Advanced Synthesis of Venetoclax Key Intermediates for Commercial Scale Manufacturing
Advanced Synthesis of Venetoclax Key Intermediates for Commercial Scale Manufacturing
The pharmaceutical landscape for oncology treatments continues to evolve rapidly, with Venetoclax (ABT-199) standing out as a pivotal BCL-2 inhibitor for treating chronic lymphocytic leukemia. The efficient production of its key building blocks is critical for meeting global demand. Patent CN108059599B discloses a groundbreaking preparation method for the vital intermediate, methyl 2-(4-chlorophenyl)-4,4-dimethylcyclohex-1-ene-1-carboxylate. This technology represents a significant leap forward in process chemistry, addressing long-standing challenges related to yield, safety, and cost-efficiency. By shifting away from hazardous reagents and complex purification steps, this innovation offers a streamlined pathway that aligns perfectly with modern green chemistry principles and industrial scalability requirements.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this critical intermediate has relied on routes that pose significant operational and economic hurdles for manufacturers. Traditional methods typically involve the reaction of methyl 4,4-dimethyl-2-cyclohexanone formate with trifluoromethanesulfonic anhydride to generate a triflate intermediate, followed by a Suzuki coupling with p-chlorobenzeneboronic acid. As illustrated in the reaction scheme below, this conventional pathway is fraught with difficulties.
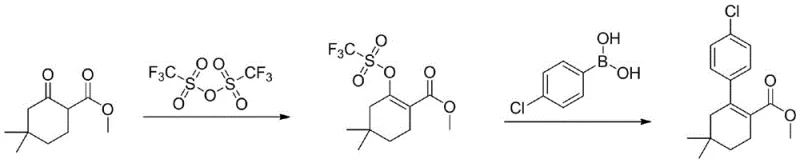
The reliance on trifluoromethanesulfonic anhydride introduces severe safety concerns due to its strong corrosivity and handling risks. Furthermore, these legacy processes often demand high loadings of palladium catalysts, sometimes as high as 0.1 equivalent, which drastically inflates raw material costs. The instability of the triflate intermediate often leads to the generation of diboron ester byproducts during coupling, resulting in a complex impurity profile. Consequently, extensive post-treatment involving silica gel column chromatography is required, which is impractical for large-scale production and makes controlling heavy metal residues to pharmaceutical standards exceptionally difficult.
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent data offers a robust and economically superior alternative. This innovative route bypasses the formation of the unstable triflate intermediate entirely. Instead, it employs a direct one-pot coupling strategy utilizing p-chlorobromobenzene, an inorganic base, and an aryl sulfonyl hydrazide under palladium catalysis. The reaction scheme for this advanced process is depicted below, highlighting the streamlined nature of the transformation.
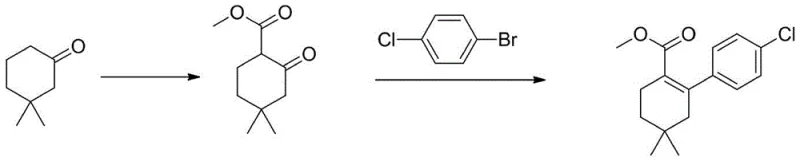
This approach not only simplifies the operational workflow but also significantly enhances the overall yield and product quality. By utilizing p-chlorobromobenzene instead of boronic acids, the process leverages more stable and cost-effective starting materials. The elimination of the triflation step removes the need for hazardous reagents, while the optimized catalytic system allows for much lower palladium loading. The final product can be purified through simple recrystallization rather than column chromatography, ensuring high purity and facilitating easier regulatory compliance for commercial manufacturing.
Mechanistic Insights into Palladium-Catalyzed One-Pot Coupling
The core of this technological breakthrough lies in the sophisticated mechanistic interplay between the palladium catalyst and the aryl sulfonyl hydrazide. The reaction initiates with the deprotonation of 3,3-dimethylcyclohexanone using a strong base such as LDA or HMDSLi at low temperatures, typically ranging from -20°C to -30°C. This generates a reactive enolate species which subsequently attacks dimethyl carbonate to form the crucial methyl 4,4-dimethyl-2-cyclohexanone formate intermediate. This first step is critical for establishing the correct carbon skeleton with high regioselectivity, ensuring that the subsequent coupling occurs at the desired position on the cyclohexane ring.
In the second stage, the palladium-catalyzed coupling proceeds through a unique mechanism involving the in situ generation of a diazo species from the aryl sulfonyl hydrazide. This species interacts with the palladium center to facilitate the cross-coupling with p-chlorobromobenzene. The use of inorganic bases like potassium tert-butoxide or cesium carbonate plays a pivotal role in neutralizing acidic byproducts and maintaining the catalytic cycle. Crucially, this mechanism minimizes the formation of homocoupling impurities often seen in traditional Suzuki reactions. The result is a cleaner reaction profile that allows for the isolation of the target molecule with purity exceeding 99% after a single recrystallization step from methanol, effectively controlling the impurity spectrum without the need for chromatographic separation.
