Advanced Manufacturing of SGLT-2 Inhibitor Intermediates via Selective Deprotection
Advanced Manufacturing of SGLT-2 Inhibitor Intermediates via Selective Deprotection
The pharmaceutical landscape for Type II diabetes treatment has been revolutionized by Sodium-Glucose Cotransporter 2 (SGLT-2) inhibitors, which offer a novel mechanism of action independent of insulin secretion. As demand for these therapeutics grows, the efficiency of their supply chain becomes paramount. Patent CN114195748A discloses a groundbreaking preparation method for key SGLT-2 inhibitor intermediates, specifically targeting compounds of Formula I' and its L-proline complex Formula I. This technology addresses critical bottlenecks in traditional synthesis by introducing a highly selective deprotection strategy and a unique derivatization-based purification protocol. Unlike prior art that relies on laborious chromatographic separation, this invention enables the production of high-purity intermediates suitable for direct API manufacturing through scalable crystallization techniques. For procurement leaders and R&D directors, this represents a significant opportunity to optimize the commercial scale-up of complex pharmaceutical intermediates while maintaining stringent quality standards.
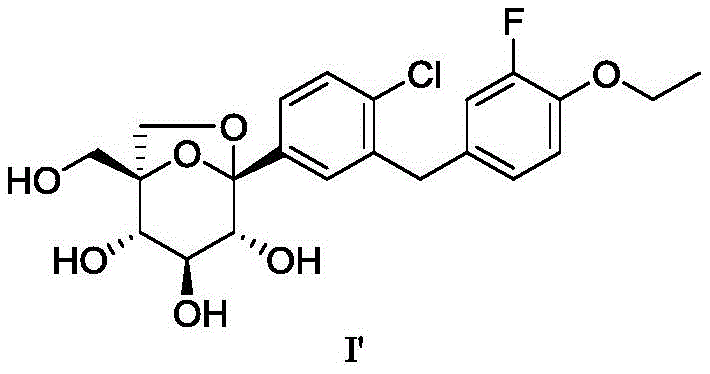
The core innovation lies in the precise manipulation of protecting groups on the tetrahydropyran ring. The patent details a route that circumvents the need for multiple orthogonal protection strategies that plague earlier generations of synthesis. By focusing on the selective removal of trimethylsilyl (TMS) groups under mild basic conditions, the process achieves high regioselectivity. Furthermore, the ability to derivatize the final intermediate into a p-nitrobenzoyl ester (Formula VI) allows for purification via recrystallization, a unit operation far more amenable to ton-scale production than flash chromatography. This technical advancement positions the technology as a cornerstone for any organization seeking a reliable SGLT-2 inhibitor intermediate supplier capable of delivering consistent quality at volume.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of glucoside-based SGLT-2 inhibitors, such as those disclosed in WO2012019496, has been fraught with inefficiencies that hinder cost-effective manufacturing. Traditional routes typically require the installation of four distinct protecting groups, often utilizing tert-butyldimethylsilyl (TBS) and benzyl (Bn) moieties, to mask the hydroxyl functionality of the sugar scaffold. The removal of these groups is particularly problematic; benzyl groups necessitate catalytic hydrogenation using expensive palladium reagents, which introduces heavy metal contamination risks and requires rigorous downstream clearance protocols. Moreover, the sequential deprotection steps often lack specificity, leading to complex mixtures that mandate purification via column chromatography. This reliance on chromatography is a major impediment to cost reduction in API manufacturing, as it is solvent-intensive, time-consuming, and notoriously difficult to scale beyond kilogram quantities without significant capital investment in simulated moving bed (SMB) systems.
The Novel Approach
In stark contrast, the methodology presented in CN114195748A streamlines the synthetic trajectory by leveraging the differential stability of silyl ethers. The process utilizes a fully TMS-protected intermediate (Formula II) which undergoes selective mono-deprotection to yield Formula III. This step is critical as it exposes a single hydroxyl group necessary for subsequent transformations while leaving others protected to prevent side reactions. The true brilliance of this approach, however, is the purification strategy. Instead of struggling to purify the polar, free-hydroxyl containing intermediate directly, the process converts the crude material into a lipophilic p-nitrobenzoyl derivative (Formula VI). This derivative exhibits superior crystallinity, allowing impurities to be washed away through simple recrystallization or beating in mixed solvent systems like acetonitrile and isopropanol. This shift from chromatography to crystallization drastically simplifies the workflow, reduces solvent waste, and enhances the overall throughput of the production line.
