Advanced Axial Chiral Binaphthyl Spirocyclic Phosphonium Salts for High-Performance Asymmetric Catalysis
Advanced Axial Chiral Binaphthyl Spirocyclic Phosphonium Salts for High-Performance Asymmetric Catalysis
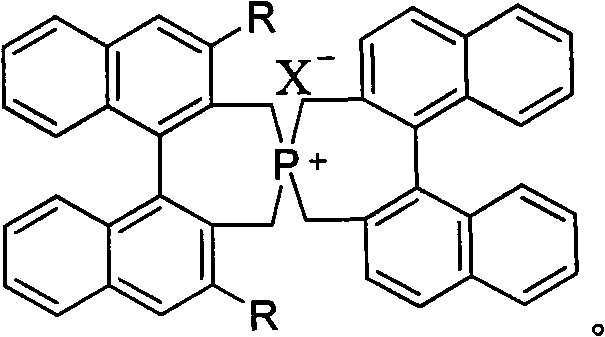
The landscape of asymmetric synthesis is continuously evolving, driven by the relentless demand for high-purity chiral intermediates in the pharmaceutical and agrochemical sectors. A significant breakthrough in this domain is documented in patent CN101537373A, which introduces a novel class of axial chiral binaphthyl-containing spirocyclic phosphonium salt phase transfer catalysts. These sophisticated molecular architectures represent a paradigm shift from traditional ammonium-based systems, offering superior thermal stability and enhanced stereocontrol. By leveraging the rigid spatial arrangement of the binaphthyl backbone combined with a quaternary phosphonium center, this technology addresses critical challenges in enantioselective transformations. For R&D directors and process chemists, understanding the nuances of this innovation is paramount for developing next-generation synthetic routes that meet stringent regulatory purity standards while maintaining economic viability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, asymmetric phase transfer catalysis has relied heavily on chiral quaternary ammonium salts derived from cinchona alkaloids or simple binaphthyl derivatives. While effective in certain contexts, these conventional catalysts often suffer from inherent limitations such as moderate thermal stability, susceptibility to Hofmann elimination under basic conditions, and limited substrate scope. Furthermore, many traditional systems exhibit lower turnover numbers, necessitating higher catalyst loadings which complicates downstream purification and increases the burden on waste management protocols. The flexibility of the linker chains in older generations of catalysts can also lead to conformational freedom that diminishes the precision of the chiral environment, resulting in variable enantiomeric excess (ee) values that may not meet the rigorous specifications required for active pharmaceutical ingredient (API) manufacturing.
The Novel Approach
The technology disclosed in CN101537373A overcomes these hurdles through the strategic design of a spirocyclic quaternary phosphonium framework. Unlike their ammonium counterparts, phosphonium salts possess exceptional thermal and chemical stability, allowing them to withstand harsher reaction conditions without degradation. The spirocyclic topology locks the binaphthyl units into a fixed, rigid conformation, creating a well-defined chiral pocket that exerts precise stereochemical control over the incoming substrates. This structural rigidity translates directly into improved catalytic performance, enabling high yields ranging from 70% to 99% and enantioselectivity values reaching up to 98% ee. This novel approach not only enhances the efficiency of the transformation but also simplifies the overall process workflow by reducing the need for extensive recrystallization steps to upgrade optical purity.
