Advanced Electrocatalytic Strategy for Scalable Production of Tetra-Substituted Sulfonated Vinyl Ethers
Introduction to Electrocatalytic Vinyl Ether Synthesis
The landscape of organic synthesis is undergoing a paradigm shift towards sustainable, metal-free methodologies, a trend exemplified by the groundbreaking technology disclosed in patent CN112410807A. This intellectual property introduces a novel preparation method for tetra-substituted sulfonated vinyl ethers, a class of compounds critical for the development of bioactive molecules and advanced pharmaceutical intermediates. Unlike traditional thermal approaches that often rely on harsh conditions and toxic reagents, this invention leverages electrocatalysis to drive the coupling of substituted alkynes, aryl sulfonyl hydrazides, and alcohols. The process operates under remarkably mild conditions, utilizing room temperature and weak electrical currents to achieve high conversion rates. For R&D directors and process chemists, this represents a significant opportunity to streamline synthetic routes while adhering to increasingly stringent environmental regulations regarding heavy metal residues in active pharmaceutical ingredients.
The strategic value of this technology lies in its ability to construct complex tetra-substituted olefin frameworks with high regio- and stereoselectivity, a challenge that has historically plagued synthetic organic chemistry due to steric hindrance. By replacing chemical oxidants with electrons as the green reactant, the method not only simplifies the workup procedure but also enhances the overall atom economy of the transformation. This aligns perfectly with the goals of modern green chemistry, offering a pathway to reduce the environmental footprint of fine chemical manufacturing. As we delve deeper into the technical specifics, it becomes clear that this electrochemical strategy is not merely a laboratory curiosity but a robust platform capable of addressing real-world supply chain and cost challenges in the production of high-value sulfonated intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of similar sulfone-containing olefins often relied on transition metal catalysis, which introduces significant bottlenecks in both process efficiency and regulatory compliance. A notable example reported by the Liyan project group involves a copper-catalyzed reaction between alkynes, aryl sulfonyl hydrazides, and diphenyl diselenide to produce seleno-sulfone compounds. While effective, this conventional approach suffers from inherent drawbacks, primarily the reliance on toxic selenium reagents and copper catalysts that are difficult to remove completely from the final product matrix. 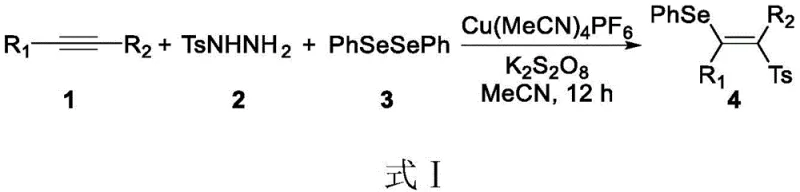 The presence of residual heavy metals like copper is a critical failure point for pharmaceutical applications, necessitating expensive and time-consuming purification steps such as scavenging or repeated recrystallization. Furthermore, the use of stoichiometric amounts of oxidants or toxic solvents in these traditional methods generates substantial chemical waste, increasing disposal costs and complicating the environmental impact assessment for large-scale production facilities.
The presence of residual heavy metals like copper is a critical failure point for pharmaceutical applications, necessitating expensive and time-consuming purification steps such as scavenging or repeated recrystallization. Furthermore, the use of stoichiometric amounts of oxidants or toxic solvents in these traditional methods generates substantial chemical waste, increasing disposal costs and complicating the environmental impact assessment for large-scale production facilities.
The Novel Approach
In stark contrast, the electrocatalytic method described in the patent offers a streamlined, metal-free alternative that fundamentally redesigns the reaction manifold. By employing an undivided electrolytic cell with graphite felt electrodes, the system utilizes electricity to generate reactive radical intermediates directly from the substrates, eliminating the need for external chemical oxidants or metal catalysts entirely. 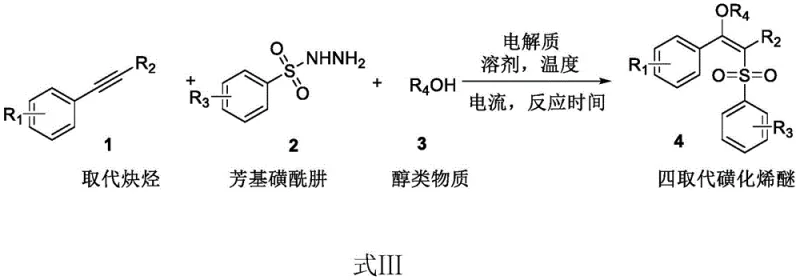 The reaction proceeds smoothly at room temperature in a nitromethane solvent system, driven by a constant low current of 5 mA, which minimizes energy consumption and thermal stress on sensitive functional groups. This approach not only avoids the toxicity issues associated with selenium and copper but also simplifies the downstream processing, as the absence of metal salts allows for direct concentration and chromatographic purification. The result is a cleaner reaction profile with higher selectivity, enabling the efficient synthesis of diverse tetra-substituted sulfonated vinyl ethers with various substituents on the aromatic rings and alkyne chains.
