Advanced Green Synthesis of Menadione Sodium Bisulfite for Industrial Scale-Up and Cost Reduction
Advanced Green Synthesis of Menadione Sodium Bisulfite for Industrial Scale-Up and Cost Reduction
The global demand for high-purity vitamin derivatives in the animal nutrition sector continues to surge, driving the need for robust and sustainable manufacturing processes. Patent CN113185433A introduces a groundbreaking preparation method for Menadione Sodium Bisulfite, a critical feed additive and pharmaceutical intermediate known chemically as 2-methyl-1,4-naphthoquinone sodium bisulfite. This innovative protocol addresses long-standing industry pain points by replacing hazardous heavy metal oxidants with a benign air oxidation system, fundamentally altering the economic and environmental landscape of production. The structural integrity and chemical identity of the target molecule are paramount for its biological efficacy in preventing hemorrhagic diseases in livestock, as illustrated by its precise molecular architecture below.
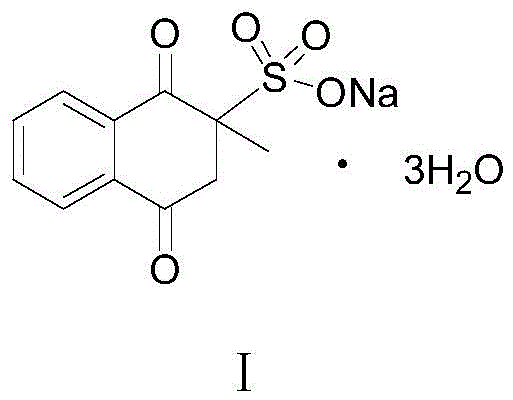
This technical disclosure provides a comprehensive roadmap for synthesizing this essential compound starting from basic petrochemical feedstocks. By leveraging a strategic sequence of Friedel-Crafts acylation, halogenation-elimination, and catalytic oxidation, the patent establishes a pathway that achieves a total yield of 88.8 percent while maintaining exceptional product stability. For R&D directors and process engineers, this represents a significant opportunity to upgrade existing facilities with a greener, more cost-effective technology that aligns with modern regulatory standards for chemical manufacturing and environmental protection.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Menadione Sodium Bisulfite has relied heavily on the oxidation of 2-methylnaphthalene, a process fraught with significant technical and environmental drawbacks. As depicted in the traditional synthesis scheme, earlier methods frequently employed chromium(VI) based oxidants, such as chromic anhydride, to convert the methyl-naphthalene precursor into the requisite quinone structure. This reliance on hexavalent chromium generates massive quantities of toxic acidic wastewater containing heavy metals, imposing severe burdens on waste treatment infrastructure and escalating operational compliance costs. Furthermore, the raw material 2-methylnaphthalene is often subject to price volatility and supply chain constraints, while alternative oxidants like m-chloroperoxybenzoic acid introduce prohibitive raw material costs that render the process economically unviable for large-scale commodity production.
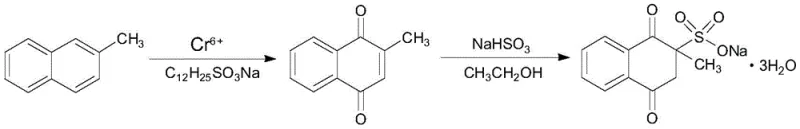
Beyond the environmental liabilities, the conventional oxidative pathways suffer from poor reaction selectivity and moderate yields, typically capping around 75.7 percent. The harsh conditions required for chromium oxidation often lead to over-oxidation and the formation of complex by-product mixtures, necessitating energy-intensive purification steps to meet the stringent purity specifications required for feed-grade and pharmaceutical applications. These inefficiencies not only erode profit margins but also create bottlenecks in production capacity, making it difficult for manufacturers to respond agilely to market demand fluctuations. Consequently, there is an urgent industry-wide imperative to transition away from these legacy technologies toward more sustainable and economically resilient synthetic strategies.
The Novel Approach
The patented methodology presented in CN113185433A offers a transformative solution by completely reimagining the synthetic backbone of Menadione Sodium Bisulfite production. Instead of relying on expensive and scarce naphthalene derivatives, this novel approach initiates the synthesis with benzene and alpha-methyl-gamma-butyrolactone, two abundant and low-cost commodity chemicals. The core innovation lies in the construction of the naphthalene ring system via a Friedel-Crafts reaction, followed by a highly selective halogenation-elimination sequence to install the hydroxyl functionality. This strategic shift not only drastically reduces raw material expenditures but also bypasses the generation of hazardous chromium waste entirely, positioning the process as a leader in green chemistry principles.
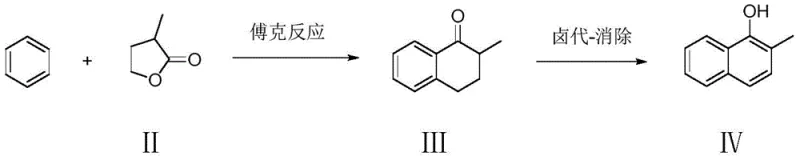
Furthermore, the new route demonstrates superior control over reaction specificity, utilizing mild air oxidation to convert the intermediate naphthol into the final quinone precursor. This step is critical as it avoids the use of stoichiometric chemical oxidants, thereby minimizing waste generation and simplifying the downstream workup procedures. The overall process is characterized by high stability of intermediates and robust reaction conditions that are easily scalable from laboratory to multi-ton production. By integrating these advancements, manufacturers can achieve a total yield of 88.8 percent with product purity levels exceeding 99 percent, ensuring a reliable supply of high-quality Menadione Sodium Bisulfite for the global market.