Mechanistic Insights into Spirocyclic Phosphonium Catalysis
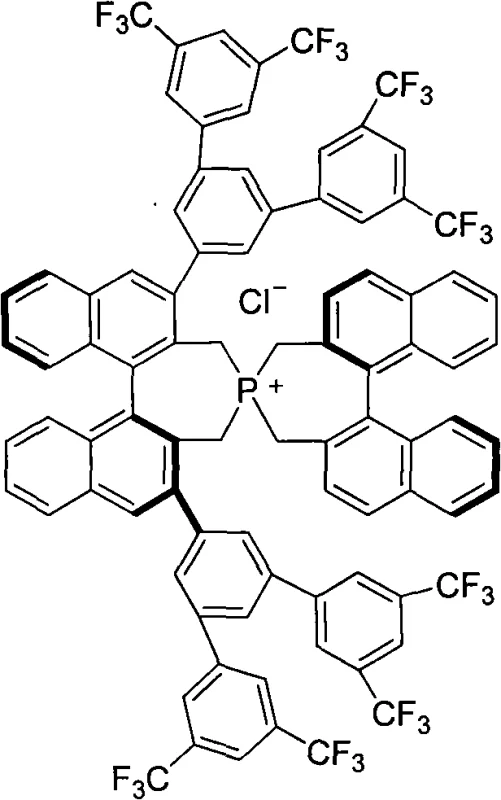
The efficacy of these catalysts stems from the unique interplay between the cationic phosphorus center and the chiral anion or substrate complex within the transition state. The quaternary phosphonium atom acts as a robust anchor, facilitating the transport of anionic nucleophiles into the organic phase where the reaction occurs. Crucially, the bulky binaphthyl wings create a steric barrier that differentiates the prochiral faces of the electrophile, thereby directing the attack of the nucleophile to a specific trajectory. This mechanism is particularly effective in conjugate addition reactions, such as the addition of oxindoles to alpha,beta-unsaturated carbonyl compounds, where the spatial arrangement of the catalyst dictates the absolute configuration of the newly formed stereocenter. The ability to tune the electronic and steric properties of the catalyst by modifying the R groups on the binaphthyl rings allows for fine-tuning of the catalytic activity to suit specific substrate requirements.
Impurity control is another critical aspect where this mechanistic design excels. The high stability of the P-C bonds in the spirocyclic structure minimizes the formation of decomposition byproducts that often plague less stable catalysts. Additionally, the rigid framework reduces the likelihood of non-selective background reactions that occur outside the chiral pocket. This results in a cleaner reaction profile with fewer side products, significantly easing the burden on purification teams. For procurement and supply chain managers, this translates to a more predictable manufacturing process with consistent quality output, reducing the risk of batch failures due to impurity spikes. The robust nature of the catalyst also implies a longer operational lifetime, potentially allowing for multiple cycles of use or continuous flow applications, further enhancing the economic attractiveness of the process.
How to Synthesize Axial Chiral Spirocyclic Phosphonium Salts Efficiently
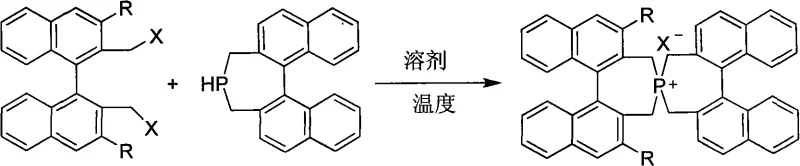
The preparation of these advanced catalysts is remarkably straightforward, relying on a direct nucleophilic substitution reaction that is highly amenable to scale-up. The process involves reacting a 3,3'-disubstituted-2,2'-dihalogenated methyl-1,1'-binaphthyl precursor with a 1,1'-binaphthyl-2,2'-bismethylene phosphine in a 1:1 molar ratio. This simplicity is a major advantage for industrial adoption, as it avoids complex multi-step syntheses or the use of exotic reagents. The reaction proceeds smoothly in common organic solvents such as toluene, acetonitrile, or tetrahydrofuran at temperatures ranging from 0°C to 120°C, providing flexibility in process optimization. Following the reaction, standard workup procedures involving aqueous washing, extraction, and silica gel chromatography yield the target catalyst in high purity. The detailed standardized synthesis steps for implementing this technology in your facility are outlined below.
- Prepare the reaction mixture by combining 3,3'-disubstituted-2,2'-dihalogenated methyl-1,1'-binaphthyl and 1,1'-binaphthyl-2,2'-bismethylene phosphine in a 1: 1 molar ratio under an inert atmosphere.
- Heat the mixture in a suitable organic solvent such as toluene, acetonitrile, or tetrahydrofuran at temperatures ranging from 0°C to 120°C until the reaction is complete as monitored by TLC.
