Advanced Synthesis of 4-Methoxymethyl-2,3,5,6-Tetrafluorobenzyl Alcohol for Commercial Scale-Up
Advanced Synthesis of 4-Methoxymethyl-2,3,5,6-Tetrafluorobenzyl Alcohol for Commercial Scale-Up
The global demand for high-efficiency, low-toxicity pyrethroid insecticides continues to drive innovation in the synthesis of their critical precursors. Patent CN102731269B discloses a robust and economically viable synthesis method for 4-methoxymethyl-2,3,5,6-tetrafluorobenzyl alcohol, a pivotal intermediate in the production of tetrafluorobenzene derivatives used in sanitary pest control. This technical breakthrough addresses long-standing challenges in selectivity and reagent toxicity that have plagued previous manufacturing protocols. By shifting from direct methylation strategies to a controlled halogenation-methoxylation-lithiation sequence, the process achieves superior reaction selectivity and product purity. For R&D directors and procurement specialists, understanding this pathway is essential for securing a reliable agrochemical intermediate supplier capable of delivering consistent quality at scale.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this fluorinated benzyl alcohol derivative has relied on routes that suffer from inherent chemical inefficiencies and safety hazards. One prevalent conventional method involves the direct selective methylation of tetrafluoro terephthalyl alcohol. As illustrated in the reaction scheme below, this approach attempts to methylate only one of the two hydroxymethyl groups. However, due to the similar reactivity of both hydroxyl groups, this reaction inevitably generates significant amounts of dimethoxy byproducts. This lack of selectivity drastically increases the difficulty of downstream purification, requiring complex chromatographic or crystallization steps that reduce overall yield and increase manufacturing costs.
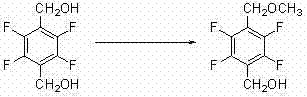
Furthermore, alternative historical routes have attempted to bypass selectivity issues by employing harsh reagents such as dimethyl sulfate or methyl chloride. These reagents pose severe occupational health risks and environmental compliance burdens. Another documented approach involves a multi-step sequence starting from 2,3,5,6-tetrafluorobenzyl alcohol, proceeding through methylation, carboxylation to an acid, and finally a reduction step back to the alcohol. As shown in the following diagram, this detour through a carboxylic acid intermediate necessitates the use of expensive reducing agents and adds unnecessary operational complexity, making it less attractive for large-scale commercial production.
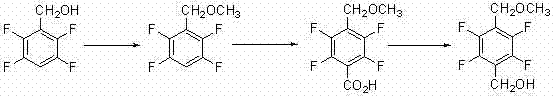
The Novel Approach
The methodology outlined in patent CN102731269B represents a paradigm shift by decoupling the functionalization steps to ensure absolute control over the molecular architecture. Instead of struggling with the similar reactivity of two hydroxyl groups, the novel process begins with the conversion of 2,3,5,6-tetrafluorobenzyl alcohol into a monohalomethyl intermediate using hydrogen halides. This transformation is highly selective and avoids the formation of di-substituted impurities. Subsequently, the halomethyl group undergoes a clean nucleophilic substitution with methanol under mild basic conditions to install the methoxymethyl moiety. This stepwise logic effectively eliminates the formation of the troublesome dimethoxy byproducts seen in direct methylation routes.
The final stage employs modern organometallic chemistry to introduce the second hydroxymethyl group with precision. By utilizing an organolithium reagent at controlled low temperatures followed by the introduction of formaldehyde gas, the process achieves high regioselectivity without the need for protecting groups or excessive reaction steps. This streamlined three-step sequence not only improves the theoretical atom economy but also significantly simplifies the workup procedures. For supply chain managers, this translates to a more robust process with fewer unit operations, reducing the potential for batch-to-batch variability and ensuring a steady supply of high-purity material.
Mechanistic Insights into Organolithium-Mediated Formylation
The cornerstone of this synthesis is the final lithiation-formylation step, which requires precise control over reaction parameters to maximize yield and minimize side reactions. The mechanism involves the deprotonation or lithium-halogen exchange (depending on the specific precursor used in the final step context, though here it is lithiation of the aromatic ring or side chain activation) to generate a reactive nucleophilic species. In this specific patent embodiment, the reaction of 3-methoxymethyl-1,2,4,5-tetrafluorobenzene with an organolithium reagent at temperatures ranging from 0 to -78°C generates a stabilized carbanion intermediate. The low temperature is critical to prevent unwanted side reactions such as nucleophilic aromatic substitution on the electron-deficient tetrafluoro ring, which could lead to defluorination impurities.
