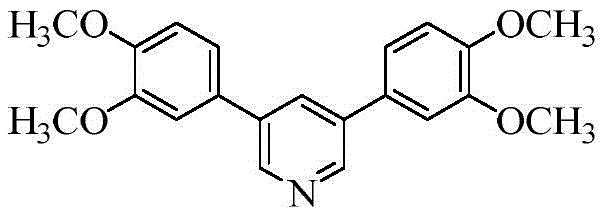
Advanced Metal-Free Synthesis of 3,5-Disubstituted Pyridines for Pharmaceutical Intermediates
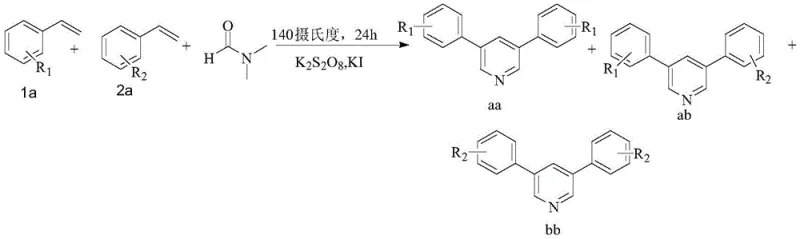
The pharmaceutical and agrochemical industries continuously seek robust, scalable, and cost-effective methodologies for constructing privileged heterocyclic scaffolds. Among these, the pyridine ring system remains a cornerstone structure, ubiquitous in bioactive molecules ranging from anti-inflammatory agents to kinase inhibitors. A significant technological breakthrough in this domain is documented in patent CN111518021A, which discloses a novel method for constructing 3,5-disubstituted pyridines through the oxidative cyclization of mixed styrene derivatives and N,N-dimethylformamide (DMF). This innovation addresses long-standing challenges in heterocycle synthesis by replacing expensive transition metal catalysts with a benign iodide salt system, thereby offering a streamlined pathway for producing high-purity pharmaceutical intermediates. The ability to access both symmetrical and asymmetrical 3,5-disubstituted pyridines in a one-pot reaction represents a substantial leap forward in process chemistry, directly impacting the efficiency of supply chains for complex fine chemicals.
For R&D directors and process chemists, the implications of this technology are profound. Traditional routes to 3,5-diarylpyridines often rely on multistep sequences involving harsh conditions or precious metal catalysis, which introduce significant impurities and cost burdens. The method described in CN111518021A utilizes readily available styrene derivatives and DMF, acting simultaneously as a solvent and a carbon-nitrogen source. This dual functionality not only simplifies the reaction setup but also enhances atom economy. By leveraging a peroxydisulfate oxidant and an iodide catalyst, the process achieves moderate to good yields under relatively mild thermal conditions, making it an attractive candidate for the commercial scale-up of complex heterocycles required in modern drug discovery pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3,5-disubstituted pyridines has been fraught with inefficiencies that hinder large-scale manufacturing. Conventional approaches frequently depend on transition metal-catalyzed cross-coupling reactions, such as those involving palladium or copper complexes, which necessitate rigorous exclusion of air and moisture and often require elevated temperatures that can degrade sensitive functional groups. Furthermore, alternative methods utilizing beta-nitrostyrenes or acryloyl azides, while chemically viable, suffer from high substrate costs and poor selectivity, often yielding mixtures of regioisomers that are difficult to separate. For instance, earlier reported syntheses involving N-alkyl-1,4-dihydropyridine derivatives require lengthy multi-step sequences including acylation, N-dealkylation, and oxidation, resulting in low overall yields and significant waste generation. These limitations create bottlenecks in the production of reliable pharmaceutical intermediate supplier networks, driving up the cost of goods sold and extending lead times for critical API precursors.
The Novel Approach
In stark contrast to these legacy methods, the innovative protocol outlined in CN111518021A offers a direct, one-pot oxidative cyclization strategy that bypasses the need for pre-functionalized substrates or expensive metal catalysts. By employing a mixture of styrene derivatives—specifically aryl ethylenes with varying electronic properties—and DMF under the catalytic influence of an iodonium salt, the reaction efficiently constructs the pyridine core. This approach is particularly advantageous for generating asymmetrical 3,5-disubstituted pyridines, a structural motif often challenging to access selectively. The use of potassium peroxydisulfate as a terminal oxidant ensures a clean reaction profile, while the iodide catalyst facilitates the generation of reactive radical species necessary for cyclization. This methodology not only reduces the environmental footprint by eliminating heavy metals but also significantly lowers the barrier to entry for cost reduction in fine chemical manufacturing, allowing producers to utilize commodity chemicals like styrene and DMF as primary feedstocks.