- Perform workup by pouring the reaction mixture into water, extracting with organic solvent, drying over anhydrous magnesium sulfate, and purifying via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this spirocyclic phosphonium catalyst technology offers compelling strategic benefits beyond mere technical performance. The synthesis route utilizes readily available binaphthyl derivatives and phosphine precursors, ensuring a stable and resilient supply chain that is not dependent on scarce natural resources or volatile commodity markets. The elimination of transition metals from the catalytic cycle is a significant cost driver, as it removes the need for expensive metal scavengers and rigorous heavy metal testing required for API release. This metal-free approach aligns perfectly with green chemistry principles and regulatory trends pushing for reduced elemental impurities in drug substances. Furthermore, the high yields and selectivity reported in the patent data suggest a substantial reduction in raw material consumption per kilogram of product, directly impacting the cost of goods sold (COGS).
- Cost Reduction in Manufacturing: The implementation of this catalyst system facilitates significant cost optimization by streamlining the synthetic pathway. By achieving high conversion rates and minimizing side reactions, the process reduces the volume of solvents and reagents required for purification, leading to lower operational expenditures. The absence of precious metal catalysts eliminates a major cost center associated with licensing, recovery, and disposal of toxic heavy metals. Additionally, the thermal stability of the phosphonium salts allows for potentially shorter reaction times or the use of less energy-intensive heating protocols, contributing to overall energy efficiency in the plant.
- Enhanced Supply Chain Reliability: Sourcing reliable chemical inputs is critical for maintaining production schedules, and the precursors for these spirocyclic catalysts are based on established industrial chemicals. The robustness of the catalyst itself means it has a longer shelf life and is less prone to degradation during storage and transportation compared to sensitive organometallic complexes. This stability reduces the risk of supply disruptions caused by spoiled inventory. Moreover, the scalability of the synthesis method ensures that suppliers can ramp up production quickly to meet surges in demand without compromising on quality, providing a secure foundation for long-term supply agreements.
- Scalability and Environmental Compliance: Scaling complex chiral syntheses often presents formidable engineering challenges, but the straightforward preparation of these phosphonium salts mitigates many of these risks. The reaction conditions are mild and do not require specialized high-pressure equipment or cryogenic facilities, making the technology accessible for existing manufacturing infrastructure. From an environmental perspective, the metal-free nature of the catalyst simplifies waste treatment protocols, reducing the environmental footprint of the manufacturing process. This ease of compliance with environmental regulations accelerates the timeline for process validation and regulatory approval, getting products to market faster.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of integrating this technology into their existing workflows, we have compiled answers to common inquiries based on the patent specifications. These questions address key concerns regarding catalyst performance, substrate compatibility, and operational parameters. Understanding these details is essential for conducting a thorough risk assessment and determining the optimal application strategy for your specific synthetic targets. The following insights are derived directly from the experimental data and claims presented in the intellectual property documentation.
Q: What is the enantioselectivity range of these novel spirocyclic catalysts?
A: According to patent CN101537373A, these catalysts demonstrate high enantioselectivity with ee values ranging from 50% to 98% depending on the specific substituents and reaction conditions.
Q: Can these catalysts be reused or recovered after the reaction?
A: While the patent highlights the stability of the quaternary phosphonium structure, phase transfer catalysts are often designed for recovery. The robust spirocyclic framework suggests potential for recycling, though specific recovery protocols depend on the downstream application.
Q: What types of reactions are best suited for this catalyst class?
A: These catalysts are specifically optimized for the conjugate addition of oxindoles to alpha,beta-unsaturated carbonyl compounds, delivering high yields between 70% and 99%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Spirocyclic Phosphonium Salt Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced chiral catalysts like the axial chiral binaphthyl spirocyclic phosphonium salts described in CN101537373A. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from bench-scale discovery to full-scale manufacturing is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including precise determination of enantiomeric excess and residual solvent levels. We are committed to delivering high-purity pharmaceutical intermediates that meet the exacting standards of the global marketplace, leveraging our deep expertise in asymmetric synthesis to optimize your supply chain.
We invite you to collaborate with us to explore how this cutting-edge catalyst technology can enhance your production capabilities. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project requirements, demonstrating the tangible economic benefits of switching to this metal-free catalytic system. Please contact us today to request specific COA data for our catalog of chiral catalysts or to discuss route feasibility assessments for your proprietary molecules. Let us help you achieve superior quality and efficiency in your asymmetric synthesis operations.
