Advanced Manufacturing of 16-Dehydroprogesterone via Biphasic Elimination for Global Pharma Supply
The pharmaceutical industry constantly seeks robust synthetic routes for critical steroid intermediates that balance high purity with economic viability. Patent CN112940062B introduces a transformative preparation method for 16-dehydroprogesterone, a pivotal precursor in the synthesis of vital corticosteroids such as dexamethasone and betamethasone. This innovation addresses long-standing challenges in steroid chemistry by utilizing a biphasic elimination strategy that operates under significantly milder conditions than traditional protocols. By leveraging a toluene-water solvent system coupled with semicarbazide hydrochloride, the process achieves exceptional selectivity and yield without the need for extreme thermal energy. This technical breakthrough not only enhances the chemical profile of the final active pharmaceutical ingredient but also aligns with modern green chemistry principles by minimizing waste and energy consumption. For R&D directors and procurement specialists, understanding this methodology is crucial for securing a reliable supply chain of high-quality steroid intermediates.
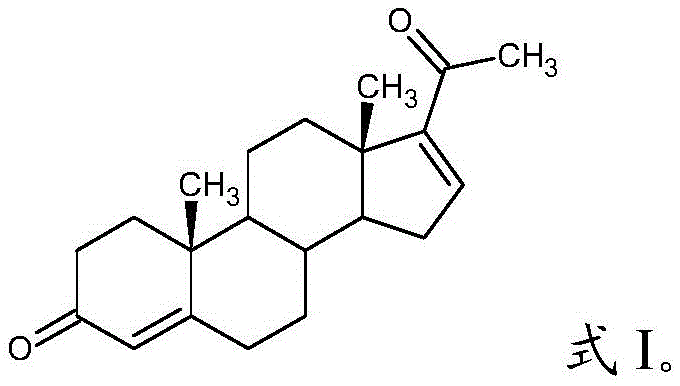
The structural integrity of 16-dehydroprogesterone is paramount for its downstream conversion into life-saving medications. As depicted in the molecular architecture, the specific positioning of the double bond and ketone groups dictates its reactivity and biological efficacy. Traditional synthesis pathways often struggle to maintain this precise configuration without generating significant impurities that require costly removal steps. The novel approach detailed in the patent ensures that the structural fidelity is preserved throughout the elimination reaction. This level of control is essential for manufacturers who must adhere to rigorous regulatory standards regarding impurity profiles. By adopting this advanced synthesis route, pharmaceutical companies can mitigate the risk of batch failures and ensure consistent quality across large-scale production runs, thereby safeguarding patient safety and brand reputation in the competitive global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical methods for producing 16-dehydroprogesterone have been plagued by inefficiencies that hinder industrial scalability and economic feasibility. Prior art, such as the method disclosed in patent application CN106977570A, relies on a multi-step sequence involving esterification followed by hydrolysis and finally a high-temperature elimination step ranging from 240°C to 300°C. These extreme thermal conditions are not only energy-intensive but also pose significant safety hazards in a manufacturing environment. Furthermore, the high heat often leads to thermal degradation of the sensitive steroid backbone, resulting in complex impurity profiles that are difficult to separate. The reliance on such harsh conditions necessitates specialized equipment capable of withstanding extreme temperatures, driving up capital expenditure and maintenance costs. Additionally, the multi-step nature of these conventional processes increases the overall processing time and material loss at each stage, ultimately reducing the overall yield and increasing the cost of goods sold for the final intermediate.
The Novel Approach
In stark contrast, the method provided in CN112940062B streamlines the synthesis into a single-step elimination reaction conducted at a mild temperature range of 80°C to 90°C. This dramatic reduction in thermal requirement eliminates the need for specialized high-temperature reactors and significantly lowers energy consumption. The use of a biphasic system comprising toluene and water creates a unique reaction environment where the elimination occurs in the aqueous layer while the product is simultaneously extracted into the organic toluene layer. This in-situ extraction mechanism protects the product from over-reaction and decomposition, which are common issues in homogeneous high-temperature systems. The simplicity of the operation, involving merely mixing reagents and heating, reduces the operational complexity and the potential for human error. Consequently, this approach offers a safer, more cost-effective, and environmentally friendlier alternative that is ideally suited for continuous large-scale industrial production without compromising on the quality of the output.