The reaction proceeds smoothly at room temperature in a nitromethane solvent system, driven by a constant low current of 5 mA, which minimizes energy consumption and thermal stress on sensitive functional groups. This approach not only avoids the toxicity issues associated with selenium and copper but also simplifies the downstream processing, as the absence of metal salts allows for direct concentration and chromatographic purification. The result is a cleaner reaction profile with higher selectivity, enabling the efficient synthesis of diverse tetra-substituted sulfonated vinyl ethers with various substituents on the aromatic rings and alkyne chains.
Mechanistic Insights into Electrocatalytic Oxidative Coupling
The success of this transformation hinges on the precise control of electrochemical potentials to facilitate the oxidative coupling of the sulfonyl hydrazide and the alkyne. Mechanistically, the anodic oxidation of the aryl sulfonyl hydrazide generates a sulfonyl radical species, which subsequently adds across the triple bond of the substituted alkyne. This radical addition step is crucial for establishing the carbon-sulfur bond and setting the stereochemistry of the resulting double bond. The use of graphite felt electrodes provides a high surface area for this electron transfer process, ensuring efficient generation of radicals without the over-oxidation that can lead to byproduct formation. The cathodic process likely involves the reduction of protons to hydrogen gas or the reduction of the supporting electrolyte species, maintaining charge balance within the undivided cell without interfering with the main synthetic pathway.
From an impurity control perspective, the electrochemical nature of the reaction offers distinct advantages over thermal radical initiators. By adjusting the current density and voltage, chemists can fine-tune the rate of radical generation to match the consumption rate, thereby minimizing the concentration of free radicals in the bulk solution and reducing dimerization or polymerization side reactions. This level of control is particularly beneficial when dealing with sterically hindered tetra-substituted olefins, where competing pathways are common. The patent data indicates that this method tolerates a wide range of electronic environments, from electron-rich methoxy groups to electron-deficient chloro and fluoro substituents, suggesting a robust radical mechanism that is not overly sensitive to substrate electronics. This mechanistic resilience ensures consistent product quality and reduces the risk of batch-to-batch variability, a key concern for process validation in regulated industries.
How to Synthesize Tetra-Substituted Sulfonated Vinyl Ether Efficiently
Implementing this electrocatalytic protocol requires careful attention to the molar ratios of the starting materials and the configuration of the electrolytic cell to ensure optimal yield and reproducibility. The patent outlines a standardized procedure where substituted alkynes, aryl sulfonyl hydrazides, and tetraethylammonium hexafluorophosphate are combined in a specific 1:3:1 molar ratio, ensuring an excess of the hydrazide to drive the reaction to completion. The choice of alcohol as both a reactant and a co-solvent plays a dual role, participating in the nucleophilic attack on the vinyl cation intermediate while helping to solubilize the ionic species. Detailed operational parameters, including electrode dimensions and stirring rates, are critical for scaling this reaction from milligram to kilogram quantities without losing efficiency. For a comprehensive guide on executing this synthesis with precision, please refer to the standardized protocol below.
- Prepare the reaction mixture by adding substituted alkyne, aryl sulfonyl hydrazide, and tetraethylammonium hexafluorophosphate in a 1: 3:1 molar ratio to an undivided cell equipped with graphite felt electrodes.
- Add 4.5 mL of the chosen alcohol substance and nitromethane as the solvent, then purge the system with nitrogen gas to ensure an inert atmosphere.
