Optimizing Irbesartan Production: A Technical Deep Dive into Novel Intermediate Synthesis
The pharmaceutical landscape for antihypertensive agents continues to evolve, driven by the demand for more efficient and cost-effective manufacturing processes for blockbuster drugs like Irbesartan. Patent CN102050761A introduces a transformative approach to synthesizing the key intermediate N-(1-cyano-cyclopentyl)-N'-((2'-cyanobiphenyl-4-yl) methyl) pentamidine, which serves as the cornerstone for Irbesartan production. This technical disclosure outlines a robust synthetic pathway that circumvents the limitations of traditional methods, offering a streamlined route characterized by mild reaction conditions and exceptional yield profiles. By leveraging specific carbonyl activation strategies and optimized cyclization protocols, this method addresses critical pain points in large-scale API manufacturing, providing a compelling value proposition for R&D directors seeking to optimize their supply chains for cardiovascular medications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Irbesartan and its precursors has relied on several distinct routes, each carrying inherent inefficiencies that impact commercial viability. Traditional methods often utilize 2-cyano-4'-bromomethylbiphenyl or protected tetrazole derivatives such as N-(4-bromomethylbiphenyl-2-yl)-1-trityl-1H-tetrazole as starting materials. These conventional pathways frequently necessitate the use of expensive protecting groups, such as the trityl group, which adds significant material costs and requires additional synthetic steps for installation and removal. Furthermore, reactions involving bromomethyl intermediates can be prone to side reactions and often require stringent control over stoichiometry to prevent over-alkylation or decomposition. The reliance on harsh conditions or complex purification sequences in these legacy methods results in lower overall throughput and increased waste generation, creating substantial bottlenecks for manufacturers aiming to scale production to meet global demand for this essential antihypertensive medication.
The Novel Approach
In stark contrast, the methodology disclosed in CN102050761A presents a paradigm shift by utilizing N-[(2'-cyanobiphenyl-4-yl) methyl] valeramide as a primary building block. This novel approach eliminates the need for cumbersome protecting groups and leverages a direct amidine formation strategy. The process involves the activation of the amide carbonyl followed by nucleophilic substitution with 1-aminocyclopentanecarbonitrile, a reaction sequence that proceeds with remarkable efficiency. By operating under moderate temperature ranges of -20°C to 80°C, the new method reduces energy consumption and minimizes thermal degradation of sensitive intermediates. The result is a significantly shortened reaction timeline and a simplified workup procedure, typically involving standard extraction and crystallization techniques, which collectively enhance the operational simplicity and economic feasibility of producing high-purity Irbesartan intermediates on an industrial scale.
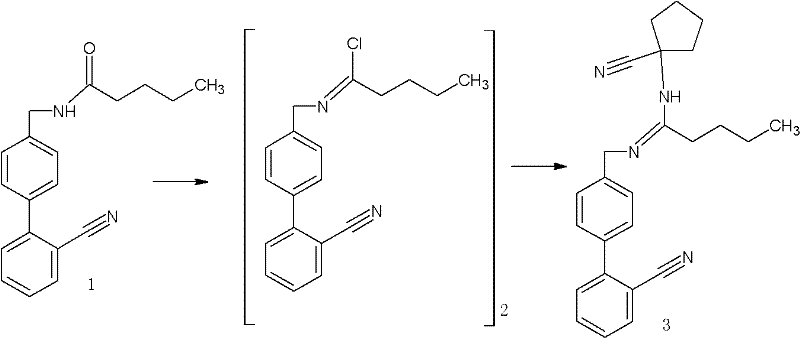
Mechanistic Insights into Amidine Formation and Cyclization
The core of this innovative synthesis lies in the precise activation of the valeramide derivative. In the first critical step, the carbonyl group of N-[(2'-cyanobiphenyl-4-yl) methyl] valeramide is activated using reagents such as oxalyl chloride, thionyl chloride, or triphosgene. This activation generates a highly reactive imidoyl chloride intermediate in situ. The choice of activating agent is pivotal; for instance, triphosgene offers a solid, easier-to-handle alternative to gaseous phosgene while maintaining high reactivity. The subsequent addition of 1-aminocyclopentanecarbonitrile facilitates a nucleophilic attack on the activated carbon, forming the stable amidine linkage found in the key intermediate. This mechanism is highly selective, minimizing the formation of urea byproducts that often plague similar condensation reactions. The use of organic bases like triethylamine or pyridine further drives the equilibrium towards product formation by scavenging the acidic byproducts generated during the activation and substitution phases, ensuring a clean reaction profile suitable for downstream processing.