How to Synthesize Methyl 2-(4-chlorophenyl)-4,4-dimethylcyclohex-1-ene-1-carboxylate Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize yield and safety. The process begins with the careful preparation of the keto-ester intermediate under inert atmosphere conditions to prevent moisture interference. Following this, the one-pot coupling reaction demands strict temperature management, ideally maintained at 110°C in solvents like dioxane or toluene, to ensure complete conversion. The detailed standardized synthetic steps, including specific molar ratios and workup procedures derived from the patent examples, are outlined in the guide below.
- Deprotonate 3,3-dimethylcyclohexanone using LDA or HMDSLi at low temperature (-20°C to -30°C) and react with dimethyl carbonate to form the keto-ester intermediate.
- In a one-pot reaction, combine the keto-ester with p-chlorobromobenzene, an inorganic base, and aryl sulfonyl hydrazide under palladium catalysis.
- Heat the mixture to 110°C in a solvent like dioxane, then purify the final product via methanol recrystallization to achieve >99% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route translates into tangible strategic benefits that extend beyond mere chemical efficiency. The shift away from corrosive and hazardous reagents like trifluoromethanesulfonic anhydride significantly reduces the regulatory burden and safety infrastructure costs associated with manufacturing. Furthermore, the ability to source stable, commodity-grade raw materials such as p-chlorobromobenzene mitigates supply chain risks often associated with specialized boronic acids. This stability ensures a more predictable production schedule and reduces the likelihood of delays caused by raw material shortages or quality fluctuations.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, primarily driven by the drastic reduction in catalyst consumption. By lowering the palladium acetate loading to as little as 0.004 to 0.008 equivalents compared to the 0.1 equivalent required in conventional methods, the direct material cost is significantly optimized. Additionally, the elimination of silica gel column chromatography in favor of recrystallization removes a major bottleneck in processing time and solvent consumption. This simplification of the downstream processing workflow leads to substantial operational expenditure savings, making the final API intermediate much more cost-competitive in the global market.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of robust and widely available starting materials. Unlike sensitive boronic acid derivatives that may degrade during storage or transport, p-chlorobromobenzene and aryl sulfonyl hydrazides are stable solids with long shelf lives. This stability allows manufacturers to maintain strategic stockpiles without significant degradation risks. Moreover, the simplified purification process reduces the dependency on specialized consumables like chromatography media, further insulating the production line from external supply disruptions and ensuring consistent delivery timelines for downstream API manufacturers.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, this route offers a cleaner profile that aligns with increasingly stringent global regulations. The avoidance of heavy waste streams associated with column chromatography and the reduction of heavy metal residues simplify wastewater treatment protocols. The process is inherently safer, avoiding the use of pyrophoric reagents like sodium hydride in critical coupling steps, which facilitates easier technology transfer from pilot plants to multi-ton commercial reactors. This scalability ensures that production capacity can be ramped up rapidly to meet market demand without compromising on safety or environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on performance metrics and operational feasibility. Understanding these details is essential for technical teams evaluating the integration of this process into their existing manufacturing portfolios.
Q: What are the primary advantages of the new synthesis route over conventional methods?
A: The new route eliminates the use of corrosive trifluoromethanesulfonic anhydride and reduces palladium catalyst loading significantly. It replaces complex silica gel column purification with simple recrystallization, drastically lowering production costs and environmental impact.
Q: How does this process ensure high purity for pharmaceutical applications?
A: The process utilizes a robust one-pot coupling mechanism that minimizes side reactions. Final purification is achieved through methanol recrystallization, consistently yielding products with purity exceeding 99%, meeting stringent pharmaceutical standards without heavy metal contamination.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method is designed for scalability. It uses readily available raw materials like p-chlorobromobenzene and avoids hazardous reagents like sodium hydride in critical steps, ensuring safe and stable operation from pilot scale to multi-ton commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Venetoclax Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust supply chains for life-saving oncology medications. Our technical team has thoroughly analyzed the potential of this patented route and is fully equipped to translate these laboratory successes into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. Our state-of-the-art facilities are designed to handle complex organic syntheses with stringent purity specifications, supported by rigorous QC labs that guarantee every batch meets the highest international standards.
We invite you to collaborate with us to leverage this advanced technology for your Venetoclax supply chain. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise can drive efficiency and reliability in your API manufacturing operations.
