Advanced One-Step Elimination Route for Commercial 16-Dehydroprogesterone Production
The pharmaceutical industry continuously seeks robust synthetic routes for critical steroid intermediates, and the preparation of 16-dehydroprogesterone stands as a pivotal challenge in this domain. As detailed in the recent patent CN112940062A, a groundbreaking methodology has emerged that fundamentally shifts the paradigm from complex, high-energy processes to a streamlined, biphasic catalytic elimination. This innovation addresses the longstanding need for a reliable pharmaceutical intermediates supplier capable of delivering high-purity materials essential for synthesizing downstream drugs like progesterone, hydrocortisone, and dexamethasone. The core of this technological leap lies in the utilization of a toluene-water biphasic system, which not only simplifies the operational workflow but also inherently protects the sensitive steroid skeleton from thermal degradation. By leveraging 17α-hydroxyprogesterone as a cost-effective starting material and employing semicarbazide hydrochloride as a specific elimination reagent, this process achieves exceptional purity levels exceeding 99% under mild thermal conditions. The structural integrity of the final product, as illustrated below, is maintained through precise control of the reaction environment, ensuring it meets the rigorous standards required for hormonal therapy manufacturing.
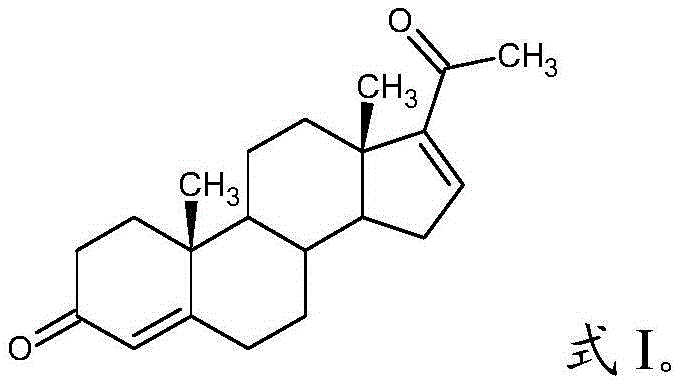
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 16-dehydroprogesterone has been plagued by inefficient methodologies that impose severe constraints on cost reduction in pharmaceutical intermediates manufacturing. Prior art, such as the method disclosed in patent application CN106977570A, relied on a cumbersome multi-step sequence involving initial esterification followed by hydrolysis to convert the 3-position ester group into a ketone. Furthermore, the critical elimination step in these traditional routes necessitated extreme thermal conditions, often requiring temperatures ranging from 240°C to 300°C. Such harsh environments not only consume excessive energy but also pose significant risks of thermal decomposition, leading to the formation of complex impurity profiles that are difficult to remove. The complexity of these legacy processes results in prolonged production cycles and lower overall yields, making them economically unviable for large-scale industrial applications where margin compression is a constant pressure. Additionally, the use of homogeneous solvent systems in older methods often traps the product in the reaction medium, exposing it to prolonged contact with reagents that promote side reactions.
The Novel Approach
In stark contrast, the novel approach described in CN112940062A introduces a sophisticated yet operationally simple one-step elimination strategy that circumvents the pitfalls of high-temperature processing. By employing a biphasic solvent system comprising toluene and water, the reaction creates a dynamic equilibrium where the elimination occurs in the aqueous phase, driven by acetic acid catalysis, while the product is continuously extracted into the organic toluene phase. This "reactive extraction" mechanism effectively shields the newly formed 16-dehydroprogesterone from the acidic aqueous environment, preventing over-reaction and the generation of degradation byproducts. The process operates at a significantly milder temperature range of 80-90°C, drastically reducing energy overheads and equipment stress. Moreover, the use of inexpensive and readily available reagents like semicarbazide hydrochloride eliminates the need for exotic catalysts, thereby streamlining the supply chain and enhancing the economic feasibility of commercial scale-up of complex pharmaceutical intermediates. This method represents a quintessential example of green chemistry principles applied to steroid synthesis, balancing efficiency with environmental stewardship.
