Advanced Axial Chiral Binaphthyl Spirocyclic Phosphonium Salts for High-Enantioselective Catalysis
Advanced Axial Chiral Binaphthyl Spirocyclic Phosphonium Salts for High-Enantioselective Catalysis
The landscape of asymmetric synthesis is continually evolving, driven by the demand for higher purity and more efficient chiral intermediates in the pharmaceutical and fine chemical sectors. A significant breakthrough in this domain is documented in patent CN101537373B, which introduces a novel class of axial chiral binaphthyl-containing spirocyclic phosphonium salt phase transfer catalysts. These compounds represent a substantial leap forward in catalyst design, leveraging the rigid stereochemical environment provided by the binaphthyl backbone to induce high levels of chirality in downstream reactions. For R&D directors and process chemists, the ability to access such well-defined chiral environments is critical for developing robust synthetic routes that minimize impurity profiles and maximize yield. The structural versatility of these catalysts, allowing for various substituents at the 3,3'-positions, provides a tunable platform for optimizing specific transformations, particularly the conjugate addition of indoles to alpha,beta-unsaturated carbonyl compounds.
From a supply chain and procurement perspective, the development of such catalysts addresses the perennial challenge of sourcing reliable high-purity chiral auxiliaries that can be produced consistently at scale. The synthesis protocol outlined in the patent utilizes relatively standard organic solvents and moderate thermal conditions, suggesting a pathway that is amenable to kilogram-to-tonne scale production without prohibitive energy costs. This aligns perfectly with the strategic goals of a reliable pharmaceutical intermediate supplier aiming to reduce lead times for high-purity chiral building blocks. By establishing a domestic or regional source for these advanced catalysts, manufacturers can mitigate the risks associated with long import lead times and ensure a continuous supply of critical reagents necessary for the production of active pharmaceutical ingredients (APIs).
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the field of phase transfer catalysis has relied heavily on ammonium salts or simpler phosphonium salts that often lack the rigorous steric definition required for high enantioselectivity in challenging substrates. Traditional chiral catalysts frequently suffer from limited substrate scope, meaning a catalyst optimized for one reaction may fail completely when applied to a slightly different molecular framework. Furthermore, many existing chiral phase transfer catalysts exhibit poor stability under prolonged reaction conditions or are difficult to recover and reuse, leading to increased operational expenditures and waste generation. The reliance on expensive transition metals in alternative asymmetric catalysis methods also introduces concerns regarding heavy metal contamination in the final product, necessitating costly purification steps to meet stringent regulatory limits for pharmaceutical applications. These limitations create bottlenecks in process development, forcing chemists to spend excessive time screening suboptimal catalysts rather than focusing on scale-up and optimization.
The Novel Approach
The innovative strategy presented in the patent overcomes these hurdles by constructing a spirocyclic architecture that locks the binaphthyl units into a fixed chiral conformation. This rigidity prevents the loss of chiral information during the catalytic cycle, ensuring consistent induction of asymmetry. The synthesis involves a direct quaternization reaction between 3,3'-disubstituted-2,2'-bis(halomethyl)-1,1'-binaphthyl and 1,1'-binaphthyl-2,2'-bis(methylene)phosphine. This convergent approach allows for the modular introduction of diverse functional groups, such as phenyl, naphthyl, or trifluoromethyl-substituted aryl rings, at the 3,3'-positions. By tuning these steric and electronic properties, chemists can precisely tailor the catalyst's pocket to accommodate specific substrates, thereby achieving yields ranging from 70% to 99% and enantiomeric excess (ee) values between 50% and 98%. This level of performance significantly reduces the need for extensive downstream purification, directly contributing to cost reduction in API manufacturing.
