Advanced Synthetic Route for High-Purity Lafutidine Intermediates via Hydroxylamine Aminolysis
Introduction to Advanced Lafutidine Manufacturing
The pharmaceutical landscape for histamine H2 receptor antagonists continues to evolve, driven by the demand for ultra-high purity active pharmaceutical ingredients (APIs) that minimize patient risk and streamline regulatory approval. A pivotal advancement in this domain is detailed in patent CN103130782A, which introduces a robust chemical synthetic method for preparing Lafutidine. This novel approach fundamentally reimagines the construction of the critical primary amine intermediate by substituting traditional hydrazinolysis reagents with hydroxylamine hydrochloride. By leveraging this specific aminolysis strategy, manufacturers can bypass the notorious reduction side reactions associated with hydrazine, thereby securing a product profile of exceptional clarity. The structural integrity of the target molecule, Lafutidine (Formula 1), is preserved with remarkable fidelity, setting a new benchmark for quality in the production of this potent gastroprotective agent.
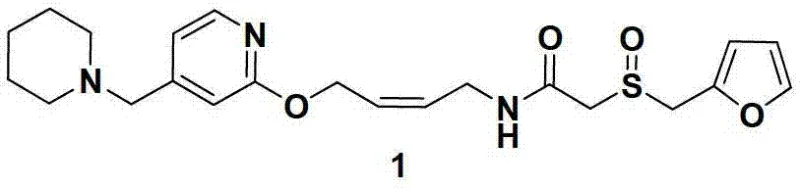
This technological breakthrough addresses long-standing challenges in the supply chain of gastro-intestinal therapeutics, offering a pathway that is not only chemically superior but also operationally safer. The shift away from volatile and unstable reagents towards solid, high-purity hydroxylamine salts represents a significant maturation of the process chemistry. For global procurement teams and R&D directors, understanding the nuances of this synthesis is critical, as it directly impacts the cost of goods sold (COGS) through reduced purification burdens and enhanced yield consistency. The following analysis dissects the mechanistic advantages and commercial implications of adopting this hydroxylamine-mediated route for large-scale Lafutidine manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of Lafutidine has relied heavily on the Gabriel synthesis method, a classic organic transformation used to generate primary amines from phthalimides. While theoretically sound, the practical execution of this route using hydrazine hydrate for the deprotection step (Ing-Manske reaction) introduces severe complications. Hydrazine is a potent reducing agent, and under the reaction conditions required for aminolysis, it indiscriminately attacks the electron-deficient double bond within the crotyl linker of the intermediate. This unintended reduction leads to the formation of dihydro-lafutidine (Formula 7) and its precursor dihydro-amine (Formula 8), which are structurally analogous impurities that are notoriously difficult to separate from the desired product.
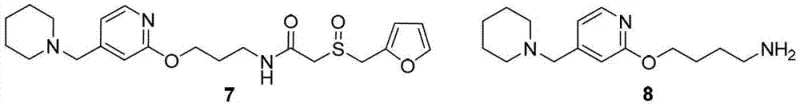
The presence of these saturated impurities necessitates rigorous and often repetitive recrystallization processes to meet stringent pharmacopeial standards, typically requiring single impurity levels below 0.1%. This reliance on extensive downstream purification drastically erodes overall process yield and inflates manufacturing costs due to solvent consumption and extended processing times. Furthermore, the instability of alternative reagents like methylamine solutions adds another layer of operational risk, as concentration fluctuations can lead to batch-to-batch variability. Consequently, the conventional hydrazine-based route creates a bottleneck in the supply chain, characterized by lower throughput, higher waste generation, and a persistent struggle to maintain the high purity profiles demanded by modern regulatory bodies.
