Industrial Synthesis of Chenodeoxycholic Acid: A Cost-Effective Route for Global API Manufacturing
Industrial Synthesis of Chenodeoxycholic Acid: A Cost-Effective Route for Global API Manufacturing
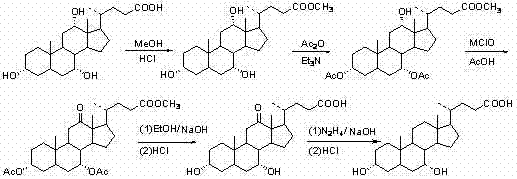
The pharmaceutical industry continuously seeks robust, scalable, and environmentally compliant pathways for producing critical bile acid intermediates. Patent CN102060902A introduces a transformative chemical synthesis method for Chenodeoxycholic Acid (CDCA), a pivotal active pharmaceutical ingredient used in treating cholesterol gallstones and as a key precursor for Ursodeoxycholic Acid (UDCA). Unlike traditional extraction methods that rely on the volatile supply of poultry or livestock bile, this patented approach utilizes Cholic Acid as a stable starting material, employing a streamlined five-step sequence that maximizes yield while minimizing environmental impact. For R&D directors and procurement strategists, this technology represents a significant leap forward in securing a reliable supply chain for high-purity bile acids, effectively decoupling production from agricultural constraints and offering a reproducible, industrial-grade solution for global markets.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of Chenodeoxycholic Acid has been plagued by significant inefficiencies inherent to both extraction and early synthetic methodologies. Traditional extraction from animal bile is not only operationally complex but also suffers from notoriously low yields and inconsistent purity profiles due to biological variability. Furthermore, earlier chemical synthesis routes often relied on harsh oxidizing agents such as chromium trioxide or Jones reagent to achieve the necessary structural modifications at the C-12 position. These heavy metal-based oxidants pose severe environmental hazards, generating toxic waste streams that require costly disposal protocols and complicate regulatory compliance. The reliance on such hazardous reagents increases the overall cost of goods sold (COGS) and introduces substantial supply chain risks related to the handling and storage of dangerous chemicals, making these conventional methods increasingly untenable for modern, sustainability-focused manufacturing facilities.
The Novel Approach
The methodology outlined in patent CN102060902A fundamentally reengineers the oxidation step, substituting toxic heavy metals with inexpensive and environmentally benign sodium hypochlorite. This innovative route begins with the esterification of Cholic Acid, followed by selective acetylation to protect the 3α and 7α hydroxyl groups, setting the stage for a highly selective oxidation at the 12α position. By utilizing a phase transfer catalytic system involving tetrabutylammonium bromide and bromide salts, the process achieves exceptional conversion rates without the need for cryogenic conditions or exotic reagents. The subsequent steps involve straightforward hydrolysis and a Wolff-Kishner reduction to finalize the deoxygenation, resulting in a final product with purity levels suitable for direct pharmaceutical application. This approach not only simplifies the operational workflow but also drastically reduces the ecological footprint of the manufacturing process.
Mechanistic Insights into Hypochlorite-Mediated Selective Oxidation
The core technical breakthrough of this synthesis lies in the third step: the selective oxidation of the 12α-hydroxyl group to a ketone using a hypochlorite-bromide system. In this mechanism, sodium hypochlorite acts as the terminal oxidant, while bromide ions serve as a mediator to generate the active brominating species in situ. The presence of tetrabutylammonium bromide (TBAB) functions as a phase transfer catalyst, facilitating the transport of the hypochlorite anion into the organic phase where the steroid substrate resides. This ensures that the oxidation occurs rapidly and selectively at the secondary alcohol position without affecting the protected acetate groups or the steroid backbone. The reaction conditions are mild, typically conducted at room temperature in a mixture of ethyl acetate and methanol, which prevents the formation of over-oxidized byproducts or chlorinated impurities that are common with free radical halogenation. This precise control over the oxidation state is critical for maintaining the stereochemical integrity of the bile acid scaffold.
Following the oxidation, the process employs a robust deprotection and reduction strategy to ensure high final purity. The hydrolysis step utilizes sodium hydroxide in ethanol to simultaneously cleave the methyl ester and the acetyl protecting groups, yielding the 12-oxo intermediate with high efficiency. The final transformation involves a Wolff-Kishner reduction, where the 12-keto group is converted to a methylene group using hydrazine hydrate and a strong base in ethylene glycol at elevated temperatures (up to 150°C). This high-temperature step is crucial for driving the equilibrium towards the fully deoxygenated product, effectively removing the oxygen functionality that distinguishes Cholic Acid from Chenodeoxycholic Acid. The rigorous control of pH during the workup, specifically acidifying to precipitate the product, ensures that residual basic impurities and hydrazine derivatives are effectively removed, resulting in a crystalline solid that meets stringent pharmaceutical specifications.