Mechanistic Insights into Biphasic Catalytic Elimination
The efficacy of this synthesis hinges on the intricate interplay between the biphasic solvent system and the specific reactivity of the elimination reagents. Acetic acid serves a dual role: it acts as a proton donor to activate the hydroxyl group at the 17α-position of the starting material, facilitating the departure of the leaving group, and it maintains the necessary acidic pH in the water layer to drive the elimination forward. Simultaneously, semicarbazide hydrochloride functions as a specialized dehydrating agent that promotes the formation of the double bond at the 16-position without attacking other sensitive functional groups on the steroid nucleus. The reaction kinetics are optimized by the phase transfer dynamics; as the 16-dehydroprogesterone forms in the water layer, its higher solubility in the non-polar toluene layer drives its immediate migration across the interface. This continuous removal of product from the reaction zone shifts the equilibrium towards completion according to Le Chatelier's principle, ensuring high conversion rates even with stoichiometric amounts of reagents. The visual representation of this transformation highlights the clean conversion of the 17α-hydroxy group into the conjugated enone system characteristic of the target molecule.
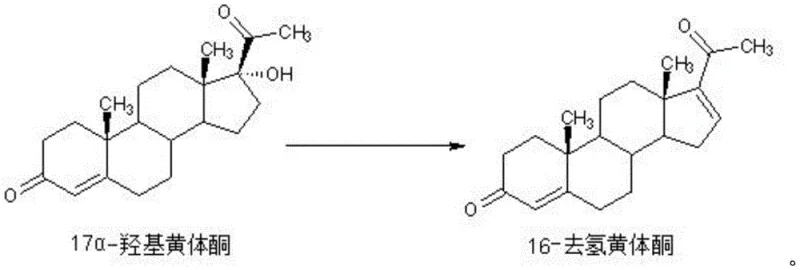
Controlling the impurity profile is paramount for any high-purity pharmaceutical intermediate, and this mechanism offers inherent selectivity advantages. In homogeneous systems, the product remains in contact with the acidic catalyst and elimination reagents for the entire duration of the reaction, increasing the probability of side reactions such as polymerization or further dehydration. However, in this biphasic design, the partition coefficient of the product favors the organic phase, effectively isolating it from the reactive aqueous environment once formed. This spatial separation minimizes the residence time of the product in the harsh reaction conditions, thereby suppressing the formation of polar impurities that typically remain in the water layer. Post-reaction workup is consequently simplified; the water layer, containing the bulk of polar impurities and spent reagents, is easily removed by phase separation. The toluene layer, enriched with the target compound, requires minimal washing before concentration, leading to a crude product of sufficient quality for final recrystallization. This mechanistic elegance translates directly into operational simplicity and consistent batch-to-batch reproducibility.
How to Synthesize 16-Dehydroprogesterone Efficiently
Implementing this synthesis route requires precise adherence to the stoichiometric ratios and thermal parameters defined in the patent to maximize yield and purity. The process begins with the careful charging of 17α-hydroxyprogesterone into a reactor equipped with efficient stirring and temperature control, followed by the addition of the biphasic solvent system. The ratio of toluene to substrate is critical, typically maintained between 5 mL to 10 mL per gram of starting material, ensuring adequate volume for product extraction without diluting the reaction rate excessively. Water is added in a controlled amount, generally 0.3 mL to 1 mL per gram of substrate, to create the distinct aqueous phase required for the catalytic cycle. Acetic acid and semicarbazide hydrochloride are then introduced to initiate the elimination, with the system heated to the optimal window of 80-90°C. Detailed standard operating procedures regarding mixing speeds, addition rates, and specific crystallization protocols are essential for technology transfer and are outlined in the comprehensive guide below.
- Mix 17α-hydroxyprogesterone with toluene, water, acetic acid, and semicarbazide hydrochloride in a reactor to form a biphasic system.
- Heat the mixture to 80-90°C and maintain for 4-8 hours to facilitate the elimination reaction in the aqueous layer while extracting product into the organic layer.
- Separate the layers, wash the toluene layer with water, concentrate to dryness, and recrystallize the residue using ethyl acetate to obtain pure 16-dehydroprogesterone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers transformative benefits that extend far beyond simple chemical conversion. The shift from multi-step, high-temperature processes to a single-step, low-temperature elimination fundamentally alters the cost structure of production. By eliminating the need for energy-intensive heating up to 300°C and removing intermediate isolation steps, the operational expenditure is significantly reduced. Furthermore, the ability to recycle the toluene solvent after concentration creates a closed-loop system that minimizes raw material waste and disposal costs. This efficiency gain allows manufacturers to offer more competitive pricing structures without compromising on the stringent quality specifications demanded by the global pharmaceutical market. The simplified workflow also reduces the dependency on complex utility infrastructure, making the process adaptable to a wider range of manufacturing facilities.
