Revolutionizing Irbesartan Production: A Deep Dive into Toxic-Free Phase Transfer Catalysis
Introduction to Next-Generation Irbesartan Manufacturing
The pharmaceutical landscape for antihypertensive agents is constantly evolving, driven by the need for safer, more efficient, and environmentally sustainable manufacturing processes. Patent CN102219779A represents a significant technological leap in the synthesis of Irbesartan, a potent angiotensin II receptor antagonist widely used to treat hypertension and diabetic nephropathy. This patent discloses a novel methodology that fundamentally alters the final cyclization step, moving away from hazardous organotin reagents toward a robust phase-transfer catalytic system. By utilizing sodium azide and inorganic ammonium salts in non-polar solvents, this process achieves exceptional purity levels exceeding 99.8 percent while maintaining total yields between 88 percent and 95 percent. For global supply chain stakeholders, this innovation addresses critical pain points regarding heavy metal contamination and solvent recovery costs.
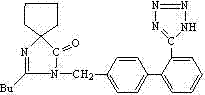
The structural complexity of Irbesartan, particularly the spiro-fused diazabicyclononane ring coupled with the biphenyl tetrazole motif, presents unique challenges in process chemistry. Traditional routes often struggle with the introduction of the tetrazole ring, a pharmacophore essential for biological activity. The breakthrough detailed in this patent lies not just in the reaction itself, but in the downstream processing strategy. By bypassing alkaline extraction—a step known to induce decomposition in this specific molecular scaffold—the inventors have secured a pathway that is both chemically elegant and commercially viable. This report analyzes the mechanistic underpinnings and commercial implications of this technology for R&D directors and procurement leaders seeking reliable irbesartan intermediate supplier partnerships.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of Irbesartan has been plagued by reliance on tributyltin azide for the critical tetrazole formation step. While chemically effective, organotin reagents introduce severe downstream complications, primarily the persistence of toxic tin residues in the final active pharmaceutical ingredient. Removing these heavy metals to meet stringent regulatory limits (often less than 10 ppm) typically necessitates complex purification techniques such as column chromatography or specialized scavenging resins, which drastically inflate production costs and reduce overall throughput. Furthermore, prior art methods, such as those described in US Patent 5629331, utilized N-methylpyrrolidone (NMP) as a solvent. NMP is not only expensive but also poses significant challenges in recovery and recycling due to its high boiling point and hygroscopic nature, leading to substantial operational expenditures in solvent management.
The Novel Approach
The methodology outlined in CN102219779A offers a transformative alternative by employing a phase-transfer catalytic system comprising sodium azide, ammonium chloride, and a quaternary ammonium salt like tetrabutylammonium bromide. This shift eliminates the need for toxic tributyltin azide entirely, thereby removing the burden of heavy metal clearance from the manufacturing workflow. Additionally, the substitution of NMP with non-polar, non-protic solvents such as toluene or xylene simplifies the reaction environment. These solvents are cheaper, easier to recover via distillation, and provide a homogeneous reaction medium that enhances kinetics. Most critically, the process introduces a direct acidification workup at pH 4-5, circumventing the alkaline conditions that previously caused degradation of the sensitive spiro-cyclic core. This holistic optimization results in a cleaner reaction profile and a more robust supply chain for high-purity irbesartan.
Mechanistic Insights into Phase-Transfer Catalyzed Tetrazole Formation
The core chemical transformation in this patent is the [2+3] cycloaddition between the nitrile group of the biphenyl precursor and the azide ion to form the tetrazole ring. In a non-polar solvent like toluene, inorganic sodium azide is inherently insoluble, which would normally halt the reaction. The addition of a phase-transfer catalyst (PTC), specifically tetrabutylammonium bromide, resolves this solubility mismatch. The lipophilic quaternary ammonium cation transports the azide anion from the solid or aqueous phase into the organic phase, where it can effectively collide and react with the dissolved nitrile substrate. This mechanism allows the reaction to proceed efficiently at reflux temperatures (110-130°C) without the need for polar aprotic solvents that are difficult to remove.
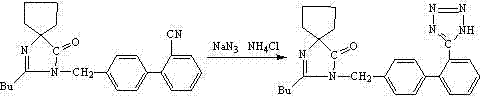
Beyond the cyclization mechanics, the patent highlights a crucial stability insight regarding the workup procedure. The Irbesartan molecule contains a spiro-fused 1,3-diazabicyclo[4.4]nonane-1-ene-4-ketone system which is susceptible to base-catalyzed hydrolysis or ring-opening. Conventional methods often attempt to extract the tetrazole as a salt using strong alkali, inadvertently triggering decomposition pathways that generate impurities and lower yield. By contrast, this novel process opts for direct acid neutralization of the reaction mixture. Adjusting the pH to the slightly acidic range of 4-5 protonates the tetrazole anion, causing the free acid form of Irbesartan to precipitate directly from the solution. This gentle isolation technique preserves the integrity of the spiro-ring system, ensuring that the final product maintains a purity profile superior to 99.8 percent with minimal impurity generation.