Mechanistic Insights into Selective TMS Deprotection and Derivatization
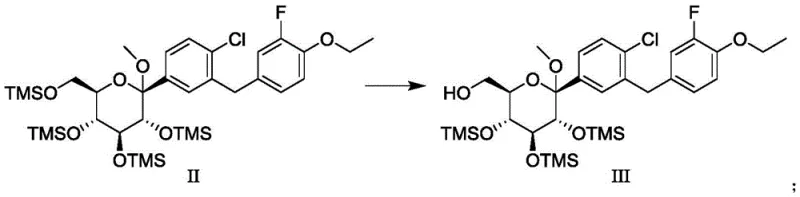
The chemical elegance of this synthesis is rooted in the controlled hydrolysis of silyl ethers. In the conversion of Formula II to Formula III, the reaction conditions are meticulously tuned to exploit the subtle steric and electronic differences between the various TMS-protected positions on the tetrahydropyran ring. The use of sodium carbonate in methanol at low temperatures (e.g., -5°C) provides a kinetic window where the most accessible or least hindered TMS group is cleaved preferentially. Comparative data within the patent highlights the sensitivity of this step; for instance, using potassium carbonate under similar conditions resulted in over-deprotection, yielding the fully deprotected compound 'a' alongside the desired product. This underscores the importance of base selection and stoichiometry in maintaining reaction specificity. The resulting intermediate, Formula III, retains three TMS groups which serve as robust handles for the subsequent oxidation and cyclization steps, ensuring that the stereochemical integrity of the sugar backbone is preserved throughout the sequence.
Following the construction of the C-glycosidic bond and subsequent cyclization to form the hemiacetal (Formula V), the process employs a "purification-by-derivatization" tactic. The crude Formula I' is reacted with p-nitrobenzoyl chloride (PNB-Cl) in the presence of a base like triethylamine. This transformation converts the polar, amorphous sugar derivative into a crystalline ester (Formula VI). The p-nitrobenzoyl group not only aids in crystallization but also serves as a temporary mask that stabilizes the molecule against degradation during purification. Once the high-purity ester is isolated (often achieving >98% purity after recrystallization), it is hydrolyzed back to the free hydroxyl form using lithium hydroxide. This two-step "mask-purify-unmask" cycle is a powerful tool in process chemistry, effectively decoupling the synthesis from the limitations of purifying highly polar carbohydrates directly. It ensures that the final high-purity SGLT-2 inhibitor intermediate meets the rigorous impurity profiles required for regulatory submission.
How to Synthesize SGLT-2 Inhibitor Intermediate Efficiently
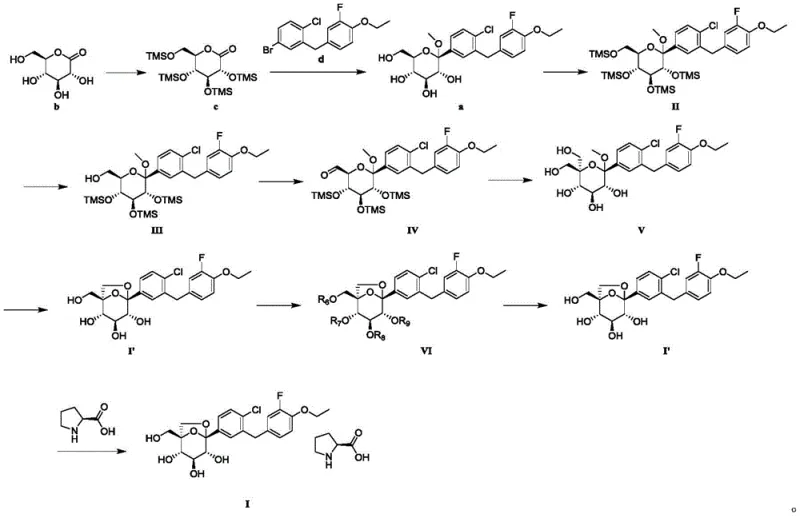
The implementation of this synthesis route requires careful attention to reaction parameters, particularly during the selective deprotection and oxidation phases. The patent outlines a robust sequence starting from a protected gluconolactone derivative, proceeding through Grignard addition to install the aglycone, followed by the critical selective deprotection and oxidation-cyclization cascade. The final stages involve the strategic derivatization for purification. While the chemical transformations are straightforward, the operational success relies on precise control of temperature, stoichiometry, and solvent composition to maximize yield and minimize the formation of regioisomers. For process chemists looking to adopt this technology, understanding the nuances of the workup procedures—such as the specific aqueous washes required to remove excess silylating agents or the exact solvent ratios for recrystallization—is essential for reproducibility. Detailed standardized synthesis steps see the guide below.