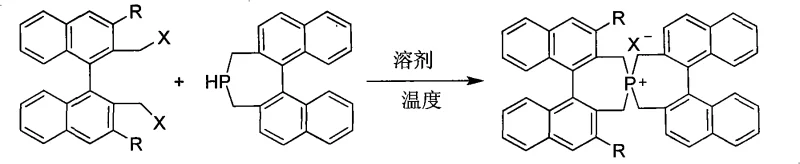
Mechanistic Insights into Spirocyclic Phosphonium Salt Catalysis
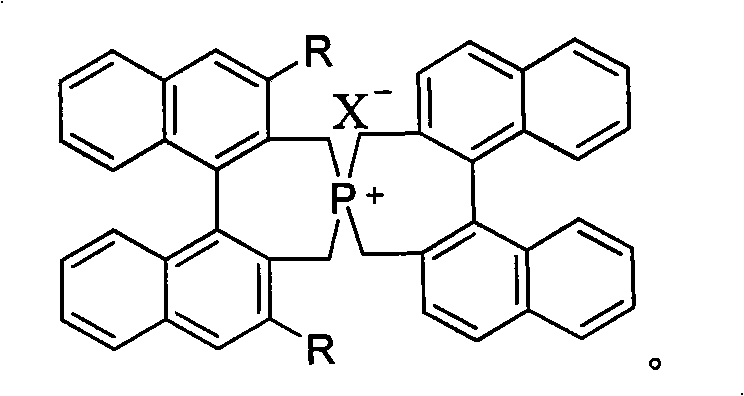
The efficacy of these spirocyclic phosphonium salts stems from their unique three-dimensional topology, which creates a highly differentiated chiral environment around the phosphorus center. In the catalytic cycle, the cationic phosphonium moiety acts as the phase transfer agent, shuttling anionic nucleophiles from the aqueous or solid phase into the organic phase where the electrophilic substrate resides. The bulky binaphthyl wings, especially when substituted with large groups like 3,5-bis(trifluoromethyl)phenyl or naphthyl rings, effectively shield one face of the approaching substrate. This steric shielding forces the reaction to proceed through a single favored transition state, thereby dictating the stereochemical outcome of the bond formation. The spiro-connection further enhances this effect by preventing free rotation that could otherwise blur the chiral distinction, ensuring that the catalyst maintains its integrity and selectivity throughout the reaction duration.
Furthermore, the electronic nature of the substituents plays a pivotal role in modulating the acidity of the proton sources and the nucleophilicity of the anions involved in the reaction. For instance, electron-withdrawing groups like trifluoromethyl can enhance the Lewis acidity of the catalyst-anion complex, potentially accelerating the reaction rate without compromising selectivity. The ability to systematically vary these substituents, as demonstrated by the diverse library of catalysts synthesized in the patent examples, provides a powerful tool for mechanistic optimization. Understanding these structure-activity relationships allows process chemists to predict catalyst behavior and select the optimal variant for a specific transformation, reducing the experimental burden during method development. This deep mechanistic understanding is crucial for scaling up reactions where minor variations in conditions can have amplified effects on product quality.
How to Synthesize Axial Chiral Spirocyclic Phosphonium Salt Efficiently
The preparation of these high-value catalysts follows a straightforward yet precise protocol that balances reaction kinetics with product stability. The process begins with the stoichiometric mixing of the halogenated binaphthyl precursor and the phosphine counterpart in a suitable organic medium. The choice of solvent is critical, with options ranging from non-polar hydrocarbons like toluene and benzene to polar aprotic solvents like acetonitrile and DMF, depending on the solubility of the specific substituents involved. The reaction is typically conducted under an inert atmosphere to prevent oxidation of the phosphine species, ensuring high conversion rates. Following the thermal treatment, the crude product undergoes a standard workup involving aqueous washing and extraction to remove unreacted starting materials and inorganic byproducts. The detailed standardized synthesis steps for specific variants are provided in the guide below.
- Mix 3,3'-disubstituted-2,2'-bis(halomethyl)-1,1'-binaphthyl and 1,1'-binaphthyl-2,2'-bis(methylene)phosphine in a 1: 1 molar ratio within an organic solvent.
- Heat the reaction mixture to a temperature between 0°C and 120°C, maintaining reflux conditions until thin-layer chromatography confirms complete consumption of starting materials.
