Advanced Catalytic Bromination Strategy for High-Purity Lesinurad Intermediate Manufacturing and Commercial Scale-Up
Introduction to Advanced Lesinurad Intermediate Manufacturing
The global pharmaceutical landscape for metabolic disorders continues to evolve, with Hyperuricemia (HUA) and gout representing a significant and growing therapeutic market. At the heart of modern gout treatment lies Lesinurad (Racinard), a potent URAT1 inhibitor approved by major regulatory bodies including the FDA and EMA. However, the commercial viability of such complex small molecules often hinges on the robustness of the synthetic route, particularly for critical intermediates. Patent CN111116501B introduces a transformative synthesis method for the key Lesinurad intermediate, methyl 2-(5-bromo-4-(1-cyclopropylnaphthalen-4-yl)-4H-1,2,4-triazol-3-ylthio)acetate. This innovation addresses long-standing challenges in impurity control, specifically targeting the elimination of persistent chlorinated byproducts and unreacted precursors that have historically plagued the manufacturing process. By shifting from a standard stoichiometric bromination to a catalytically enhanced protocol, this technology offers a pathway to significantly higher purity profiles, directly impacting the safety and efficacy of the final Active Pharmaceutical Ingredient (API).
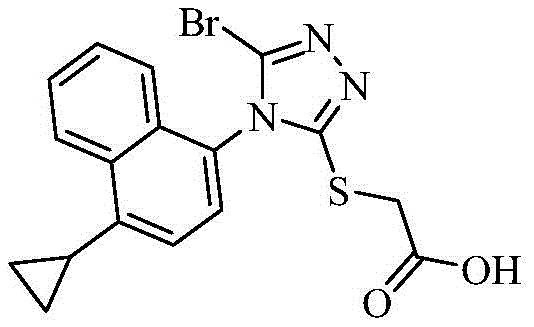
For R&D directors and process chemists, the ability to control the impurity profile at the intermediate stage is paramount. The presence of structurally similar impurities at the intermediate level often leads to carryover into the final drug substance, where separation becomes exponentially more difficult and costly. The methodology described in this patent not only optimizes the chemical transformation but also fundamentally alters the physical properties of the product, converting it from an intractable oil into a crystalline solid. This physical state change is a critical, yet often overlooked, parameter in process chemistry that dictates downstream handling, filtration efficiency, and drying kinetics. As we delve deeper into the technical specifics, it becomes evident that this approach represents a substantial leap forward in the reliable supply of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
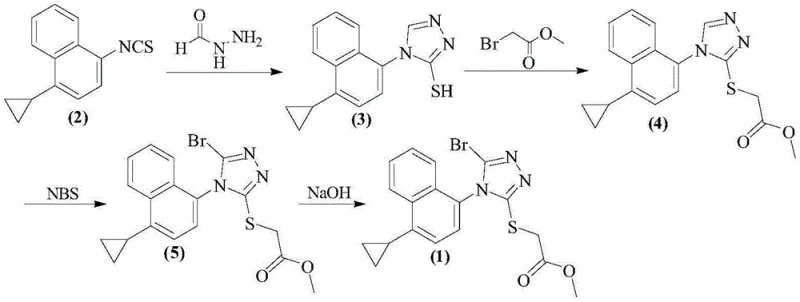
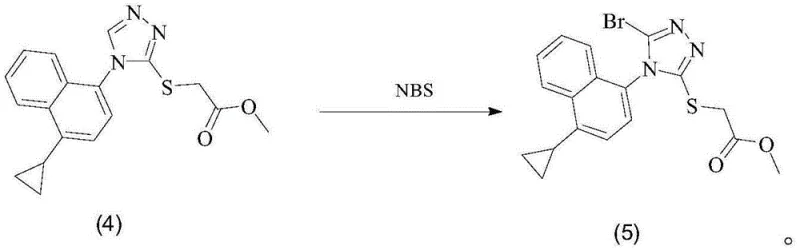
Historically, the synthesis of the brominated triazole intermediate has relied on the direct reaction of the precursor thioether with N-bromosuccinimide (NBS) in tetrahydrofuran (THF). While conceptually straightforward, this conventional approach suffers from severe selectivity issues. The primary drawback is the formation of a chlorinated impurity, 2-((5-chloro-4-(1-cyclopropylnaphthalen-4-yl)-4H-1,2,4-triazol-3-yl)thio)acetic acid, which arises during the bromination step. This chlorinated analog is a nightmare for purification teams because its polarity is nearly identical to that of the desired brominated product and the final Lesinurad API. Consequently, standard recrystallization techniques fail to effectively purge this contaminant, leading to potential failures in meeting stringent pharmacopeial specifications. Furthermore, the conventional method often results in a product that is a yellow oily substance rather than a solid. Oily intermediates are notoriously difficult to handle on a large scale; they trap solvent residues, complicate filtration, and hinder accurate weighing and dosing in subsequent reaction steps. Additionally, residual amounts of the starting material, Intermediate (4), often persist at levels around 5%, necessitating extensive and yield-eroding purification protocols.
