Advanced Industrial Synthesis of Gastrodin: Overcoming Traditional Toxicity and Yield Barriers
The pharmaceutical industry continuously seeks robust synthetic pathways that balance efficiency with environmental safety, particularly for high-demand neurological agents like gastrodin. Patent CN103275146A introduces a transformative chemical synthesis method that fundamentally restructures the production landscape for this critical active pharmaceutical ingredient. By shifting away from the historically hazardous use of red phosphorus and elemental bromine, this technology leverages a sophisticated Lewis acid-catalyzed glycosylation strategy coupled with a novel radical bromination technique. This approach not only mitigates severe safety risks associated with traditional manufacturing but also addresses the chronic issue of low overall yields that have plagued the sector for decades. For global procurement teams and R&D directors, this patent represents a pivotal shift towards greener, more cost-effective, and scalable production methodologies. The ability to synthesize high-purity gastrodin through stable intermediates without heavy metal contamination offers a compelling value proposition for supply chains aiming to reduce lead time for high-purity pharmaceutical intermediates while maintaining rigorous quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of gastrodin has been hindered by reliance on archaic and dangerous chemical protocols that pose significant operational and environmental challenges. Traditional routes typically necessitate the preparation of tetraacetyl-bromo-glucose using large quantities of toxic red phosphorus and bromine, or alternatively, phosphorus tribromide, creating severe waste disposal issues and safety hazards for plant personnel. Furthermore, the tetraacetyl-bromo-glucose intermediate generated in these processes is inherently unstable, complicating storage and handling while leading to inconsistent reaction outcomes in subsequent glycosylation steps. The glycosylation reaction itself, often conducted under alkaline phase-transfer conditions, suffers from notoriously low yields ranging between 35% and 46.5%, drastically inflating the cost of goods sold. Additionally, the final reduction step to convert the formyl group to a hydroxymethyl group traditionally requires expensive reducing agents like potassium borohydride or the use of Raney nickel, which introduces the risk of heavy metal residues in the final API, necessitating costly and time-consuming purification protocols to meet regulatory specifications.
The Novel Approach
In stark contrast to these legacy methods, the patented process described in CN103275146A employs a streamlined, one-pot strategy that begins with the direct acetylation of D-glucose followed immediately by glycosylation with p-cresol. This eliminates the need for isolating unstable brominated sugar intermediates, thereby enhancing process stability and throughput. The introduction of a radical substitution reaction using 1,3-dibromo-5,5-dimethylhydantoin (DBDMH) allows for the precise functionalization of the aromatic methyl group under mild conditions, either through thermal initiation with AIBN or even sunlight irradiation. This innovative step bypasses the need for hazardous phosphorus reagents entirely and avoids the use of heavy metal catalysts during the reduction phase. Instead, a selective hydrolysis under weak alkaline conditions converts the bromomethyl group to the desired hydroxymethyl functionality with exceptional efficiency. The entire sequence is designed such that every intermediate can be purified through simple recrystallization, drastically simplifying the isolation process and ensuring a final product of superior purity suitable for direct pharmaceutical application without extensive downstream polishing.
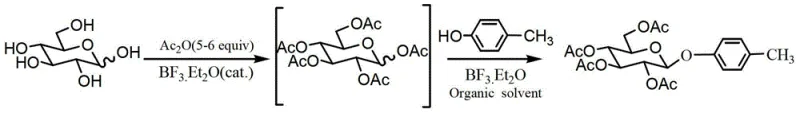
Mechanistic Insights into Lewis Acid-Catalyzed Glycosylation and Radical Functionalization
The core of this technological breakthrough lies in the precise manipulation of stereoelectronic effects during the glycosidic bond formation, facilitated by boron trifluoride etherate (BF3·Et2O) as a potent Lewis acid catalyst. In the initial stage, D-glucose reacts with acetic anhydride to form pentaacetyl-D-glucose, which serves as a stable glycosyl donor. Unlike the unstable bromo-sugar donors used in older methods, this peracetylated species maintains structural integrity, allowing for controlled nucleophilic attack by p-cresol. The Lewis acid activates the anomeric center, promoting the formation of the β-glycosidic linkage with high stereoselectivity, which is critical for the biological activity of the final gastrodin molecule. The reaction conditions are meticulously optimized, with temperature control between 10-30°C preventing the isomerization of the glycosidic bond, a common side reaction that generates difficult-to-remove impurities. This mechanistic control ensures that the crude product contains a high proportion of the desired beta-anomer, reducing the burden on purification steps and maximizing the overall mass balance of the process.
Following glycosylation, the process employs a free-radical substitution mechanism to functionalize the para-methyl group of the phenyl ring, a step that replaces the traditional and inefficient reduction of an aldehyde. By utilizing 1,3-dibromo-5,5-dimethylhydantoin as a bromine source in the presence of a radical initiator like AIBN, the reaction selectively targets the benzylic position to install a bromine atom. This radical pathway is highly advantageous because it proceeds under neutral conditions, preserving the acid-sensitive glycosidic bond and the ester protecting groups on the sugar moiety. The resulting bromomethyl intermediate is then subjected to a mild hydrolysis using aqueous sodium bicarbonate in acetone, which selectively displaces the bromide with a hydroxyl group without affecting the acetyl esters. This chemoselectivity is paramount; it prevents the premature deprotection of the sugar hydroxyls, which would complicate purification. Finally, the removal of the acetyl protecting groups is achieved under standard Zemplén deacetylation conditions using sodium methoxide in methanol, yielding the target gastrodin molecule with high fidelity and minimal byproduct formation.
