Advancing Pharmaceutical Intermediates: Metal-Free Electrocatalytic Synthesis of Tetra-Substituted Sulfonated Vinyl Ethers
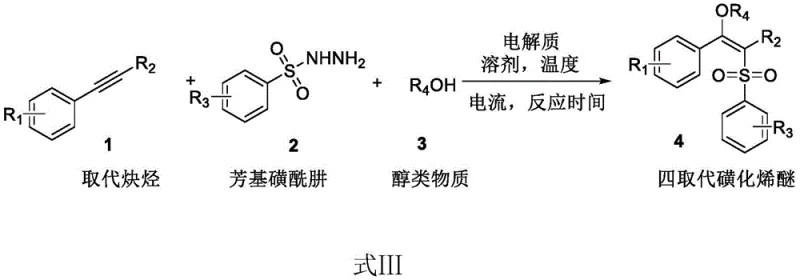
The landscape of organic synthesis for high-value pharmaceutical intermediates is undergoing a significant transformation, driven by the urgent need for greener, more efficient, and cost-effective methodologies. A pivotal advancement in this domain is detailed in patent CN112410807B, which discloses a novel preparation method for tetra-substituted sulfonated vinyl ethers under electrocatalysis. This technology represents a paradigm shift from traditional transition-metal catalysis to a sustainable electro-organic approach. Tetrasubstituted alkenes are critical structural motifs found extensively in biologically active compounds, drugs, and advanced materials, serving as indispensable key intermediates in the synthesis of valuable molecules. However, their construction has historically been plagued by challenges related to steric hindrance and the difficulty of achieving high regioselectivity and stereoselectivity. The invention described in CN112410807B overcomes these hurdles by utilizing an electrocatalytic strategy that promotes the three-component reaction of substituted alkynes, aryl sulfonyl hydrazides, and alcohols. This method not only delivers high yields of the target compounds but also aligns perfectly with the modern principles of green chemistry by operating at room temperature and avoiding the use of hazardous heavy metal catalysts.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
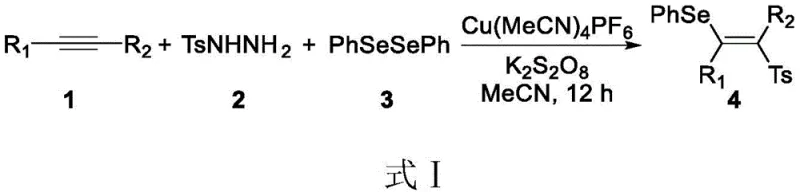
Historically, the synthesis of functionalized alkenes, particularly those with complex substitution patterns like tetra-substituted sulfonated vinyl ethers, has relied heavily on transition metal catalysis. For instance, prior art such as the work by the Li Yan group (J. Org. Chem. 2017) demonstrated the synthesis of selenosulfone compounds via a three-component reaction involving alkynes, arylsulfonylhydrazides, and diphenyl diselenide under copper catalysis. While effective, these conventional methods suffer from inherent and significant defects that limit their utility in large-scale industrial applications. The primary drawback is the reliance on toxic and expensive transition metal catalysts, such as copper salts, which necessitate rigorous downstream processing to remove trace metal residues to meet stringent pharmaceutical purity standards. Furthermore, these reactions often require harsh conditions, including elevated temperatures and the use of toxic organic solvents, which pose safety risks and environmental burdens. The reaction times are frequently prolonged, and the presence of metal catalysts can lead to side reactions that complicate the impurity profile, making purification difficult and costly.
The Novel Approach
In stark contrast to the limitations of metal-catalyzed routes, the novel electrocatalytic approach disclosed in patent CN112410807B offers a transformative solution for the synthesis of tetra-substituted sulfonated vinyl ethers. This method leverages electricity as a clean and traceless reagent to drive the chemical transformation, effectively replacing the need for stoichiometric or catalytic amounts of oxidizing or reducing agents. The reaction proceeds smoothly in an undivided cell using graphite felt electrodes under a constant weak current of 5 mA at room temperature. This mild operational window significantly reduces energy consumption and eliminates the thermal stress often associated with traditional heating methods. By avoiding transition metals entirely, the process inherently produces a cleaner crude product, drastically simplifying the purification workflow. The use of nitromethane as a solvent and simple alcohols as reactants further underscores the practicality and accessibility of this route. This electro-organic strategy not only enhances the sustainability of the manufacturing process but also improves the overall economic viability by reducing raw material costs and waste disposal expenses associated with heavy metal contamination.