The Novel Approach
In stark contrast to the legacy methods, the innovative process disclosed in patent CN103130782A utilizes hydroxylamine hydrochloride as the exclusive aminolysis reagent to convert the phthalimide intermediate (Formula 4) into the crucial primary amine (Formula 2). This strategic substitution effectively decouples the nucleophilic attack required for deprotection from any reducing capability, thereby preserving the integrity of the Z-alkene configuration in the carbon chain. The reaction proceeds smoothly under mild conditions, typically at room temperature in ethanol, utilizing sodium hydroxide to generate the reactive hydroxylamine species in situ. This methodological shift eliminates the root cause of dihydro-impurity formation, resulting in a crude product that is significantly cleaner and requires far less intensive purification.
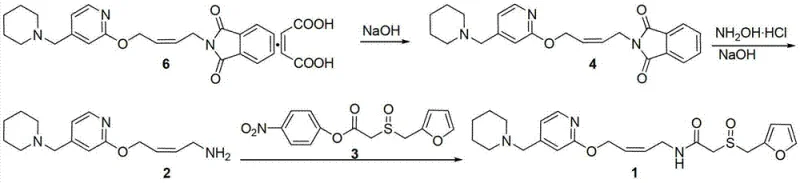
The operational simplicity of this new route is equally compelling for industrial scale-up. Hydroxylamine hydrochloride is a stable, solid reagent with high purity and excellent shelf-life, contrasting sharply with the handling hazards and stability issues of liquid hydrazine or gaseous methylamine. The process demonstrates remarkable efficiency, achieving high conversion rates with stoichiometric control that minimizes waste. By integrating this aminolysis step with a subsequent condensation using an activated ester (Formula 3), the entire synthesis converges on Lafutidine with a total recovery rate that is commercially attractive. This holistic improvement in process design translates directly into a more resilient supply chain, capable of delivering high-quality intermediates with greater predictability and reduced environmental footprint.
Mechanistic Insights into Hydroxylamine-Mediated Aminolysis
The core chemical innovation lies in the specific reactivity profile of hydroxylamine compared to hydrazine. In the conventional Gabriel synthesis, the nucleophilic nitrogen of hydrazine attacks the carbonyl carbons of the phthalimide ring, forming a tetrahedral intermediate that collapses to release the primary amine and phthalhydrazide. However, the adjacent N-N bond in hydrazine renders it susceptible to oxidation-reduction cycles, allowing it to act as a hydrogen donor to the conjugated alkene system of the substrate. Hydroxylamine, possessing an N-O bond, lacks this specific reducing potential under the basic conditions employed (pH ~12 with NaOH). Instead, it functions purely as a nitrogen nucleophile, attacking the imide carbonyls to form a transient hydroxamic acid intermediate which rapidly decomposes to release the free amine and phthalohydroxamic acid byproducts.
This mechanistic distinction is vital for impurity control. By removing the reducing agent from the reaction matrix, the thermodynamic pathway leading to the saturation of the double bond is effectively blocked. The result is a reaction mixture where the primary amine (Formula 2) is generated with high selectivity, free from the dihydro-contaminants that plague other methods. Furthermore, the byproducts of hydroxylamine aminolysis are generally more water-soluble and easier to extract than the bulky phthalhydrazide derivatives formed in hydrazinolysis. This facilitates a cleaner work-up procedure, often requiring simple aqueous washes to remove inorganic salts and polar organic byproducts, leaving the desired lipophilic amine in the organic phase ready for the final coupling step.
How to Synthesize Lafutidine Efficiently
The implementation of this synthesis requires precise control over stoichiometry and reaction conditions to maximize the benefits of the hydroxylamine route. The process begins with the preparation of the phthalimide precursor, followed by the critical aminolysis step where hydroxylamine hydrochloride is reacted with the imide in the presence of a base such as sodium hydroxide. The choice of solvent, typically ethanol or methanol, plays a crucial role in solubilizing the reagents while maintaining a homogeneous reaction environment. Following the isolation of the pure amine intermediate, the final step involves a condensation reaction with 2-[(2-furylmethyl)sulfinyl]acetic acid p-nitrophenyl ester. Detailed standard operating procedures for each stage, including specific molar ratios and temperature profiles, are essential for reproducibility.