The Novel Approach
The innovative strategy disclosed in the patent circumvents these pitfalls through the introduction of specific radical catalysts and a tailored mixed-solvent system. By incorporating catalysts such as TEMPO, BPO, or AIBN into the reaction matrix, the bromination process becomes highly selective and efficient. The use of a mixed solvent system, typically combining THF with methanol or dichloromethane, appears to modulate the solubility and reactivity of the species involved, favoring the formation of the desired bromo-derivative while suppressing the formation of the chlorinated side product. The result is a dramatic improvement in product quality: the intermediate is isolated as a free-flowing off-white or white solid with purity levels exceeding 99.9%. Crucially, analytical data indicates that both the chlorinated impurity and the residual starting material are reduced to undetectable levels. This transition from an oily, impure mixture to a high-purity crystalline solid simplifies the entire downstream process, reducing the burden on purification units and ensuring a consistent, high-quality feed for the final hydrolysis step to generate Lesinurad.
Mechanistic Insights into Catalytic Radical Bromination
The core of this technological advancement lies in the modulation of the radical bromination mechanism. In the absence of a catalyst, the homolytic cleavage of NBS can be sluggish or non-selective, potentially allowing trace chloride sources (perhaps from solvent degradation or reagent impurities) to compete with bromine for the reactive position on the triazole ring. The addition of a radical initiator like TEMPO (2,2,6,6-tetramethylpiperidin-1-yl)oxyl) serves to facilitate the generation of bromine radicals in a controlled manner. TEMPO acts as a stable free radical that can interact with the succinimide radical or the bromine species, effectively lowering the activation energy for the propagation step of the bromination. This ensures that the bromine radical attacks the C-5 position of the 1,2,4-triazole ring with high fidelity. The mixed solvent environment further supports this mechanism; methanol, being a polar protic solvent, may assist in stabilizing transition states or solvating the succinimide byproduct, driving the equilibrium towards product formation. The net effect is a kinetic preference for bromination over any competing halogenation pathways, effectively shutting down the formation of the chlorinated impurity at the source.
From an impurity control perspective, the mechanism also explains the near-total consumption of the starting material. The catalytic cycle ensures that the concentration of active brominating species remains optimal throughout the reaction duration, preventing the stalling of the reaction that often leaves unreacted starting material in conventional batch processes. The rapid and complete conversion means that Intermediate (4) does not accumulate, eliminating the need for complex recycling streams. Furthermore, the crystallization behavior of the product is enhanced by the absence of oily impurities. In crystallization science, the presence of structurally similar impurities (like the chloro-analog) can inhibit crystal growth or lead to oiling out. By virtually eliminating these impurities, the new method allows the product to nucleate and grow into well-defined crystals upon the addition of anti-solvents like n-hexane. This mechanistic understanding provides R&D teams with the confidence to scale the process, knowing that the reaction is robust against minor variations in reagent quality due to the buffering effect of the catalyst.
How to Synthesize Methyl 2-(5-bromo-4-(1-cyclopropylnaphthalen-4-yl)-4H-1,2,4-triazol-3-ylthio)acetate Efficiently
Implementing this synthesis requires precise control over reaction parameters to maximize the benefits of the catalytic system. The process begins with the dissolution of the precursor Intermediate (4) in a specific ratio of tetrahydrofuran and a co-solvent such as methanol. The mixture is heated to a moderate temperature range of 30-40°C to ensure homogeneity before the introduction of the catalyst. Once the catalyst (preferably TEMPO) is added, N-bromosuccinimide is introduced portion-wise to manage the exotherm and maintain steady radical concentration. Following the reaction completion, a standard aqueous workup removes the succinimide byproduct, and the crude product is subjected to a recrystallization step using ethyl acetate and n-hexane. This final purification step is critical for locking in the high purity and achieving the desired solid-state form. For detailed operational parameters and safety considerations, please refer to the standardized synthesis guide below.