How to Synthesize Irbesartan Efficiently
The synthesis protocol described in the patent provides a clear, scalable roadmap for producing Irbesartan with high efficiency. The process begins by charging the nitrile precursor, ammonium chloride, phase transfer catalyst, and sodium azide into a reactor containing toluene or xylene. The mixture is heated to reflux, typically between 110°C and 130°C, and stirred for a period ranging from 24 to 48 hours depending on the specific solvent and temperature profile chosen. Upon confirmation of reaction completion, the mixture is cooled, and water is added to facilitate phase separation. Instead of basic extraction, dilute acid is introduced to adjust the pH to 4-5, inducing precipitation of the crude product. The detailed standardized synthesis steps, including specific molar ratios and crystallization parameters, are provided in the guide below.
- React the nitrile precursor with sodium azide and ammonium chloride in toluene or xylene using a phase transfer catalyst like tetrabutylammonium bromide at reflux temperatures (110-130°C).
- Upon completion, cool the reaction mixture and add water, then directly adjust the pH to 4-5 using dilute acid (e.g., hydrochloric acid) to precipitate the crude product.
- Filter the precipitate, wash, and dry, followed by recrystallization from an ethanol/water mixture to obtain high-purity Irbesartan (>99.8%).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers profound strategic benefits beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the purification train. By eliminating tributyltin azide, manufacturers remove the necessity for expensive heavy metal scavenging steps and the associated validation testing for tin residues. This reduction in processing steps translates directly into lower operational costs and shorter batch cycle times. Furthermore, the switch from NMP to toluene or xylene leverages commodity solvents that are globally available and easily recycled, insulating the production process from the price volatility and supply constraints often associated with specialty solvents like NMP.
- Cost Reduction in Manufacturing: The economic impact of removing organotin reagents cannot be overstated. Tributyltin azide is a high-cost specialty reagent, whereas sodium azide and ammonium chloride are inexpensive bulk commodities. Additionally, the avoidance of column chromatography—a low-throughput, high-solvent-consumption technique—in favor of simple filtration and crystallization significantly reduces the cost of goods sold (COGS). The ability to recover and reuse toluene or xylene further enhances the economic viability of the process, making cost reduction in pharmaceutical manufacturing a tangible reality rather than a theoretical projection.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available raw materials. Sodium azide and quaternary ammonium salts are produced at scale by numerous chemical suppliers globally, reducing the risk of single-source dependency. Moreover, the robustness of the reaction conditions—tolerating standard reflux temperatures and simple workup procedures—minimizes the risk of batch failures due to sensitive operating parameters. This reliability ensures consistent delivery schedules for high-purity irbesartan intermediates, allowing downstream formulation teams to plan with greater confidence and reducing lead time for high-purity api batches.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process is markedly superior. The elimination of tin waste streams simplifies wastewater treatment and reduces the environmental footprint of the manufacturing site. The use of toluene or xylene, while requiring standard VOC controls, is well-understood in industrial settings compared to the disposal challenges of tin-containing sludge. This alignment with green chemistry principles facilitates easier regulatory approval and supports corporate sustainability goals, ensuring that commercial scale-up of complex pharmaceutical intermediates proceeds without environmental bottlenecks.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and comparative analysis presented in CN102219779A, offering clarity on how this technology outperforms legacy methods in terms of purity, yield, and operational safety. Understanding these nuances is essential for technical teams evaluating process transfers.
Q: Why is the elimination of tributyltin azide critical in Irbesartan manufacturing?
A: Tributyltin azide leaves toxic tin residues that are difficult to remove, often requiring expensive chromatography. The new method ensures tin levels are below 5ppm or undetectable, significantly reducing purification costs and environmental hazards.
Q: What are the advantages of using toluene or xylene over NMP in this synthesis?
A: N-methylpyrrolidone (NMP) is expensive and difficult to recover efficiently. Toluene and xylene are cost-effective, non-polar solvents that facilitate easier product isolation and solvent recycling, lowering overall production costs.
Q: How does the direct acidification step improve product yield?
A: Traditional alkaline extraction can cause decomposition of the spiro-fused ring system. Direct acidification to pH 4-5 avoids base-catalyzed degradation, preserving the molecular integrity and achieving yields of 88-95%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Irbesartan Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes requires a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the efficiencies demonstrated in patent CN102219779A can be fully realized at an industrial level. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the detection of trace impurities like tin and triethylamine at parts-per-million levels. We are committed to delivering high-purity Irbesartan that meets the exacting standards of global regulatory bodies.
We invite pharmaceutical companies and generic manufacturers to collaborate with us to leverage this cost-effective and environmentally friendly synthesis technology. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments, ensuring that your supply chain for this critical antihypertensive agent is both robust and economically optimized for the future.