- React N-[4-[4-(piperidinomethyl)-2-pyridyl]oxy-(Z)-2-butenyl]phthalimide (Formula 4) with hydroxylamine hydrochloride and sodium hydroxide in ethanol to obtain the primary amine intermediate (Formula 2).
- Condense the resulting amine (Formula 2) with 2-[(2-furylmethyl)sulfinyl]acetic acid p-nitrophenyl ester (Formula 3) under mild conditions.
- Purify the final crude product via recrystallization to achieve Lafutidine (Formula 1) with purity exceeding 99.88%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this hydroxylamine-based synthesis offers tangible economic and logistical benefits that extend beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the purification train. By preventing the formation of hard-to-remove dihydro-impurities at the source, manufacturers can eliminate multiple recrystallization cycles, which are resource-intensive and yield-dilutive. This streamlining of the downstream process directly correlates to a significant reduction in manufacturing costs, driven by lower solvent usage, reduced energy consumption for heating and cooling, and decreased labor hours for filtration and drying operations. The ability to achieve >99% purity with fewer unit operations creates a leaner, more cost-effective production model.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents like hydrazine hydrate, coupled with the use of stable, commodity-grade hydroxylamine hydrochloride, lowers raw material costs. Furthermore, the avoidance of complex purification steps reduces the overall cost of goods sold (COGS) by minimizing material loss during processing. The higher selectivity of the reaction ensures that a greater proportion of input materials are converted into saleable product, enhancing the overall economic efficiency of the manufacturing campaign without compromising on quality standards.
- Enhanced Supply Chain Reliability: Relying on solid, stable reagents like hydroxylamine hydrochloride mitigates the risks associated with the degradation of liquid amine solutions during storage and transport. This stability ensures consistent batch quality and reduces the likelihood of production delays caused by out-of-spec raw materials. Additionally, the robustness of the reaction conditions, which tolerate ambient temperatures and common solvents, makes the process less sensitive to minor operational fluctuations, thereby guaranteeing a steady and reliable supply of high-purity Lafutidine intermediates to meet market demand.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing standard reactor configurations and avoiding the need for specialized high-pressure or cryogenic equipment. From an environmental perspective, the reduction in solvent waste and the absence of toxic hydrazine residues simplify effluent treatment and disposal, aligning with increasingly stringent global environmental regulations. This green chemistry profile not only reduces compliance costs but also enhances the sustainability credentials of the supply chain, a factor of growing importance to downstream pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthetic route. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these specifics helps stakeholders evaluate the feasibility of integrating this technology into their existing manufacturing portfolios.
Q: Why is hydroxylamine hydrochloride preferred over hydrazine hydrate for Lafutidine synthesis?
A: Hydrazine hydrate possesses strong reducing properties that inadvertently reduce the double bond in the crotyl chain, generating difficult-to-remove dihydro-lafutidine impurities. Hydroxylamine hydrochloride acts purely as a nucleophile for aminolysis without reducing the alkene, ensuring higher chemical purity.
Q: What is the expected purity profile of Lafutidine produced via this novel route?
A: The patented method achieves a final product purity of up to 99.88% by HPLC. Crucially, it eliminates the formation of dihydro-lafutidine (Formula 7), a major impurity in conventional Gabriel synthesis routes that often requires complex recrystallization steps to control.
Q: Is this synthesis route scalable for industrial production?
A: Yes, the process utilizes stable solid reagents like hydroxylamine hydrochloride instead of unstable liquid methylamine or hazardous hydrazine. The reaction proceeds efficiently at room temperature in common solvents like ethanol, facilitating safe and straightforward scale-up from pilot to commercial tonnage.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Lafutidine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes requires a partner with deep technical expertise and proven scale-up capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the hydroxylamine aminolysis method are fully realized in a GMP-compliant environment. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify that every batch of Lafutidine intermediate meets the highest global standards for potency and impurity control.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this superior manufacturing technology. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to request specific COA data and comprehensive route feasibility assessments, ensuring that your supply chain is optimized for both quality and efficiency in the competitive landscape of gastrointestinal therapeutics.