- Perform workup by pouring the reaction mixture into water, extracting with organic solvent, drying over anhydrous magnesium sulfate, and purifying via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology offers tangible benefits beyond mere technical performance. The synthesis route relies on binaphthyl derivatives, which are increasingly available from established chemical suppliers due to their widespread use in ligand chemistry. This abundance of raw materials ensures a stable supply chain, reducing the risk of production stoppages caused by precursor shortages. Moreover, the reaction conditions are relatively mild, operating between 0°C and 120°C, which means that existing reactor infrastructure can be utilized without the need for specialized cryogenic or high-pressure equipment. This compatibility with standard manufacturing assets translates to lower capital expenditure and faster time-to-market for new processes utilizing these catalysts.
- Cost Reduction in Manufacturing: The elimination of expensive transition metals such as palladium or rhodium, which are often required for comparable asymmetric transformations, results in substantial cost savings. Transition metal catalysts not only carry a high price tag but also require rigorous removal steps to meet ppm-level specifications in pharmaceutical products. By utilizing an organocatalytic approach based on phosphonium salts, manufacturers can bypass these costly purification stages, streamlining the overall production workflow. Additionally, the high yields reported (up to 99%) minimize material waste, further enhancing the economic efficiency of the process and lowering the cost of goods sold for the final active ingredient.
- Enhanced Supply Chain Reliability: The robustness of the spirocyclic structure contributes to improved shelf-life and handling characteristics compared to more fragile chiral complexes. This stability simplifies logistics, allowing for bulk storage and transportation without stringent temperature controls that might otherwise inflate shipping costs. For global supply chains, this reliability is paramount, ensuring that critical catalyst batches arrive at manufacturing sites in optimal condition. The ability to produce these catalysts in multi-kilogram batches using scalable chromatography or crystallization techniques further secures the supply line, enabling manufacturers to respond swiftly to fluctuating market demands for chiral intermediates.
- Scalability and Environmental Compliance: The synthetic methodology generates minimal hazardous waste, primarily consisting of benign halide salts and organic solvents that can be recovered and recycled. This aligns with modern green chemistry principles and helps facilities maintain compliance with increasingly strict environmental regulations. The absence of heavy metals simplifies waste treatment protocols, reducing the environmental footprint of the manufacturing site. Furthermore, the high atom economy of the quaternization reaction ensures that the majority of the input mass is incorporated into the final product, supporting sustainable manufacturing practices that are increasingly demanded by end-users and regulatory bodies alike.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of these spirocyclic phosphonium salts in industrial processes. The answers are derived directly from the experimental data and scope defined in the underlying patent literature, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the fit of this technology within their existing portfolios and identify potential areas for process intensification.
Q: What enantioselectivity can be achieved with these spirocyclic catalysts?
A: According to patent CN101537373B, these catalysts demonstrate high enantioselectivity with ee values ranging from 50% to 98% depending on the specific substituents and reaction conditions.
Q: Which solvents are compatible with the synthesis of these phosphonium salts?
A: The preparation method supports a wide range of organic solvents including toluene, acetonitrile, tetrahydrofuran, chloroform, benzene, and ethyl acetate, offering flexibility for process optimization.
Q: Are these catalysts suitable for large-scale industrial applications?
A: Yes, the synthesis utilizes readily available binaphthyl precursors and standard heating conditions (0-120°C), making the commercial scale-up of complex chiral catalysts feasible without requiring exotic equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Spirocyclic Phosphonium Salt Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced chiral catalysts play in the development of next-generation therapeutics and fine chemicals. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of catalyst meets the exacting standards required for GMP manufacturing. Our commitment to quality ensures that your synthetic routes remain robust and reproducible, minimizing the risk of batch failures and ensuring consistent product quality for your downstream customers.
We invite you to collaborate with us to explore how these axial chiral binaphthyl spirocyclic phosphonium salts can optimize your current synthetic strategies. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and process constraints. Please contact us to request specific COA data for our available catalyst grades and to discuss route feasibility assessments for your target molecules. Together, we can drive innovation and efficiency in your chemical supply chain.