Mechanistic Insights into Electrocatalytic Vinyl Ether Formation
The mechanistic underpinning of this electrocatalytic transformation is rooted in the precise manipulation of electron transfer processes at the electrode surface. In this system, the anodic oxidation likely generates reactive radical species from the aryl sulfonyl hydrazide, which then engage with the substituted alkyne substrate. The application of a constant current allows for the fine-tuning of the reaction rate, ensuring that the generation of reactive intermediates matches their consumption, thereby minimizing unproductive side reactions such as polymerization or over-oxidation. The cathodic process complements this by balancing the charge and potentially activating the alcohol nucleophile or facilitating proton reduction. This dual-electrode synergy creates a unique chemical environment that favors the formation of the tetra-substituted double bond with high regioselectivity. Unlike thermal activation, which distributes energy randomly across all vibrational modes of the molecules, electrochemical activation targets specific redox potentials, offering a level of chemoselectivity that is difficult to achieve with conventional reagents. This precision is crucial when dealing with complex substrates containing multiple functional groups, as it ensures that only the desired transformation occurs while leaving other sensitive moieties intact.
From an impurity control perspective, the absence of metal catalysts is a game-changer for pharmaceutical manufacturing. In traditional copper-catalyzed reactions, metal-ligand complexes can persist in the final product, requiring additional steps like chelation or specialized chromatography to reduce metal levels to parts-per-million (ppm) specifications. The electrocatalytic method described here inherently avoids this issue, as the only "reagent" added is electricity. The primary byproducts are typically benign gases or simple salts that are easily removed during the aqueous workup or concentration steps. Furthermore, the mild room temperature conditions prevent thermal degradation of the product or the formation of heat-induced byproducts, which are common in high-temperature syntheses. The result is a product with a superior purity profile right out of the reactor, reducing the burden on quality control laboratories and accelerating the release of batches for downstream processing. This mechanistic elegance translates directly into operational efficiency and cost savings for manufacturers.
How to Synthesize Tetra-Substituted Sulfonated Vinyl Ethers Efficiently
The practical implementation of this electrocatalytic synthesis is straightforward and designed for ease of operation in both laboratory and pilot-scale settings. The protocol involves mixing the substituted alkyne, aryl sulfonyl hydrazide, and supporting electrolyte (tetraethyl ammonium hexafluorophosphate) in a specific molar ratio, typically 1:3:1, within a reactor. An alcohol substance, serving as both reactant and co-solvent, is added along with nitromethane. The reaction is then initiated by applying a constant current across graphite felt electrodes in an undivided cell under a nitrogen atmosphere. This setup minimizes the complexity of the equipment required, avoiding the need for specialized divided cells or membrane separators. The reaction proceeds at room temperature for approximately 8 hours, after which the solvent is removed via rotary evaporation. The resulting crude product is purified using standard silica gel column chromatography to yield the high-purity target compound. For a detailed breakdown of the standardized synthetic steps and specific parameters, please refer to the guide below.
- Prepare the reaction mixture by adding substituted alkyne, aryl sulfonyl hydrazide, and tetraethyl ammonium hexafluorophosphate in a molar ratio of 1: 3:1 into a reactor, followed by the addition of an alcohol substance and nitromethane solvent.
- Conduct the electrolysis in an undivided cell using graphite felt electrodes at a constant current of 5 mA under a nitrogen atmosphere at room temperature for approximately 8 hours.