- Perform selective deprotection of the fully protected tetrahydropyran intermediate (Formula II) using sodium carbonate in methanol to yield the mono-deprotected species (Formula III).
- Oxidize the primary alcohol of Formula III to an aldehyde (Formula IV) using sulfur trioxide-pyridine complex, followed by cyclization to form the hemiacetal structure (Formula V).
- Convert the crude product (Formula I') into a crystalline derivative (Formula VI) using p-nitrobenzoyl chloride for purification via recrystallization, then hydrolyze back to the final high-purity API intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers tangible benefits that extend beyond mere chemical yield. The elimination of column chromatography is perhaps the most significant economic driver. Chromatographic purification is a major cost center in fine chemical manufacturing, consuming vast quantities of silica gel and organic solvents while limiting batch sizes. By replacing this with crystallization, the process significantly reduces raw material costs and waste disposal fees. Furthermore, the removal of palladium-catalyzed hydrogenation steps eliminates the need for expensive noble metal catalysts and the associated analytical testing for residual metals, thereby streamlining the quality control workflow. These factors combine to create a manufacturing process that is inherently more cost-efficient and environmentally sustainable, aligning with modern green chemistry principles.
- Cost Reduction in Manufacturing: The streamlined synthetic route minimizes the number of unit operations and avoids the use of costly reagents such as palladium on carbon. By utilizing inexpensive inorganic bases like sodium carbonate for deprotection and avoiding chromatographic purification, the overall cost of goods sold (COGS) is substantially lowered. The ability to purify via recrystallization also reduces solvent consumption, which is a major variable cost in large-scale production. This efficiency translates directly into better pricing stability for downstream API manufacturers.
- Enhanced Supply Chain Reliability: The reagents required for this synthesis, including TMS-Cl, sodium carbonate, and p-nitrobenzoyl chloride, are commodity chemicals with robust global supply chains. Unlike specialized chiral catalysts or sensitive organometallic reagents that may face supply disruptions, these materials are readily available from multiple vendors. This diversity in sourcing mitigates the risk of production delays due to raw material shortages, ensuring a consistent and reliable supply of the critical SGLT-2 inhibitor intermediate to meet market demand.
- Scalability and Environmental Compliance: The process is designed with scale-up in mind. Crystallization is a standard unit operation in the pharmaceutical industry that scales linearly from the laboratory to the plant floor without the engineering challenges associated with chromatography. Additionally, the reduction in solvent usage and the avoidance of heavy metal catalysts simplify waste treatment and environmental compliance. This makes the technology highly attractive for manufacturing in regions with strict environmental regulations, facilitating smoother regulatory approvals and long-term operational sustainability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific experimental data and beneficial effects outlined in the patent documentation, providing clarity on purity profiles, scalability, and process robustness. Understanding these aspects is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: How does this novel process improve purity compared to conventional methods?
A: Conventional methods often rely on column chromatography which is difficult to scale. This patent introduces a derivatization strategy where the crude product is converted into a p-nitrobenzoyl ester (Formula VI). This derivative can be purified via simple recrystallization or beating, removing impurities effectively before hydrolyzing back to the final product, achieving purities exceeding 99%.
Q: What are the key cost-saving drivers in this synthesis route?
A: The primary cost reductions come from eliminating expensive palladium-catalyzed hydrogenation steps required for benzyl group removal in older routes. Additionally, the use of inexpensive inorganic bases like sodium carbonate for selective deprotection and the avoidance of large-scale column chromatography significantly lower operational expenditures and solvent consumption.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is specifically designed for scalability. By replacing chromatographic purification with crystallization-based techniques and utilizing robust, non-sensitive reagents (such as TMS-Cl and Na2CO3), the method avoids the bottlenecks typically associated with scaling up complex carbohydrate chemistry, ensuring consistent supply continuity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable SGLT-2 Inhibitor Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes in the competitive landscape of diabetes therapeutics. Our team of expert process chemists has extensively evaluated the technology disclosed in CN114195748A and possesses the technical capability to implement this advanced selective deprotection and derivatization strategy. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless. Our facilities are equipped with state-of-the-art reactors and purification units capable of handling the specific solvent systems and temperature controls required for this synthesis, guaranteeing adherence to stringent purity specifications and rigorous QC labs protocols.
We invite pharmaceutical partners to collaborate with us to leverage this innovative manufacturing approach. By optimizing the production of SGLT-2 inhibitor intermediates, we can help you achieve significant operational efficiencies and secure a stable supply of high-quality materials. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our implementation of this patent can enhance your supply chain resilience and reduce overall manufacturing costs.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
