.
Impurity control is another cornerstone of this mechanistic pathway. The formation of di-bromo isomers is a common pitfall in pyridine chemistry, often requiring costly removal strategies. The new method mitigates this risk by optimizing the molar ratio of the brominating agent to the substrate, typically keeping it below 1 equivalent to prevent over-reaction. Additionally, the use of inert organic solvents for extraction, such as methyl tert-butyl ether or toluene, ensures that the product is isolated cleanly from the acidic reaction mass. The subsequent purification via distillation under reduced pressure further refines the quality, removing any trace volatile impurities or residual solvents. This rigorous control over the impurity profile is essential for meeting the stringent specifications required by regulatory bodies for pharmaceutical intermediates. It ensures that the material entering the next stage of synthesis is of consistent quality, reducing the risk of batch failures in the production of the final active ingredient.
How to Synthesize Brominated Pyridine Derivatives Efficiently
Implementing this synthesis route requires careful attention to reaction conditions and reagent stoichiometry to maximize efficiency. The process begins with the dissolution of the pyridine precursor in 65% oleum, followed by the controlled addition of the solid brominating agent at low temperatures to manage the initial exotherm. Once the addition is complete, the mixture is heated to the optimal reaction temperature and maintained for a specific duration to ensure complete conversion. Following the reaction, the mixture is quenched into ice water, and the pH is carefully adjusted to alkaline conditions to facilitate the extraction of the free base product. The detailed standardized synthesis steps see the guide below for specific parameters regarding temperature ramps and workup procedures.
- React formula II pyridine compounds with NBS or DBDMH in 65% oleum at controlled temperatures.
- Quench the reaction mixture into ice and adjust pH to alkaline conditions for extraction.
- Purify the crude product via distillation under reduced pressure to achieve high purity specifications.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this bromination technology offers substantial benefits for procurement managers and supply chain heads looking to optimize their sourcing strategies. The primary advantage lies in the significant reduction of processing complexity, which translates directly into lower manufacturing costs. By avoiding the use of hazardous elemental bromine, facilities can reduce their spending on specialized safety equipment and waste disposal services associated with halogen handling. The higher selectivity of the process means that less raw material is wasted on byproduct formation, improving the overall atom economy of the synthesis. This efficiency gain allows for more competitive pricing models without compromising on margin, providing a strategic advantage in negotiations with downstream pharmaceutical clients. Furthermore, the robustness of the reaction conditions ensures consistent batch-to-batch quality, reducing the likelihood of supply disruptions caused by failed production runs.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous elemental bromine reduces both raw material costs and the overhead associated with safety compliance. The simplified purification process, often requiring only extraction and distillation rather than chromatography, significantly lowers solvent consumption and energy usage. These operational efficiencies accumulate to provide substantial cost savings over the lifecycle of the product. Additionally, the higher yields achieved through improved selectivity mean that less starting material is required to produce the same amount of final product, further driving down the cost of goods sold. This economic advantage is critical in the highly competitive market for pharmaceutical intermediates where price pressure is constant.
- Enhanced Supply Chain Reliability: The use of stable solid brominating agents like DBDMH improves supply chain resilience by reducing dependence on volatile liquid chemicals that may face transportation restrictions. The milder reaction conditions also extend the lifespan of reactor equipment by reducing corrosion, leading to less downtime for maintenance and repairs. This reliability ensures a steady flow of materials to customers, minimizing the risk of stockouts that could delay drug development timelines. The ability to scale this process from kilogram to multi-ton quantities without significant re-engineering provides confidence to partners planning long-term commercial campaigns. Consistent availability of high-quality intermediates is a key factor in building trust and securing long-term contracts.
- Scalability and Environmental Compliance: This process is inherently designed for commercial scale-up, with reaction parameters that are easily transferable from pilot plants to full-scale production facilities. The reduced generation of hazardous waste aligns with increasingly strict environmental regulations, lowering the burden of waste treatment and disposal. By minimizing the use of toxic reagents and solvents, the process supports sustainability goals and reduces the environmental footprint of manufacturing operations. This compliance not only avoids potential regulatory fines but also enhances the corporate reputation of the manufacturer as a responsible partner. The ease of scaling ensures that supply can be rapidly increased to meet surges in demand without compromising on quality or safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this bromination technology. They are derived from the specific pain points identified in traditional synthesis methods and the beneficial effects documented in the patent literature. Understanding these details helps stakeholders make informed decisions about integrating this process into their supply chains. The answers reflect the practical realities of running this chemistry at an industrial level, focusing on safety, efficiency, and quality outcomes.
Q: How does this bromination method improve selectivity over conventional Br2 processes?
A: By utilizing solid brominating agents like DBDMH or NBS instead of elemental bromine, the process minimizes the formation of di-bromo isomers and reduces reaction times significantly.
Q: What are the scalability advantages of using DBDMH in oleum?
A: The use of DBDMH allows for better handling safety and more controlled exotherms compared to gaseous or liquid bromine, facilitating safer commercial scale-up in standard reactor setups.
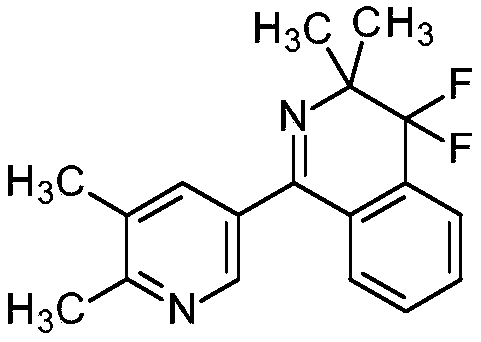
Q: Can this process be adapted for fluorinated isoquinoline coupling?
A: Yes, the resulting brominated pyridines serve as robust coupling partners for palladium-catalyzed reactions with fluorinated isoquinoline carboxylates to form complex bi-aryl structures.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Brominated Pyridine Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your drug development programs. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition smoothly from lab scale to full commercialization. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of brominated pyridine derivatives meets the highest industry standards. Our commitment to technical excellence means we can navigate the complexities of this specific bromination chemistry with precision, delivering materials that facilitate your downstream synthesis without interruption.
We invite you to engage with our technical procurement team to discuss how this advanced manufacturing route can benefit your specific project requirements. Request a Customized Cost-Saving Analysis to understand the potential economic impact of switching to this more efficient process. We are ready to provide specific COA data and route feasibility assessments to support your decision-making. By partnering with us, you gain access to a supply chain that is not only reliable and compliant but also optimized for cost and performance.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
