Scalable Production of 4-Methoxymethyl-2,3,5,6-Tetrafluorobenzyl Alcohol via Selective Monomethylation
Scalable Production of 4-Methoxymethyl-2,3,5,6-Tetrafluorobenzyl Alcohol via Selective Monomethylation
The global demand for high-performance agrochemical intermediates continues to drive innovation in fluorinated organic synthesis, particularly for compounds exhibiting potent insecticidal activity. A pivotal advancement in this domain is documented in Chinese Patent CN1176055C, which discloses a highly efficient production method for 4-methoxymethyl-2,3,5,6-tetrafluorobenzyl alcohol. This specific intermediate is critical for the synthesis of ester compounds known for their excellent biological efficacy, yet traditional manufacturing pathways have historically been plagued by excessive step counts and poor economic feasibility. The referenced patent introduces a breakthrough selective monomethylation strategy that utilizes 2,3,5,6-tetrafluoro-1,4-benzenedimethanol as a symmetric, readily available starting material. By shifting the paradigm from complex multi-step functionalization to a direct, controlled alkylation, this technology offers a robust solution for manufacturers seeking to optimize yield and purity simultaneously. For R&D directors and procurement specialists alike, understanding the nuances of this process is essential for securing a reliable agrochemical intermediate supplier capable of delivering consistent quality at scale.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies for constructing the 4-methoxymethyl-2,3,5,6-tetrafluorobenzyl alcohol scaffold often relied on 1,2,4,5-tetrafluorobenzene as the foundational feedstock. This conventional approach necessitates the sequential and differentiated attachment of methoxymethyl and hydroxymethyl functional groups onto the benzene ring, a chemically arduous task that inherently suffers from low regioselectivity and cumbersome protection-deprotection sequences. As illustrated in the reaction scheme below, the requirement to distinguish between identical positions on the aromatic ring leads to a proliferation of synthetic steps, each introducing potential yield losses and impurity profiles that complicate downstream purification. Consequently, these legacy methods fail to meet the rigorous economic criteria required for large-scale industrial production, resulting in inflated manufacturing costs and extended lead times that strain supply chain continuity.
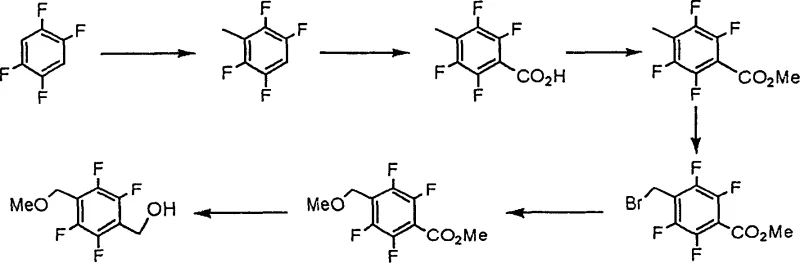
The Novel Approach
In stark contrast to the convoluted legacy pathways, the novel approach detailed in the patent leverages the inherent symmetry of 2,3,5,6-tetrafluoro-1,4-benzenedimethanol to streamline the synthesis into a concise, high-yielding operation. By utilizing a substrate where both hydroxymethyl groups are chemically equivalent, the process eliminates the need for complex regio-control strategies associated with asymmetric starting materials. The core innovation lies in the selective mono-alkylation of one hydroxyl group while preserving the other, achieved through precise control of reaction conditions rather than steric hindrance or protecting groups. This strategic pivot not only drastically reduces the number of unit operations but also enhances the overall atom economy of the process. The result is a manufacturing protocol that is significantly more economical and environmentally benign, positioning it as the preferred route for cost reduction in agrochemical manufacturing for forward-thinking enterprises.
Mechanistic Insights into Selective Monomethylation
The chemical elegance of this process resides in the biphasic reaction system comprising water and a water-immiscible organic solvent, such as toluene or methyl tert-butyl ether. The mechanism initiates with the formation of a mono-alkali metal salt of the starting diol using inorganic bases like sodium hydroxide or potassium hydroxide. Crucially, the reaction temperature is maintained within a moderate range of 15°C to 65°C to balance reaction kinetics with selectivity. The subsequent addition of dimethyl sulfate acts as the methylating agent, reacting preferentially with the generated alkoxide species in the aqueous phase or at the interface. The presence of the organic solvent facilitates the extraction of the product as it forms, driving the equilibrium forward and preventing the accumulation of reactive intermediates that could lead to side reactions. This biphasic design is instrumental in managing the exothermic nature of the alkylation while ensuring efficient mass transfer between the inorganic base and the organic electrophile.
