Revolutionizing Venetoclax Intermediate Production: A Deep Dive into Patented Synthetic Efficiency
The pharmaceutical landscape for oncology treatments has been profoundly transformed by the advent of BCl-2 selective inhibitors, most notably Venetoclax, which has demonstrated exceptional clinical efficacy in treating relapsed or refractory chronic lymphocytic leukemia. As the demand for this life-saving medication continues to surge globally, the efficiency and robustness of its supply chain have become paramount concerns for major pharmaceutical stakeholders. In this context, the technical disclosure found in patent CN111892591A represents a pivotal advancement in the synthetic methodology for key Venetoclax intermediates. This patent details a novel approach that fundamentally reimagines the construction of the critical ether linkage found in the drug's core structure, moving away from hazardous traditional methods toward a more sustainable and operationally simple catalytic process. For R&D directors and procurement strategists alike, understanding the nuances of this patented technology is essential for securing a reliable pharmaceutical intermediates supplier capable of meeting the rigorous demands of modern API manufacturing. The shift from Grignard-based chemistry to a DMAP-catalyzed esterification followed by nucleophilic substitution not only enhances chemical safety but also opens new avenues for cost reduction in API manufacturing by streamlining process controls and minimizing waste generation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the critical benzoate intermediate required for Venetoclax production has relied heavily on organometallic chemistry, specifically utilizing Grignard reagents such as triisopropyl magnesium chloride. While chemically effective in laboratory settings, this traditional pathway presents severe logistical and safety challenges when translated to an industrial scale. The inherent reactivity of Grignard reagents necessitates strictly anhydrous and anoxic environments, requiring specialized equipment and rigorous inert gas purging systems that significantly inflate capital expenditure and operational complexity. Furthermore, these reactions typically must be conducted at cryogenic temperatures to manage exothermicity and prevent side reactions, leading to substantial energy consumption and extended batch cycle times. The sensitivity of the Grignard species also means that even minor deviations in moisture content can lead to catastrophic yield losses and the formation of difficult-to-remove impurities, thereby compromising the purity profile required for downstream API synthesis. These factors collectively create a fragile supply chain vulnerable to disruptions, making the conventional route less attractive for long-term commercial partnerships seeking stability and predictability.
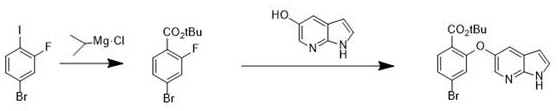
The Novel Approach
In stark contrast to the volatility of organometallic pathways, the innovative method disclosed in the patent utilizes a mild, catalytic esterification strategy followed by a robust nucleophilic aromatic substitution. By employing 4-bromo-2-fluorobenzoic acid as the starting material and reacting it with di-tert-butyl dicarbonate (Boc anhydride) in the presence of a catalytic amount of DMAP, the process achieves efficient protection of the carboxylic acid without the need for hazardous reagents. This esterification step proceeds smoothly in common organic solvents like tetrahydrofuran at temperatures ranging from ambient to moderately elevated levels, completely eliminating the need for cryogenic cooling. The subsequent coupling with 5-hydroxy-7-azaindole is facilitated under basic conditions using inexpensive inorganic bases such as potassium carbonate, further simplifying the reaction setup. This paradigm shift not only mitigates safety risks associated with pyrophoric materials but also dramatically simplifies the work-up procedure, allowing for straightforward aqueous washes and crystallization. For a reliable pharmaceutical intermediates supplier, adopting this methodology translates directly into enhanced process reliability and the ability to deliver high-purity Venetoclax intermediate batches with consistent quality attributes.