Once the organolithium species is formed, the introduction of dry formaldehyde gas serves as the electrophile. The use of gaseous formaldehyde, generated in situ from paraformaldehyde or trioxymethylene, ensures a high concentration of the reactive monomer without introducing water or methanol stabilizers that could quench the sensitive lithiated intermediate. The subsequent quenching with a protic solvent protonates the alkoxide intermediate to yield the target 4-methoxymethyl-2,3,5,6-tetrafluorobenzyl alcohol. This mechanistic pathway is superior to the older acid-reduction routes because it constructs the carbon-carbon bond directly, avoiding the redox inefficiencies of converting an alcohol to an acid and back to an alcohol. The result is a cleaner impurity profile, which is paramount for meeting the stringent specifications required by pharmaceutical and agrochemical clients.
How to Synthesize 4-Methoxymethyl-2,3,5,6-Tetrafluorobenzyl Alcohol Efficiently
The patented procedure offers a clear roadmap for laboratory and pilot-scale synthesis, emphasizing the importance of temperature control and reagent stoichiometry. The process begins with the halogenation of the starting alcohol, followed by methoxylation, and concludes with the lithiation step. Each stage has been optimized to balance reaction rate with selectivity, ensuring that the final product meets high-purity standards suitable for downstream coupling reactions. The detailed standardized synthesis steps, including specific solvent ratios and addition rates, are provided in the guide below to assist technical teams in replicating this efficient route.
- React 2,3,5,6-tetrafluorobenzyl alcohol with hydrogen halide at 40-120°C to obtain the halomethyl intermediate.
- Perform nucleophilic substitution with methanol and inorganic base at 0-65°C to form the methoxymethyl derivative.
- Conduct lithiation at low temperature followed by formaldehyde gas introduction and quenching to yield the final alcohol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis method offers tangible benefits beyond mere chemical elegance. The primary advantage lies in the substantial cost reduction in agrochemical intermediate manufacturing achieved by eliminating expensive and hazardous reagents. By replacing toxic dimethyl sulfate with inexpensive hydrogen halides and methanol, the raw material costs are significantly lowered. Furthermore, the avoidance of the reduction step found in prior art removes the need for costly reducing agents and the associated waste disposal costs, leading to a more economically sustainable process.
- Cost Reduction in Manufacturing: The elimination of the carboxylic acid reduction step fundamentally alters the cost structure of the synthesis. Traditional routes requiring reduction consume significant amounts of energy and expensive hydride reagents, whereas this novel route utilizes a direct formylation strategy. Additionally, the high selectivity of the halogenation-methoxylation sequence minimizes the loss of valuable fluorinated starting materials to byproducts. This improvement in yield directly correlates to a lower cost per kilogram of the final active intermediate, providing a competitive edge in pricing negotiations.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as hydrogen chloride, methanol, and formaldehyde ensures that the supply chain is not vulnerable to the shortages often associated with specialized or highly regulated reagents. The process operates under relatively mild pressure and temperature conditions in the early stages, reducing the risk of equipment failure and unplanned downtime. This robustness ensures consistent delivery schedules, allowing downstream manufacturers to maintain lean inventory levels without fear of supply disruption.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route is markedly superior. The removal of dimethyl sulfate, a known carcinogen, simplifies regulatory compliance and reduces the burden on waste treatment facilities. The process generates fewer heavy metal wastes compared to reduction-based routes, facilitating easier disposal and lowering environmental fees. The scalability is further enhanced by the use of standard unit operations like reflux and gas sparging, which are easily transferred from pilot plants to multi-ton commercial reactors without significant engineering hurdles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of this fluorinated intermediate. These answers are derived directly from the technical specifications and comparative data provided in the patent literature, offering clarity on process capabilities and quality assurance measures.
Q: Why is this new synthesis method superior to direct methylation?
A: Direct methylation of tetrafluoro terephthalyl alcohol often leads to dimethoxy byproducts due to poor selectivity between the two hydroxyl groups. The patented method uses a stepwise halogenation-methoxylation sequence that ensures mono-substitution, significantly simplifying purification.
Q: Does this process avoid toxic reagents like dimethyl sulfate?
A: Yes, the novel route utilizes hydrogen halides and methanol with inorganic bases, completely avoiding the use of highly toxic dimethyl sulfate or gaseous methyl chloride found in conventional methods, thereby improving workplace safety and environmental compliance.
Q: How does the lithiation step impact product purity?
A: The use of organolithium reagents at controlled low temperatures (-30 to -70°C) allows for precise regioselective functionalization. Coupled with formaldehyde gas introduction, this avoids the multi-step oxidation-reduction sequences of older routes, resulting in higher overall purity and yield.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Methoxymethyl-2,3,5,6-Tetrafluorobenzyl Alcohol Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of next-generation agrochemicals depends on the availability of high-quality intermediates. Our technical team has extensively analyzed the pathway described in CN102731269B and possesses the expertise to implement this advanced synthesis at an industrial level. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 4-methoxymethyl-2,3,5,6-tetrafluorobenzyl alcohol meets the exacting standards required for pyrethroid synthesis.
We invite you to collaborate with us to optimize your supply chain for this critical building block. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this novel route can improve your bottom line. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us support your journey toward more efficient and sustainable chemical manufacturing.