How to Synthesize Chenodeoxycholic Acid Efficiently
Implementing this synthesis requires careful attention to reaction stoichiometry and temperature control, particularly during the oxidation and reduction phases. The patent details a scalable protocol that begins with the conversion of Cholic Acid to its methyl ester using methanol and concentrated hydrochloric acid under reflux, achieving yields exceeding 95%. Subsequent acetylation is performed at room temperature using acetic anhydride and a catalytic amount of DMAP, ensuring complete protection of the A-ring hydroxyls. The critical oxidation step utilizes a molar excess of sodium hypochlorite relative to the substrate to drive the reaction to completion, followed by a simple aqueous workup. For the final reduction, the gradual removal of water and excess hydrazine via distillation prior to reaching the final reaction temperature of 150°C is essential to prevent side reactions and ensure safety. Detailed standardized operating procedures for each unit operation are provided below to guide process engineers in replicating these results.
- Esterification of Cholic Acid with methanol and HCl catalyst to form methyl cholate.
- Acetylation of hydroxyl groups at positions 3 and 7 using acetic anhydride and DMAP catalyst.
- Selective oxidation of the 12-alpha-hydroxyl group to a ketone using sodium hypochlorite and phase transfer catalysts.
- Hydrolysis of the ester and acetyl groups using sodium hydroxide to yield 12-oxochenodeoxycholic acid.
- Wolff-Kishner reduction using hydrazine hydrate and ethylene glycol at 150°C to remove the 12-keto group.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers compelling economic and logistical advantages over traditional extraction or heavy-metal synthesis. The primary driver for cost reduction is the substitution of expensive and regulated oxidizing agents with commodity chemicals like sodium hypochlorite (bleach) and acetic acid. This shift eliminates the need for specialized hazardous waste disposal contracts associated with chromium or manganese waste, leading to substantial operational savings. Furthermore, the raw materials required for this process, including Cholic Acid, methanol, and hydrazine, are widely available on the global chemical market, reducing the risk of supply disruptions. The simplicity of the workup procedures, which rely largely on filtration and recrystallization rather than complex chromatography, further enhances the throughput capacity of manufacturing plants, allowing for faster turnaround times and improved inventory management.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts and toxic heavy metal oxidants significantly lowers the direct material costs associated with production. By utilizing a phase transfer catalytic system with inexpensive quaternary ammonium salts, the process avoids the high expense of noble metals while maintaining high catalytic efficiency. Additionally, the high yields reported in the patent, particularly in the final reduction step, minimize the loss of valuable intermediate materials, ensuring that the maximum amount of starting Cholic Acid is converted into saleable product. This efficiency translates directly into a lower cost per kilogram of the final API, providing a competitive edge in price-sensitive markets.
- Enhanced Supply Chain Reliability: Relying on animal bile extraction subjects manufacturers to seasonal fluctuations and disease outbreaks in livestock populations, creating unpredictable supply volatility. In contrast, this chemical synthesis route depends on stable, petrochemical-derived reagents and bulk fermentation products that can be sourced from multiple suppliers globally. The robustness of the reaction conditions, which do not require sensitive inert atmospheres or ultra-low temperatures, means that production can be maintained consistently across different geographic locations. This diversification of the supply base ensures business continuity and allows for long-term contracting with greater confidence, shielding the organization from raw material price spikes.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing standard reactor types and separation equipment found in most fine chemical facilities. The use of aqueous hypochlorite generates salt and water as primary byproducts, which are far easier to treat in standard wastewater facilities compared to heavy metal sludge. This alignment with green chemistry principles simplifies the permitting process for new manufacturing lines and reduces the regulatory burden on environmental health and safety teams. The ability to scale from pilot batches to multi-ton commercial production without significant process re-engineering accelerates time-to-market for new drug formulations requiring CDCA.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity on yield expectations, impurity profiles, and scalability. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers reflect the specific advantages of the hypochlorite oxidation method and the Wolff-Kishner reduction strategy employed in this novel process.
Q: What is the primary advantage of using hypochlorite oxidation in CDCA synthesis?
A: The use of sodium hypochlorite replaces traditional chromium-based oxidants, significantly reducing heavy metal contamination and environmental waste disposal costs while maintaining high selectivity for the 12-position.
Q: How does this synthetic route compare to bile extraction methods?
A: Unlike extraction from animal bile which suffers from low yields and supply variability, this chemical synthesis offers consistent quality, scalable production volumes, and independence from agricultural supply chains.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process utilizes common industrial reagents like methanol, acetic anhydride, and bleach, avoiding exotic catalysts, which simplifies procurement and facilitates scaling from pilot plants to multi-ton manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chenodeoxycholic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering Chenodeoxycholic Acid with stringent purity specifications, utilizing rigorous QC labs to verify every batch against international pharmacopoeia standards. Our facility is equipped to handle the specific reagents and conditions required for this advanced synthesis, guaranteeing a consistent supply of material that meets the exacting demands of pharmaceutical R&D and commercial manufacturing.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project requirements. By leveraging our expertise in process optimization, we can provide a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating exactly how switching to this method can improve your margins. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on real-world performance metrics and our proven track record in bile acid chemistry.