Mechanistic Insights into Friedel-Crafts Acylation and Air Oxidation
The cornerstone of this advanced synthesis is the initial Friedel-Crafts acylation between benzene and alpha-methyl-gamma-butyrolactone, catalyzed by Lewis acids such as aluminum trichloride or titanium tetrachloride. This reaction proceeds through the formation of an acylium ion intermediate which attacks the electron-rich benzene ring, followed by intramolecular cyclization to form the tetralone derivative, 2-methyl-3,4-dihydro-1(2H)-naphthalenone. The choice of catalyst and the precise control of reaction temperature between 60 to 90 degrees Celsius are vital to suppress polyacylation side reactions and ensure high conversion rates. The subsequent halogenation step targets the alpha-position relative to the carbonyl group with high regioselectivity, utilizing reagents like chlorine or N-bromosuccinimide, followed by a base-mediated elimination that aromatizes the ring to yield 2-methyl-1-naphthol.
Following the formation of the naphthol intermediate, the process employs a catalytic air oxidation mechanism to generate 2-methyl-1,4-naphthoquinone. Unlike chemical oxidants that leave behind reduced by-products, molecular oxygen from air serves as the terminal oxidant, producing water as the only benign by-product. This transformation is facilitated by heating the naphthol in solvents like toluene or isopropanol under an oxygen atmosphere, where the phenolic hydroxyl group is oxidized to the quinone system. The final step involves a nucleophilic addition of sodium bisulfite to the quinone double bond, forming the stable sulfonate adduct. This sequence ensures minimal impurity carryover, as each step is designed to maximize selectivity and facilitate easy purification through crystallization or distillation.
How to Synthesize Menadione Sodium Bisulfite Efficiently
The implementation of this synthesis route requires careful attention to reaction parameters to maximize yield and safety. The process begins with the acylation step, followed by the functionalization of the ring system and final oxidation. Detailed operational guidelines regarding solvent ratios, catalyst loading, and temperature profiles are critical for successful scale-up.
- Perform a Friedel-Crafts reaction between alpha-methyl-gamma-butyrolactone and benzene using a Lewis acid catalyst to form 2-methyl-3,4-dihydro-1(2H)-naphthalenone.
- Subject the naphthalenone intermediate to halogenation followed by base-mediated elimination to generate 2-methyl-1-naphthol.
- Oxidize 2-methyl-1-naphthol using air oxygen in a solvent system to obtain 2-methyl-1,4-naphthoquinone.
- React the quinone with sodium bisulfite solution to finalize the production of Menadione Sodium Bisulfite.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented synthesis route offers compelling economic advantages driven by raw material optimization and waste reduction. The substitution of expensive 2-methylnaphthalene with commodity benzene and lactone significantly lowers the direct material cost per kilogram of finished product. Additionally, the elimination of chromium-based reagents removes the need for costly heavy metal removal units and hazardous waste disposal services, leading to substantial operational expenditure savings. The simplified workflow, characterized by fewer purification steps and higher overall yields, enhances throughput capacity and reduces the manufacturing cycle time, allowing for more responsive inventory management.
- Cost Reduction in Manufacturing: The utilization of widely available petrochemical feedstocks such as benzene creates a more stable and predictable cost structure compared to specialized aromatic intermediates. By avoiding the use of premium-priced oxidants like m-chloroperoxybenzoic acid and eliminating the expense associated with treating toxic chromium effluent, the overall production cost is drastically reduced. This economic efficiency allows manufacturers to maintain competitive pricing even in volatile raw material markets, securing better margins for downstream applications in the feed and pharmaceutical sectors.
- Enhanced Supply Chain Reliability: Sourcing strategies are greatly improved as the primary inputs are bulk chemicals with established global supply chains, reducing the risk of shortages that often plague specialty reagents. The robustness of the reaction conditions means that production is less susceptible to disruptions caused by sensitive reagent handling or strict storage requirements. This reliability ensures consistent delivery schedules for customers, strengthening long-term supplier relationships and mitigating the risks associated with supply chain bottlenecks common in the fine chemical industry.
- Scalability and Environmental Compliance: The green nature of the process, particularly the use of air oxidation and the absence of heavy metals, simplifies regulatory compliance and environmental permitting for new or expanded production facilities. The reduced volume of process wastewater and the non-toxic nature of the by-products make it easier to scale operations from pilot plants to commercial multi-ton reactors without incurring prohibitive environmental mitigation costs. This scalability supports business growth and aligns with corporate sustainability goals, making the supply chain more resilient to future regulatory changes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Menadione Sodium Bisulfite using this advanced methodology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on process capabilities and product quality.
Q: Why is the new synthesis route for Menadione Sodium Bisulfite considered more environmentally friendly?
A: The novel route eliminates the use of toxic chromium(VI) oxidants found in traditional methods, thereby preventing the generation of hazardous heavy metal wastewater and simplifying waste treatment protocols.
Q: What are the primary raw materials used in this patented preparation method?
A: The process utilizes inexpensive and readily available commodity chemicals, specifically benzene and alpha-methyl-gamma-butyrolactone, avoiding the high cost and supply volatility of 2-methylnaphthalene.
Q: How does this method improve product purity compared to conventional techniques?
A: By employing highly selective Friedel-Crafts acylation and controlled air oxidation steps, the method minimizes side reactions, resulting in final product purity exceeding 99% with significantly reduced impurity profiles.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Menadione Sodium Bisulfite Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting next-generation synthesis technologies to meet the evolving demands of the global feed and pharmaceutical industries. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of Menadione Sodium Bisulfite performs reliably in your formulations.
We invite you to collaborate with us to leverage this cutting-edge preparation method for your supply chain needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced manufacturing capabilities can drive value and efficiency for your organization.