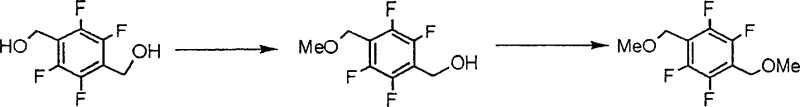
Impurity control is arguably the most critical aspect of this mechanism, specifically regarding the suppression of the dialkylated by-product, 1,4-di(methoxymethyl)-2,3,5,6-tetrafluorobenzene. The patent explicitly highlights that the absence of quaternary ammonium salts, which are commonly used as phase transfer catalysts, is vital for maintaining high selectivity. The presence of such catalysts would aggressively promote the second alkylation step, leading to significant contamination that is difficult to remove. Furthermore, maintaining the aqueous phase pH above 10, and ideally above 13, ensures that the concentration of the mono-anion remains optimal relative to the neutral diol, thereby kinetically favoring the formation of the mono-ether. Post-reaction, the crude product can be purified to exceptional standards, with GC area percentages reaching 98.5% after crystallization, demonstrating the robustness of this mechanistic design in delivering high-purity agrochemical intermediates suitable for sensitive biological applications.
How to Synthesize 4-Methoxymethyl-2,3,5,6-Tetrafluorobenzyl Alcohol Efficiently
Implementing this synthesis requires strict adherence to the sequential addition protocols and stoichiometric ratios defined in the patent to maximize the yield of the mono-alkylated species. The process begins with the dissolution of the symmetric diol in an aqueous base, followed by the controlled introduction of the methylating agent in the presence of an extracting solvent. Operators must monitor the pH and temperature closely to prevent the runaway formation of the dimethyl ether impurity. While the general workflow is straightforward, the specific nuances of workup and purification—such as acidification of the aqueous layer to pH 3 prior to extraction—are critical for recovering the maximum amount of product. For a detailed breakdown of the standardized operating procedures, including specific reagent grades and mixing times, please refer to the technical guide below.
- React 2,3,5,6-tetrafluoro-1,4-benzenedimethanol with an inorganic base (NaOH/KOH) in water at 15°C-65°C to form the mono-salt.
- Add dimethyl sulfate and a water-immiscible organic solvent (e.g., toluene) to the mixture, maintaining pH > 10 to ensure selective mono-alkylation.
- Separate the organic layer, extract the aqueous phase if necessary, and purify the crude product via crystallization to achieve >98% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this patented methodology represents a strategic opportunity to de-risk the supply of critical fluorinated building blocks. The shift from scarce, multi-step precursors to a commoditized, symmetric diol fundamentally alters the cost structure of the final intermediate. By eliminating the need for expensive protecting groups and reducing the total number of reaction vessels required, the process inherently lowers the capital expenditure and operational overhead associated with production. This efficiency translates directly into a more competitive pricing model without compromising on the stringent quality specifications demanded by the agrochemical sector. Furthermore, the reliance on common industrial solvents like toluene and ethyl acetate ensures that the supply chain remains resilient against fluctuations in specialty chemical availability.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and complex protection steps significantly simplifies the bill of materials, leading to substantial cost savings in raw material procurement. The high selectivity of the reaction minimizes the generation of waste by-products, thereby reducing the burden and expense associated with waste treatment and disposal. Additionally, the ability to recover unreacted starting material and recycle solvents further enhances the overall economic viability of the process, making it a superior choice for cost-sensitive large-volume applications.
- Enhanced Supply Chain Reliability: Utilizing 2,3,5,6-tetrafluoro-1,4-benzenedimethanol as a starting material leverages a supply chain that is more mature and stable compared to specialized fluorinated benzenes. The robustness of the reaction conditions, which tolerate moderate temperatures and standard pressures, reduces the risk of batch failures due to equipment sensitivity. This reliability ensures consistent delivery schedules, allowing downstream manufacturers to maintain lean inventory levels while securing a steady flow of high-quality intermediates for their own synthesis campaigns.
- Scalability and Environmental Compliance: The process is designed with commercial scale-up in mind, utilizing standard stirred-tank reactors and liquid-liquid separation techniques that are easily adaptable from pilot to multi-ton scales. The avoidance of hazardous phase transfer catalysts and the use of aqueous workups align with modern green chemistry principles, facilitating easier regulatory compliance and environmental permitting. This scalability ensures that the production capacity can be rapidly expanded to meet surging market demand without the need for bespoke engineering solutions or prolonged validation periods.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this intermediate into their existing supply chains. For further clarification on specific batch records or customization options, direct consultation with our technical team is recommended.
Q: How does this method prevent over-alkylation to the dimethyl ether?
A: The process strictly controls the pH above 10 (preferably >13) and avoids quaternary ammonium phase transfer catalysts, which significantly suppresses the formation of the 1,4-di(methoxymethyl) by-product.
Q: What is the expected purity after crystallization?
A: According to patent data, crystallization from solvents like toluene/hexane can elevate the GC area percentage of the target compound to 98.5%, with by-products reduced to 0.2%.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the use of readily available symmetric starting materials, aqueous inorganic bases, and standard organic solvents like toluene makes the process economically viable and easily scalable compared to multi-step conventional routes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Methoxymethyl-2,3,5,6-Tetrafluorobenzyl Alcohol Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced agrochemicals depends on the availability of high-purity intermediates produced via robust and scalable pathways. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch meets the exacting standards required for pesticide synthesis. Our commitment to quality assurance means that clients can rely on us not just as a vendor, but as a strategic partner dedicated to the success of their final product formulations.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic advantages tailored to your volume needs. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific excellence and commercial reliability. Let us help you secure the high-purity agrochemical intermediates necessary to drive your innovation forward.