Mechanistic Insights into Iodide-Catalyzed Oxidative Cyclization
Understanding the mechanistic underpinnings of this transformation is crucial for optimizing process parameters and ensuring consistent product quality. The reaction proceeds through a complex radical cascade initiated by the interaction between the iodide catalyst and the peroxydisulfate oxidant. Initially, DMF undergoes oxidative decomposition in the presence of potassium iodide and potassium peroxydisulfate to generate formaldehyde and dimethylamine in situ. Concurrently, the styrene derivative is oxidized by the iodine radical species to form a phenylacetaldehyde intermediate. This aldehyde then condenses with the generated formaldehyde to form an alpha,beta-unsaturated ketone or aldehyde intermediate, which subsequently undergoes ammonia-aldehyde condensation with dimethylamine. The resulting iminium ion intermediate reacts with a second molecule of styrene derivative (either the same or different, depending on the feed ratio) to close the pyridine ring via a cyclocondensation pathway. This intricate interplay of radical oxidation and polar condensation steps allows for the diverse substitution patterns observed in the final products.
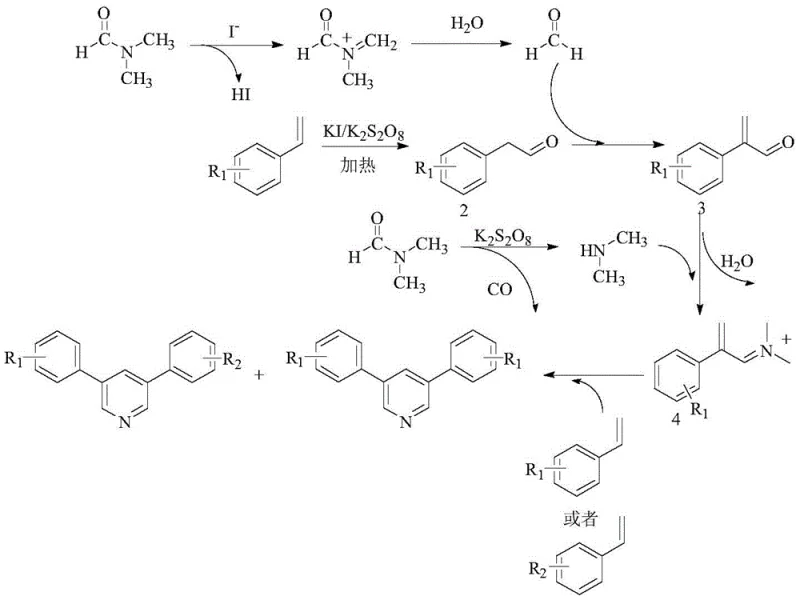
From an impurity control perspective, the mechanism dictates that the ratio of symmetrical to asymmetrical products is heavily influenced by the electronic nature of the substituents on the styrene rings. Electron-donating groups, such as tert-butyl or methyl, enhance the nucleophilicity of the styrene double bond, facilitating faster oxidation and subsequent coupling, which generally leads to higher yields compared to electron-withdrawing halogen substituents. The reaction conditions, particularly the temperature and oxidant loading, must be tightly controlled to prevent over-oxidation of the aldehyde intermediates to carboxylic acids, which would terminate the cyclization sequence. By maintaining the reaction at 140°C and optimizing the molar ratio of oxidant to substrate, the process minimizes side reactions and maximizes the formation of the desired 3,5-disubstituted pyridine scaffold, ensuring a cleaner impurity profile suitable for downstream pharmaceutical applications.
How to Synthesize 3,5-Disubstituted Pyridines Efficiently
The practical execution of this synthesis requires careful attention to reagent stoichiometry and thermal management to achieve reproducible results. The standardized protocol involves charging a reaction vessel with the selected mixed styrene derivatives, excess DMF, potassium peroxydisulfate, and potassium iodide. The mixture is then heated under sealed conditions to maintain solvent integrity and pressure. Detailed operational parameters, including specific workup procedures and purification techniques, are critical for isolating the target compounds with high purity. For a comprehensive, step-by-step laboratory guide tailored to your specific substrate requirements, please refer to the technical documentation below.