- Cost Reduction in Manufacturing: The economic advantages of this method are driven by the drastic simplification of the process flow and the use of commodity chemicals. Unlike legacy methods that require expensive solvents like tetrahydrofuran or dichloromethane in large volumes, this process utilizes toluene, which is not only cheaper but also easier to recover and recycle. The elimination of the esterification and hydrolysis steps removes the need for additional reagents and the associated labor and time costs. Moreover, the high selectivity of the reaction means that less starting material is wasted on byproduct formation, improving the overall atom economy. These factors combine to deliver substantial cost savings that can be passed down the supply chain, enhancing the competitiveness of the final API.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by complex syntheses that have multiple points of failure; this robust one-step method mitigates those risks. The reaction time is significantly shortened to a window of 4 to 8 hours, compared to the 16 hours or more required by comparative methods using homogeneous solvents. This faster throughput increases plant capacity and allows for more flexible production scheduling to meet fluctuating market demands. Additionally, the raw materials, specifically 17α-hydroxyprogesterone and semicarbazide hydrochloride, are widely available commodities, reducing the risk of supply bottlenecks associated with specialty reagents. The reliability of the process ensures that delivery timelines are met consistently, fostering stronger partnerships between suppliers and downstream drug manufacturers.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges, but the biphasic nature of this reaction facilitates smooth scale-up from laboratory to commercial tonnage. The clear phase separation allows for easy handling of large volumes using standard industrial centrifuges and separators, avoiding the emulsions that often plague homogeneous systems. From an environmental perspective, the ability to recycle toluene and the reduction in aqueous waste volume due to the concentrated reaction conditions align with modern sustainability goals. The process avoids the use of chlorinated solvents like dichloromethane, which are increasingly regulated due to their environmental impact. This compliance with green chemistry principles future-proofs the supply chain against tightening environmental regulations and reduces the burden of waste treatment.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is crucial for stakeholders evaluating its integration into their supply chains. The following questions address common inquiries regarding the operational parameters, purity profiles, and scalability of the method described in CN112940062A. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering a transparent view of the technology's capabilities. By clarifying these aspects, we aim to facilitate informed decision-making for R&D and procurement teams looking to optimize their steroid intermediate sourcing strategies.
Q: How does the biphasic toluene-water system improve product purity?
A: The biphasic system allows the elimination reaction to occur in the water layer while the generated 16-dehydroprogesterone is immediately extracted into the toluene layer. This physical separation prevents the product from undergoing excessive reaction or degradation in the acidic aqueous environment, significantly reducing impurity formation.
Q: What are the optimal reaction conditions for this elimination process?
A: The process operates efficiently at temperatures between 80-90°C for a duration of 4-8 hours. Acetic acid serves as the catalyst to provide the necessary acidic environment, while semicarbazide hydrochloride acts as the elimination reagent.
Q: Why is this method considered superior to previous high-temperature methods?
A: Unlike prior art requiring temperatures of 240-300°C and multi-step esterification/hydrolysis sequences, this method achieves conversion in a single step at mild temperatures (80-90°C). This reduces energy consumption, simplifies operation, and avoids thermal degradation of the steroid backbone.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 16-Dehydroprogesterone Supplier
As the global demand for high-quality steroid intermediates continues to rise, partnering with a technically proficient manufacturer is essential for securing a stable supply of critical materials. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging advanced synthetic methodologies like the biphasic elimination process to deliver superior value to our clients. Our facility is equipped with state-of-the-art reactors and purification systems capable of executing complex organic transformations with precision. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether you are in the clinical trial phase or full-scale commercialization. Our commitment to quality is underscored by our stringent purity specifications and rigorous QC labs, which utilize advanced analytical techniques to verify every batch against the highest industry standards.
We invite you to explore how our optimized production capabilities can enhance your project's economics and timeline. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume needs, demonstrating the tangible financial benefits of switching to our efficient manufacturing route. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments. By collaborating with us, you gain access to a supply chain that prioritizes innovation, reliability, and cost-effectiveness, positioning your organization for success in the competitive pharmaceutical landscape.