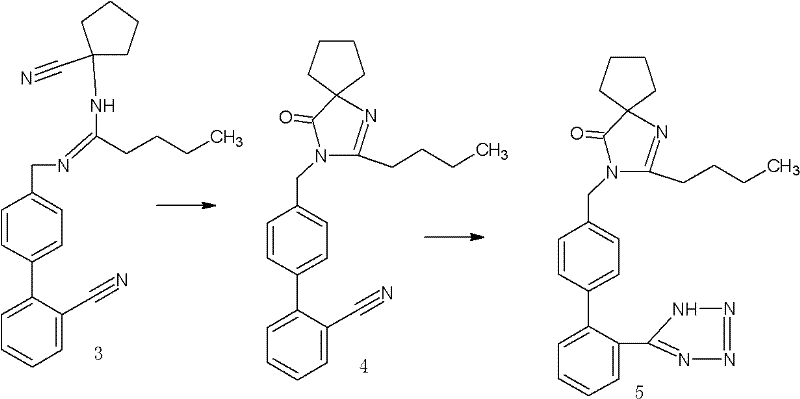
Following the formation of the key amidine intermediate, the pathway proceeds through a sophisticated cyclization sequence to construct the spiro-imidazolinone ring system. This transformation is initiated under a saturated HCl gas atmosphere, which promotes the intramolecular cyclization of the nitrile and amidine functionalities. The subsequent alkaline workup, adjusting the pH to between 8 and 11 using bases like sodium hydroxide or potassium carbonate, neutralizes the reaction mixture and isolates the spiro compound. The final step involves the conversion of the cyano group into a tetrazole ring using sodium azide and triethylamine hydrochloride in a high-boiling solvent such as toluene or xylene. This [2+3] cycloaddition is conducted at elevated temperatures (80°C to 150°C) to overcome the activation energy barrier, ensuring complete conversion to the final Irbesartan structure. The robustness of this mechanistic pathway allows for tight control over impurity profiles, consistently delivering product with purity levels exceeding 99% as verified by HPLC analysis in the patent embodiments.
How to Synthesize N-(1-cyano-cyclopentyl)-N'-((2'-cyanobiphenyl-4-yl) methyl) pentamidine Efficiently
The synthesis of this critical intermediate is designed for operational ease and scalability, making it an ideal candidate for technology transfer. The process begins with the dissolution of the valeramide precursor in a suitable solvent such as toluene or dichloromethane, followed by cooling to control the exotherm during the activation phase. Careful addition of the carbonyl activating reagent ensures the formation of the reactive species without excessive heat generation. Once the activation is complete, the amine component is introduced, and the mixture is allowed to stir, often overnight, to ensure full conversion. The detailed standardized synthesis steps below outline the specific molar ratios, temperature controls, and workup procedures required to replicate the high yields reported in the patent data, providing a clear roadmap for process chemists aiming to implement this route.
- Activate N-[(2'-cyanobiphenyl-4-yl) methyl] valeramide using a carbonyl activating reagent such as oxalyl chloride or triphosgene in a solvent like toluene or dichloromethane at temperatures between -20°C and 80°C.
- React the resulting imidoyl chloride intermediate with 1-aminocyclopentanecarbonitrile in the presence of an organic base to yield the key amidine intermediate with high purity.