- Apply a constant current of 5 mA at room temperature for 8 hours, then concentrate the crude product and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this electrocatalytic technology translates into tangible operational improvements and risk mitigation strategies. The most immediate impact is the drastic simplification of the raw material portfolio; by eliminating the need for expensive transition metal catalysts like palladium, copper, or toxic selenium reagents, the bill of materials becomes more stable and less susceptible to geopolitical supply shocks affecting rare metals. Furthermore, the removal of heavy metals from the process flow significantly reduces the burden on quality control laboratories, as there is no longer a need for sophisticated ICP-MS testing to verify ppm-level metal residuals, accelerating the release of batches for downstream formulation. This streamlining of the quality assurance workflow directly contributes to shorter lead times and improved responsiveness to market demand fluctuations.
- Cost Reduction in Manufacturing: The elimination of metal catalysts and toxic oxidants leads to substantial cost savings by removing the expensive purification steps typically required to meet pharmaceutical heavy metal specifications. Without the need for metal scavengers or complex extraction protocols to remove copper or selenium, the overall processing time is reduced, and solvent consumption is minimized, leading to a leaner and more cost-effective manufacturing process. Additionally, the use of electricity as a traceless reagent avoids the generation of stoichiometric chemical waste, further lowering waste disposal costs and improving the overall process mass intensity metrics.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as substituted alkynes, sulfonyl hydrazides, and simple alcohols ensures a robust and diversified supply base that is not dependent on single-source specialty catalyst vendors. Since the reaction operates at room temperature and atmospheric pressure, it does not require specialized high-pressure reactors or cryogenic cooling systems, allowing for production in a wider range of standard chemical manufacturing facilities. This flexibility enhances supply continuity and reduces the capital expenditure required for equipment upgrades, making it easier to onboard new contract manufacturing partners if capacity expansion is needed.
- Scalability and Environmental Compliance: The electrochemical setup described uses simple undivided cells and graphite electrodes, which are easily scalable from laboratory benchtop units to industrial flow electrolysis reactors without significant re-engineering. The green nature of the process, characterized by the absence of toxic heavy metals and the use of mild conditions, aligns perfectly with modern environmental, social, and governance (ESG) goals, facilitating smoother regulatory approvals and reducing the carbon footprint of the manufacturing site. This sustainability advantage is increasingly becoming a competitive differentiator when supplying to major multinational corporations with strict vendor sustainability codes of conduct.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electrocatalytic synthesis method, derived directly from the experimental data and beneficial effects outlined in the patent documentation. These insights are intended to clarify the practical feasibility of the technology for industrial application and to highlight its superiority over legacy synthetic routes. Understanding these nuances is essential for technical teams evaluating the integration of this process into existing production lines or new product development pipelines.
Q: What are the primary advantages of this electrocatalytic method over traditional metal-catalyzed routes?
A: The primary advantage is the complete elimination of transition metal catalysts, such as copper or selenium reagents, which removes the need for complex and costly heavy metal removal steps required for pharmaceutical grade purity. Additionally, the reaction proceeds at room temperature under weak current, significantly reducing energy consumption compared to high-thermal methods.
Q: Can this synthesis method accommodate diverse functional groups on the alkyne substrate?
A: Yes, the patent data demonstrates excellent substrate tolerance, successfully synthesizing derivatives with electron-donating groups like methyl and methoxy, as well as electron-withdrawing groups like chlorine and fluorine, yielding products with high purity and consistent yields ranging up to 91%.
Q: Is the electrochemical setup suitable for large-scale industrial production?
A: The method utilizes a simple undivided cell with commercially available graphite felt electrodes and standard supporting electrolytes, indicating high potential for scale-up without the need for specialized high-pressure or high-temperature reactors often associated with traditional thermal cyclizations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetra-Substituted Sulfonated Vinyl Ether Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrocatalytic synthesis in the production of high-value pharmaceutical intermediates. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN112410807A can be seamlessly translated into robust industrial processes. We maintain stringent purity specifications and operate rigorous QC labs equipped to handle the unique analytical requirements of electrochemically synthesized compounds, guaranteeing that every batch meets the highest standards of quality and consistency required by the global pharmaceutical industry.
We invite you to collaborate with us to leverage this advanced technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and detailed route feasibility assessments to demonstrate how our expertise in electrocatalytic manufacturing can optimize your supply chain and reduce your overall cost of goods sold.