Mechanistic Insights into DMAP-Catalyzed Esterification and Substitution
The core of this technological breakthrough lies in the mechanistic efficiency of the 4-dimethylaminopyridine (DMAP) catalyzed esterification. DMAP acts as a potent nucleophilic catalyst, accelerating the acylation of the carboxylic acid by forming a highly reactive acylpyridinium intermediate. This intermediate is significantly more electrophilic than the original anhydride, allowing the reaction to proceed rapidly even at lower catalyst loadings and milder thermal conditions. The patent specifies that DMAP loading can be optimized between 3% and 50% molar equivalents, providing flexibility for process engineers to balance reaction kinetics against catalyst recovery costs. The use of Boc anhydride serves a dual purpose: it acts as the protecting group source and drives the equilibrium forward through the release of stable byproducts. This mechanism ensures that the conversion of 4-bromo-2-fluorobenzoic acid to its tert-butyl ester is nearly quantitative, minimizing the carryover of unreacted acid which could interfere with subsequent steps. The robustness of this catalytic cycle is a key factor in achieving the high yields reported in the patent examples, demonstrating a level of control that is often absent in stoichiometric organometallic transformations.
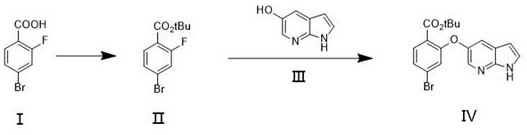
Following the esterification, the formation of the critical ether bond via nucleophilic aromatic substitution (SNAr) is equally critical for maintaining product integrity. The electron-withdrawing nature of the fluorine atom on the aromatic ring activates the substrate for displacement by the phenolic oxygen of the 7-azaindole moiety. The patent highlights the importance of base selection, with potassium carbonate proving effective in deprotonating the phenol to generate the nucleophilic phenoxide ion in situ. This reaction is performed in polar aprotic solvents like acetonitrile, which stabilize the transition state and facilitate ion pairing. Crucially, the mild basic conditions avoid the degradation of the sensitive azaindole ring system, a common issue in harsher alkaline environments. Impurity control is inherently built into this mechanism; by avoiding the aggressive conditions of Grignard chemistry, the formation of homocoupling byproducts or debromination impurities is significantly suppressed. This results in a crude product profile that is much cleaner, reducing the burden on downstream purification units and ensuring that the final high-purity Venetoclax intermediate meets stringent regulatory specifications with minimal processing.
How to Synthesize Venetoclax Key Intermediate Efficiently
Implementing this synthetic route in a production environment requires a clear understanding of the operational parameters defined in the patent to ensure reproducibility and safety. The process is divided into two distinct stages: the catalytic esterification to form the protected benzoate, and the subsequent etherification to construct the final heterocyclic scaffold. Operators must pay close attention to the molar ratios of reagents, particularly the excess of Boc anhydride which drives the first step to completion, and the stoichiometry of the base in the second step to ensure full conversion of the phenol. The simplicity of the work-up procedures, involving standard acid-base washes and solvent evaporation, makes this route particularly attractive for facilities looking to optimize throughput without investing in exotic containment systems. Detailed standardized synthesis steps see the guide below for specific procedural instructions.
- Perform DMAP-catalyzed esterification of 4-bromo-2-fluorobenzoic acid with Boc anhydride in THF at 20-100°C.
- Conduct nucleophilic substitution with 5-hydroxy-7-azaindole under basic conditions (K2CO3) in acetonitrile at 0-80°C.
- Execute post-treatment via water quenching, filtration, and ethyl acetate recrystallization to achieve high purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this patented synthetic route offers compelling advantages that extend far beyond simple chemical yield improvements. The elimination of cryogenic requirements and pyrophoric reagents fundamentally alters the cost structure of manufacturing, removing the need for specialized low-temperature reactors and complex inert gas handling infrastructure. This simplification allows for the utilization of standard multipurpose reactor trains, thereby increasing asset utilization rates and reducing the capital barrier for production. Furthermore, the reliance on commercially abundant and stable reagents like Boc anhydride and potassium carbonate insulates the supply chain from the volatility often associated with specialized organometallic suppliers. For supply chain heads, this translates into a more resilient sourcing strategy where raw material availability is guaranteed, and lead times are predictable. The overall process intensification achieved through higher yields and simpler purification directly contributes to cost reduction in API manufacturing, enabling more competitive pricing models for the final drug product without compromising on quality or compliance standards.