Mechanistic Insights into Semicarbazide-Mediated Biphasic Elimination
The core of this technological advancement lies in the sophisticated interplay between the reagents and the biphasic solvent system. Semicarbazide hydrochloride acts as the primary elimination reagent, facilitating the dehydration of 17α-hydroxyprogesterone to form the desired double bond at the 16-position. Acetic acid serves as a catalyst, providing the necessary acidic environment to protonate the hydroxyl group, making it a better leaving group. The reaction specifically takes place within the water layer where the polar reagents are dissolved. As the 16-dehydroprogesterone is formed, its lipophilic nature drives it to partition into the toluene layer immediately. This dynamic phase transfer is critical because it removes the product from the reactive aqueous environment, effectively quenching further reaction and preventing the formation of side products. This mechanism ensures that the reaction equilibrium is shifted towards product formation while maintaining high chemical selectivity.
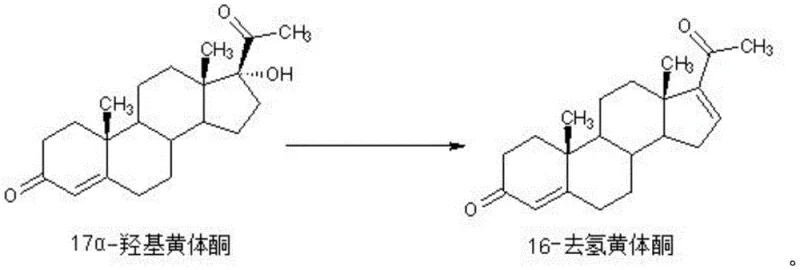
Impurity control is inherently built into this mechanistic design, addressing a major pain point for R&D directors focused on purity specifications. In conventional homogeneous systems, the product remains in contact with the reagents and catalyst for the entire duration of the reaction, increasing the likelihood of secondary reactions and degradation. However, in this biphasic system, the continuous extraction of the product into the toluene layer acts as a protective shield. The toluene layer essentially serves as a reservoir for the pure product, isolating it from the acidic aqueous phase where degradation could occur. Experimental results from the patent indicate that this method consistently achieves purity levels exceeding 99%, with specific examples demonstrating purity as high as 99.3%. This high level of purity reduces the burden on downstream purification processes, such as chromatography or extensive recrystallization, thereby simplifying the overall manufacturing workflow and reducing solvent usage and waste generation associated with additional purification steps.
How to Synthesize 16-Dehydroprogesterone Efficiently
Implementing this synthesis route requires precise adherence to the optimized ratios and conditions outlined in the patent to maximize yield and purity. The process begins with the careful charging of 17α-hydroxyprogesterone, toluene, water, acetic acid, and semicarbazide hydrochloride into a standard reactor equipped with heating and stirring capabilities. The specific mass ratios are critical; for instance, the ratio of raw material to toluene is optimized between 1g to 5-10mL to ensure adequate solubility and phase separation. Once the mixture is homogenized, the temperature is raised to the 80-90°C range and maintained for a period of 4 to 8 hours. Following the reaction, the mixture is allowed to settle, allowing the distinct toluene and water layers to separate clearly. The detailed standardized synthesis steps see the guide below.
- Mix 17α-hydroxyprogesterone with toluene, water, acetic acid, and semicarbazide hydrochloride in a reactor to form a biphasic system.
- Heat the mixture to 80-90°C for 4-8 hours to facilitate the elimination reaction within the aqueous layer while extracting product to the organic layer.
- Separate the layers, wash the toluene layer, concentrate to dryness, and recrystallize the residue using ethyl acetate to obtain pure 16-dehydroprogesterone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthesis method offers tangible benefits that extend beyond mere chemical efficiency. The primary advantage lies in the substantial cost reduction in pharmaceutical intermediate manufacturing driven by the simplification of the process. By eliminating the need for high-temperature operations and multi-step sequences, the facility can reduce energy consumption and equipment wear and tear significantly. The use of common, inexpensive solvents like toluene and water, which are easily recoverable and recyclable, further drives down the raw material costs. The ability to recycle toluene after concentration means less solvent purchase and less waste disposal cost, contributing to a leaner and more sustainable production model. These factors combine to create a more competitive cost structure, allowing suppliers to offer better pricing without sacrificing margins.