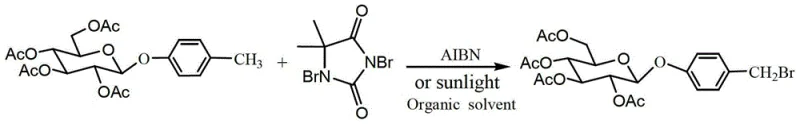
How to Synthesize Gastrodin Efficiently
The synthesis of gastrodin via this patented route involves a logical sequence of four distinct chemical transformations that prioritize safety, yield, and ease of purification. The process begins with the in situ generation of the glycosyl donor, followed by coupling, functionalization, and final deprotection. Each step has been engineered to minimize waste and maximize throughput, making it an ideal candidate for commercial scale-up of complex glycosides. The detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating these results.
- Perform one-pot acetylation of D-glucose followed by Lewis acid-catalyzed glycosylation with p-cresol to form the tetraacetyl glucoside intermediate.
- Execute radical bromination on the methyl group using 1,3-dibromo-5,5-dimethylhydantoin (DBDMH) under AIBN initiation or sunlight.
- Conduct selective hydrolysis of the bromomethyl group to hydroxymethyl, followed by deacetylation using sodium methoxide to yield pure gastrodin.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthesis methodology offers profound strategic benefits that extend beyond mere technical feasibility. The elimination of hazardous reagents like red phosphorus and elemental bromine significantly reduces the regulatory burden and insurance costs associated with handling dangerous chemicals, directly impacting the bottom line. Furthermore, the stability of the intermediates allows for more flexible inventory management and reduces the risk of batch failures due to reagent degradation, ensuring a more consistent supply of critical raw materials. The ability to purify intermediates via recrystallization rather than column chromatography translates to substantial reductions in solvent consumption and processing time, which are key drivers of manufacturing costs in the fine chemical sector. This process optimization enables manufacturers to offer more competitive pricing while maintaining high margins, a crucial factor in the highly price-sensitive generic pharmaceutical market.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the substitution of expensive and hazardous reagents with commodity chemicals. By replacing potassium borohydride and Raney nickel with a radical bromination and hydrolysis sequence, the material costs are drastically simplified. The avoidance of heavy metal catalysts also removes the necessity for specialized metal scavenging resins and the associated validation testing for residual metals, which are costly and time-consuming procedures. Additionally, the high yields reported in the patent examples indicate a more efficient use of starting materials, reducing the cost per kilogram of the final API. The one-pot nature of the initial acetylation and glycosylation steps further reduces labor and utility costs by minimizing unit operations and solvent swaps.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the use of widely available and stable starting materials such as D-glucose and p-cresol. Unlike specialized reagents that may suffer from supply disruptions or long lead times, these commodities are produced on a massive global scale, ensuring continuous availability. The stability of the tetraacetyl-glucoside intermediate allows manufacturers to produce and stockpile key precursors during periods of low demand, smoothing out production schedules and mitigating the risk of bottlenecks. Moreover, the robustness of the reaction conditions means that the process is less susceptible to variations in raw material quality or minor fluctuations in operating parameters, leading to higher first-pass yields and more predictable delivery timelines for customers.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this route is exceptionally well-suited for large-scale industrial production. The absence of phosphorus-containing waste streams simplifies wastewater treatment and reduces the environmental footprint of the manufacturing facility, aligning with increasingly stringent global environmental regulations. The reliance on recrystallization for purification minimizes the generation of silica gel waste and reduces the volume of organic solvents required compared to chromatographic methods. This green chemistry approach not only lowers waste disposal costs but also enhances the corporate sustainability profile of the manufacturer. The process has been demonstrated to work effectively with varying scales of reagents, indicating that translation from pilot plant to multi-ton commercial production can be achieved with minimal technical risk.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this gastrodin synthesis technology. These insights are derived directly from the experimental data and beneficial effects outlined in the patent documentation, providing a clear understanding of the process capabilities and limitations for potential partners and licensees.
Q: How does this new synthesis method improve upon traditional gastrodin production?
A: Traditional methods rely on toxic red phosphorus and bromine to generate unstable tetraacetyl-bromo-glucose, resulting in low yields and heavy metal contamination risks. This patented route utilizes stable pentaacetyl-D-glucose and avoids hazardous reagents entirely.
Q: What are the purification advantages of this specific chemical pathway?
A: Unlike conventional routes requiring complex chromatography, every intermediate in this process can be purified via simple recrystallization. This significantly simplifies downstream processing and ensures high purity suitable for pharmaceutical applications.
Q: Is this process scalable for commercial API manufacturing?
A: Yes, the process is designed for industrialization. It uses readily available raw materials like D-glucose and p-cresol, operates under mild conditions, and eliminates the need for expensive reducing agents like potassium borohydride, making it highly scalable.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Gastrodin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic technologies to meet the evolving demands of the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to delivering high-purity gastrodin that meets stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our expertise in Lewis acid catalysis and radical chemistry allows us to optimize this patented route further, ensuring maximum yield and minimal impurity profiles for our clients.
We invite you to collaborate with us to leverage this cutting-edge synthesis method for your supply chain needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your product portfolio while reducing overall procurement costs.