- Cost Reduction in Manufacturing: The transition away from Grignard chemistry eliminates the substantial costs associated with maintaining anhydrous and anoxic conditions, which typically require expensive drying agents, nitrogen blankets, and specialized monitoring equipment. By operating at ambient or moderately elevated temperatures, the energy footprint of the process is drastically reduced, leading to significant utility savings over the lifecycle of the product. Additionally, the higher selectivity of the DMAP-catalyzed route minimizes the formation of side products, which reduces the volume of solvents and adsorbents required for purification. This cumulative effect of simplified operations, lower energy consumption, and reduced waste disposal costs creates a leaner manufacturing model that delivers substantial economic value to the procurement organization.
- Enhanced Supply Chain Reliability: The raw materials utilized in this novel pathway, such as 4-bromo-2-fluorobenzoic acid and Boc anhydride, are commodity chemicals produced by multiple global vendors, ensuring a diversified and secure supply base. Unlike specialized Grignard reagents which may have limited suppliers and long lead times due to stability issues, these inputs can be sourced readily, mitigating the risk of production stoppages due to material shortages. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, further enhancing supply continuity. For a reliable pharmaceutical intermediates supplier, this stability is crucial for maintaining long-term contracts and ensuring that downstream API production schedules are met without interruption, thereby strengthening the overall resilience of the pharmaceutical value chain.
- Scalability and Environmental Compliance: The inherent safety of avoiding pyrophoric reagents makes this process exceptionally well-suited for commercial scale-up of complex heterocyclic compounds. Large-scale reactors can be operated with greater confidence, as the risks of thermal runaway or fire are significantly diminished compared to traditional methods. From an environmental standpoint, the reduction in hazardous waste generation aligns with increasingly stringent global regulations regarding chemical manufacturing emissions and effluent treatment. The use of recyclable solvents like THF and acetonitrile, combined with aqueous work-ups, facilitates easier waste stream management and potential solvent recovery loops. This alignment with green chemistry principles not only reduces regulatory compliance burdens but also enhances the corporate sustainability profile of the manufacturing entity, a factor of growing importance to stakeholders and investors in the modern pharmaceutical industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic technology. They are derived from the specific pain points identified in the background of the patent and the beneficial effects observed in the experimental examples. Understanding these aspects is vital for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers provided reflect the objective data and logical deductions presented in the patent documentation, offering clarity on performance expectations and operational requirements.
Q: How does the new DMAP-catalyzed method improve upon traditional Grignard routes?
A: The patented method eliminates the need for hazardous Grignard reagents and strict anhydrous/anoxic conditions, significantly simplifying operation and improving yield stability compared to conventional low-temperature processes.
Q: What are the critical reaction parameters for maximizing yield in this synthesis?
A: Optimal yields are achieved by controlling the DMAP catalyst loading between 3%-50%, maintaining esterification temperatures between 20°C-100°C, and utilizing precise molar ratios of Boc anhydride to acid.
Q: Is this synthetic route scalable for commercial API manufacturing?
A: Yes, the avoidance of cryogenic conditions and the use of common solvents like THF and acetonitrile make this route highly amenable to large-scale commercial production with reduced safety risks.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Venetoclax Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex oncology drugs like Venetoclax depends on a partnership grounded in technical excellence and supply chain integrity. Our team of expert process chemists has extensively analyzed patented methodologies such as CN111892591A to ensure that our manufacturing capabilities are aligned with the most advanced and efficient synthetic routes available. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that whether you require clinical trial materials or full-scale commercial supply, our facilities are equipped to deliver. Our commitment to quality is underscored by our stringent purity specifications and rigorous QC labs, which employ state-of-the-art analytical instrumentation to verify every batch against the highest industry standards. We understand that consistency is key in API synthesis, and our robust quality management systems are designed to guarantee that every gram of intermediate we supply meets the exacting requirements of global regulatory bodies.
We invite you to engage with our technical procurement team to discuss how our manufacturing capabilities can support your specific project needs. By leveraging our expertise in process optimization and scale-up, we can provide a Customized Cost-Saving Analysis tailored to your volume requirements and timeline constraints. We encourage potential partners to request specific COA data and route feasibility assessments to validate our technical claims and explore the potential for collaboration. Whether you are looking to secure a secondary source for an existing program or develop a new supply chain for an emerging therapy, NINGBO INNO PHARMCHEM is positioned to be your strategic partner in delivering high-quality chemical solutions efficiently and reliably.