- Perform cyclization under saturated HCl gas atmosphere followed by alkaline treatment to form the spiro compound, and finally react with sodium azide to complete the Irbesartan synthesis.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthesis route offers tangible strategic benefits that extend beyond simple yield improvements. The elimination of expensive protecting groups and the use of commodity chemicals like valeramide derivatives and common activating agents drastically simplify the raw material sourcing landscape. This simplification reduces dependency on niche suppliers and mitigates the risk of supply disruptions associated with specialized reagents. Furthermore, the mild reaction conditions reduce the burden on facility infrastructure, allowing for production in standard glass-lined or stainless steel reactors without the need for extreme cryogenic or high-pressure equipment. These factors combine to create a more resilient and cost-efficient supply chain capable of responding rapidly to market fluctuations in the demand for antihypertensive therapies.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven primarily by the reduction in step count and the avoidance of costly reagents. By bypassing the need for trityl protection and deprotection sequences, manufacturers save significantly on material costs and labor hours associated with additional unit operations. The high yields reported, often exceeding 90% for the intermediate formation, mean that less raw material is wasted, directly improving the cost of goods sold (COGS). Additionally, the use of solvents like toluene and dichloromethane, which are easily recoverable and recyclable, further enhances the economic profile by minimizing solvent purchase and disposal expenses, leading to substantial overall cost optimization in pharmaceutical intermediate manufacturing.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials ensures a stable and continuous supply flow. Unlike routes dependent on custom-synthesized bromides or protected tetrazoles, the precursors for this method are accessible from multiple global chemical suppliers, reducing single-source risk. The robustness of the reaction conditions also implies a lower rate of batch failures, which is critical for maintaining consistent inventory levels. This reliability allows supply chain planners to forecast production schedules with greater confidence, ensuring that downstream API manufacturing lines remain operational without interruption, thereby securing the continuity of supply for finished dosage forms.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this route is exceptionally well-suited for commercial expansion. The reaction generates fewer hazardous byproducts compared to traditional methods, simplifying waste treatment and disposal protocols. The ability to operate at moderate temperatures reduces energy consumption, aligning with green chemistry principles and corporate sustainability goals. Moreover, the straightforward workup procedures, involving simple filtration and crystallization, are easily adaptable from kilogram to multi-ton scales. This scalability ensures that the process can grow alongside market demand without requiring disproportionate capital investment in new specialized equipment, facilitating a smooth transition from pilot plant to full commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on reaction parameters and product quality. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the primary advantages of this synthesis route over conventional methods?
A: This route utilizes readily available raw materials and avoids complex protecting group strategies like trityl groups. It operates under moderate conditions (-20°C to 80°C) and achieves high yields (up to 93% for the intermediate), significantly simplifying the purification process compared to older bromomethyl-biphenyl routes.
Q: Which carbonyl activating reagents are most effective for this process?
A: The patent data indicates that oxalyl chloride, thionyl chloride, and triphosgene are highly effective. Triphosgene and oxalyl chloride are particularly preferred due to their ability to drive the reaction to completion with minimal side products when used in molar ratios of 0.5 to 3.0 equivalents.
Q: How is the final tetrazole ring constructed in this methodology?
A: The tetrazole ring is formed in the final step by reacting the spiro compound precursor with sodium azide and triethylamine hydrochloride in a high-boiling solvent like toluene or xylene at temperatures between 80°C and 150°C, ensuring complete cyclization to the active pharmaceutical ingredient.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Irbesartan Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and reliable supply chains in the pharmaceutical industry. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent, high-quality intermediates regardless of volume requirements. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of Irbesartan intermediate meets the highest international standards. Our expertise in process optimization allows us to deliver solutions that not only meet technical specifications but also drive down overall production costs for our partners.
We invite you to collaborate with us to leverage this advanced synthesis technology for your Irbesartan production needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume and quality requirements. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation and quality can strengthen your supply chain and enhance your competitive advantage in the global marketplace.