- Upon completion, concentrate the reaction mixture using a rotary evaporator to obtain the crude product, and purify the target tetra-substituted sulfonated vinyl ether via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this electrocatalytic technology offers compelling strategic advantages that extend beyond mere technical feasibility. The shift away from precious and base metal catalysts fundamentally alters the cost structure of manufacturing these valuable intermediates. By eliminating the need for expensive copper salts and the associated ligands, the raw material costs are significantly reduced. Moreover, the removal of metal scavenging agents and the simplification of the purification process lead to substantial savings in downstream processing time and consumables. This streamlined workflow enhances the overall throughput of the production facility, allowing for faster turnaround times on orders. The reliance on electricity as a primary reagent also insulates the process from the volatility of the chemical reagent market, providing a more stable and predictable cost base for long-term supply agreements.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts removes a major cost driver from the bill of materials. Traditional methods often require expensive metal salts and specialized ligands, the prices of which can fluctuate wildly based on global mining outputs. By replacing these with electricity, the variable cost per kilogram of product is drastically lowered. Additionally, the simplified workup procedure means less solvent consumption and reduced waste disposal fees, contributing to a leaner and more cost-efficient manufacturing operation that maximizes margin potential for high-volume contracts.
- Enhanced Supply Chain Reliability: The starting materials for this electrocatalytic process, such as substituted alkynes and aryl sulfonyl hydrazides, are commodity chemicals that are widely available from multiple global suppliers. This diversification of the supply base reduces the risk of single-source bottlenecks that can plague specialized catalyst-dependent syntheses. Furthermore, the robustness of the reaction conditions—operating at room temperature and atmospheric pressure—means that the process is less susceptible to disruptions caused by equipment failure or utility fluctuations. This reliability ensures consistent delivery schedules, which is critical for maintaining the continuity of downstream pharmaceutical production lines.
- Scalability and Environmental Compliance: Scaling electrochemical reactions is increasingly feasible with modern flow chemistry technologies, allowing for seamless transition from gram-scale development to multi-ton commercial production. The green nature of this process, characterized by zero heavy metal pollution and mild conditions, aligns perfectly with tightening global environmental regulations. This compliance reduces the regulatory burden and the risk of shutdowns due to environmental non-compliance. The ability to produce high-purity intermediates with a minimal environmental footprint enhances the corporate social responsibility profile of the supply chain, making it more attractive to top-tier pharmaceutical partners who prioritize sustainability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the electrocatalytic synthesis of tetra-substituted sulfonated vinyl ethers. These answers are derived directly from the experimental data and beneficial effects reported in patent CN112410807B, providing clarity on the method's capabilities and limitations. Understanding these details is essential for R&D teams evaluating the feasibility of integrating this technology into their existing pipelines and for procurement specialists assessing the long-term viability of the supply source.
Q: What are the primary advantages of this electrocatalytic method over traditional copper-catalyzed routes?
A: The electrocatalytic method eliminates the need for toxic transition metal catalysts like copper, thereby removing the requirement for expensive and complex metal scavenging steps. It operates under mild conditions (room temperature, weak current) and utilizes electrons as clean reagents, resulting in a greener process with easier product purification and higher environmental compliance.
Q: How does this synthesis address the challenge of steric hindrance in tetrasubstituted alkenes?
A: Traditional methods often struggle with the steric bulk of tetrasubstituted double bonds, leading to poor regioselectivity. This electrocatalytic strategy allows for precise tuning of reaction rates via current and voltage adjustments, facilitating the formation of the desired tetra-substituted sulfonated vinyl ether structure with high selectivity despite the steric challenges.
Q: Is this process scalable for industrial production of pharmaceutical intermediates?
A: Yes, the process is highly amenable to scale-up. It uses simple undivided cells, commercially available starting materials, and operates at ambient temperatures without high-pressure equipment. The absence of sensitive metal catalysts further simplifies the supply chain and quality control, making it robust for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetra-Substituted Sulfonated Vinyl Ether Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the electrocatalytic synthesis method described in CN112410807B for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped to handle electro-organic synthesis, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the exacting standards required by the global pharmaceutical industry. We are committed to delivering not just a product, but a reliable supply solution that integrates seamlessly into your manufacturing value chain.
We invite you to collaborate with us to leverage this advanced technology for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this metal-free route can optimize your budget without compromising quality. Please contact our technical procurement team today to request specific COA data for our electrocatalytically produced intermediates and to discuss route feasibility assessments for your target molecules. Let us be your partner in driving efficiency and innovation in your chemical supply chain.