- Prepare the reaction mixture by combining mixed styrene derivatives (such as styrene and 4-tert-butylstyrene), N,N-dimethylformamide (DMF) as both solvent and nitrogen source, potassium peroxydisulfate as the oxidant, and potassium iodide as the catalyst in a sealed tube.
- Heat the reaction mixture in an oil bath at an optimized temperature of 140°C for approximately 24 hours to facilitate the oxidative cyclization and formation of the pyridine ring.
- Upon completion, cool the mixture, extract the organic phase with ethyl acetate, dry over anhydrous sodium sulfate, and purify the crude product via silica gel column chromatography to isolate the target 3,5-disubstituted pyridines.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this metal-free synthesis route offers transformative economic and logistical benefits. The elimination of transition metal catalysts removes a significant cost center associated with both the purchase of precious metals and the downstream processing required to meet strict residual metal specifications in pharmaceutical products. Furthermore, the reliance on commodity chemicals like styrene and DMF ensures a stable and resilient supply chain, mitigating risks associated with the scarcity of specialized reagents. The simplicity of the one-pot process also translates to reduced operational complexity, lower energy consumption, and minimized waste disposal costs, all of which contribute to a more sustainable and cost-efficient manufacturing model.
- Cost Reduction in Manufacturing: The replacement of expensive palladium or copper catalysts with inexpensive potassium iodide results in substantial raw material savings. Additionally, the simplified workup procedure, which avoids complex metal scavenging steps, reduces labor and consumable costs. The use of DMF as both solvent and reactant further streamlines the bill of materials, lowering the overall cost of goods sold for high-volume production runs.
- Enhanced Supply Chain Reliability: By utilizing widely available styrene derivatives and bulk solvents, manufacturers can secure long-term supply contracts with multiple vendors, reducing dependency on single-source suppliers. This diversification enhances supply chain resilience against market fluctuations and geopolitical disruptions, ensuring consistent availability of critical pyridine intermediates for continuous API production schedules.
- Scalability and Environmental Compliance: The reaction operates under relatively mild thermal conditions without the need for high-pressure equipment, facilitating straightforward scale-up from kilogram to tonne scales. The absence of heavy metals simplifies wastewater treatment and aligns with increasingly stringent environmental regulations, reducing the regulatory burden and potential liabilities associated with hazardous waste management.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oxidative cyclization technology. These insights are derived directly from the experimental data and optimization studies presented in the patent literature, providing a factual basis for decision-making.
Q: What are the primary advantages of this metal-free synthesis method over traditional transition metal catalysis?
A: This method eliminates the need for expensive and toxic transition metal catalysts like palladium or copper, significantly reducing raw material costs and simplifying the purification process by removing heavy metal residues, which is critical for pharmaceutical compliance.
Q: Can this process produce asymmetrical 3,5-disubstituted pyridines efficiently?
A: Yes, by utilizing a mixture of two different styrene derivatives with distinct substituents, the reaction successfully generates asymmetrical 3,5-disubstituted pyridines alongside symmetrical byproducts, with the asymmetrical product often favored depending on the electronic properties of the substituents.
Q: What represents the optimal reaction condition for maximizing yield in this cyclization?
A: Experimental optimization indicates that using potassium peroxydisulfate (1.5 mmol) and potassium iodide (0.75 mmol) at a temperature of 140°C for 24 hours provides the highest total yield of pyridine products, balancing reaction rate and byproduct formation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,5-Disubstituted Pyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced synthetic methodologies like the one described in CN111518021A for developing next-generation therapeutic agents. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are seamlessly translated into robust industrial processes. We are committed to delivering high-purity 3,5-disubstituted pyridines that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Whether you require custom synthesis of novel analogs or large-scale supply of established intermediates, our infrastructure is designed to support your development timelines and commercial goals.
We invite you to collaborate with us to leverage this cost-effective and environmentally friendly synthesis route for your specific projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this metal-free approach can optimize your budget. Please contact us today to request specific COA data for our existing inventory or to discuss route feasibility assessments for your proprietary targets, and let us help you accelerate your path to market with reliable, high-quality chemical solutions.