- Cost Reduction in Manufacturing: The elimination of extreme thermal requirements and complex multi-step procedures directly translates to lower operational expenditures. The mild reaction conditions reduce the energy load on the facility, while the high selectivity minimizes the loss of expensive starting materials to by-products. Furthermore, the simplified workup procedure reduces labor hours and solvent consumption associated with purification. This holistic reduction in process complexity ensures that the cost of goods sold is optimized, providing a strong buffer against market volatility in raw material prices. The economic efficiency of this route makes it a preferred choice for long-term supply contracts where price stability is a key negotiation point.
- Enhanced Supply Chain Reliability: The robustness of this synthesis method enhances the reliability of the supply chain by reducing the risk of production delays. Conventional methods prone to impurity formation often require re-processing or extended purification times, which can bottleneck production schedules. In contrast, the high consistency and yield of this biphasic method ensure predictable output volumes. The use of readily available and stable reagents like semicarbazide hydrochloride and acetic acid mitigates the risk of supply disruptions associated with specialized or hazardous chemicals. This stability allows supply chain planners to forecast inventory levels with greater accuracy, ensuring continuous availability of high-purity pharmaceutical intermediates for downstream drug manufacturing.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is straightforward due to the use of standard unit operations like mixing, heating, and phase separation. The environmental footprint is significantly reduced as the process avoids the generation of hazardous waste associated with high-temperature degradation products. The recyclability of the toluene solvent aligns with increasingly stringent environmental regulations regarding volatile organic compound emissions and waste disposal. This compliance reduces the regulatory burden and potential fines, making the manufacturing process more sustainable. The ease of scale-up ensures that suppliers can rapidly respond to increased market demand, scaling production from 100 kgs to 100 MT annually without the need for fundamental process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation. Understanding these details helps stakeholders evaluate the feasibility of integrating this technology into their existing supply chains. The answers reflect the consensus on the method's superiority in terms of safety, efficiency, and product quality compared to legacy technologies.
Q: Why is the biphasic toluene-water system superior to conventional high-temperature elimination methods?
A: Conventional methods often require extreme temperatures ranging from 240°C to 300°C, which pose significant safety risks and energy costs. The biphasic system operates at a mild 80-90°C, significantly reducing energy consumption and thermal degradation risks while maintaining high reaction efficiency.
Q: How does this process ensure high purity levels suitable for pharmaceutical applications?
A: The unique phase separation mechanism ensures that the product is continuously extracted into the toluene layer as it forms, preventing over-reaction and the formation of by-products. Experimental data demonstrates purity levels reaching 99.3% after simple recrystallization, meeting stringent pharmaceutical standards.
Q: Is this synthesis route scalable for industrial commercial production?
A: Yes, the process utilizes common solvents like toluene and water which are easily recoverable and recyclable. The mild reaction conditions and straightforward workup procedure, involving simple phase separation and crystallization, make it highly suitable for large-scale manufacturing from 100 kgs to 100 MT annually.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 16-Dehydroprogesterone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global pharmaceutical landscape. Our technical team has extensively evaluated the biphasic elimination route described in CN112940062B and confirmed its potential for robust commercial manufacturing. As a leading CDMO partner, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this efficient synthesis to life. Our facilities are equipped with state-of-the-art rigorous QC labs that ensure every batch meets stringent purity specifications, consistently delivering material that exceeds the 99% purity benchmark demonstrated in the patent. We are committed to providing a secure and compliant supply of 16-dehydroprogesterone that supports your drug development and commercialization goals.
We invite you to collaborate with us to optimize your supply chain for this critical steroid intermediate. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. By partnering with us, you gain access to specific COA data and route feasibility assessments that validate the commercial viability of this advanced manufacturing process. Contact us today to discuss how we can support your production needs with reliable, high-purity 16-dehydroprogesterone synthesized via this cutting-edge technology.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