- Dissolve reactant Intermediate (4) in a mixed solvent system comprising tetrahydrofuran and methanol, heating to 30-40°C to ensure complete solubility.
- Introduce a radical catalyst such as TEMPO, followed by the portion-wise addition of N-bromosuccinimide (NBS), maintaining the reaction temperature between 30-40°C for approximately 60 minutes.
- Perform aqueous workup to remove succinimide byproducts, concentrate the organic phase, and recrystallize the crude solid from ethyl acetate and n-hexane to obtain the refined white solid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the shift to this catalytic synthesis method translates into tangible operational efficiencies and risk mitigation. The primary value driver is the drastic simplification of the purification workflow. In the conventional process, removing chlorinated impurities and residual starting materials often requires multiple recrystallizations or even preparative chromatography, both of which are time-consuming and expensive. By delivering an intermediate with undetectable levels of these critical impurities, the new method eliminates these costly purification bottlenecks. This streamlining directly contributes to cost reduction in pharmaceutical intermediate manufacturing by reducing solvent consumption, labor hours, and equipment occupancy time. Furthermore, the conversion of the product from a viscous oil to a free-flowing solid has profound implications for logistics and handling. Solids are far easier to transport, store, and dose accurately than oils, reducing the risk of material loss and cross-contamination between batches.
- Cost Reduction in Manufacturing: The elimination of difficult-to-remove impurities means that downstream processing becomes significantly more efficient. Without the need for aggressive purification steps to strip out chlorinated analogs, manufacturers can achieve higher overall yields and lower cost of goods sold (COGS). The use of common, inexpensive solvents like THF, methanol, and hexane further ensures that raw material costs remain stable and predictable, avoiding the volatility associated with specialized or hazardous reagents.
- Enhanced Supply Chain Reliability: The robustness of the catalytic reaction ensures consistent batch-to-batch quality. In supply chain management, consistency is key to maintaining production schedules. The high purity of the intermediate reduces the likelihood of batch failures during the final API synthesis, thereby securing the continuity of supply for the finished drug product. Additionally, the solid form of the intermediate improves shelf-life stability, reducing the risk of degradation during storage and transit, which is a common issue with oily chemical intermediates.
- Scalability and Environmental Compliance: The process utilizes standard industrial solvents and operates at mild temperatures (30-40°C), making it inherently safer and easier to scale from pilot plant to commercial tonnage. The reduction in waste generation, stemming from higher selectivity and fewer purification cycles, aligns with green chemistry principles. This not only lowers waste disposal costs but also simplifies regulatory compliance regarding environmental emissions, a critical factor for modern chemical manufacturing facilities aiming for sustainability certifications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical benefits of adopting this catalytic route over traditional methods.
Q: Why is the removal of chlorinated impurities critical in Lesinurad synthesis?
A: Chlorinated impurities possess polarity characteristics nearly identical to the final Lesinurad API, making them exceptionally difficult to separate via standard crystallization or chromatography, thereby posing a significant risk to drug safety and regulatory compliance.
Q: What represents the primary advantage of the catalytic method over conventional NBS bromination?
A: The catalytic method utilizing TEMPO or BPO in mixed solvents transforms the product from a difficult-to-handle yellow oil into a free-flowing off-white solid, while simultaneously reducing residual starting material and halogenated byproducts to undetectable levels.
Q: Which catalysts are effective for this specific bromination transformation?
A: The patent identifies three effective radical initiators: TEMPO (2,2,6,6-Tetramethylpiperidin-1-yl)oxyl), BPO (Benzoyl Peroxide), and AIBN (Azobisisobutyronitrile), with TEMPO demonstrating superior performance in purity enhancement.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Lesinurad Intermediate Supplier
The synthesis of complex heterocyclic intermediates like the Lesinurad precursor requires not just chemical knowledge but deep engineering expertise. NINGBO INNO PHARMCHEM stands at the forefront of this capability, leveraging advanced catalytic technologies to deliver superior chemical building blocks. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless. We understand that in the pharmaceutical sector, purity is non-negotiable. Our stringent purity specifications and rigorous QC labs guarantee that every batch of intermediate meets the highest international standards, minimizing the risk of downstream processing issues for our partners.
We invite global pharmaceutical companies and contract manufacturing organizations to collaborate with us on optimizing their supply chains for gout therapeutics. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific production volumes and quality requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us demonstrate how our mastery of catalytic bromination and impurity control can enhance the efficiency and reliability of your Lesinurad manufacturing program.
