Advanced Green Manufacturing of Menadione Sodium Bisulfite for Global Feed Additive Supply Chains
Advanced Green Manufacturing of Menadione Sodium Bisulfite for Global Feed Additive Supply Chains
The global demand for high-purity vitamin intermediates is intensifying, driven by the expanding livestock industry and stringent regulatory requirements for feed safety. A pivotal breakthrough in this sector is detailed in Chinese patent CN113185431B, which discloses a novel, environmentally benign preparation method for Menadione Sodium Bisulfite, widely known as Vitamin K3. This compound, chemically defined as 2-methyl-1,4-naphthoquinone sodium bisulfite, serves as an indispensable nutrient element for animal health, playing a critical role in blood coagulation and bone mineralization. The structural integrity of this molecule is paramount for its biological efficacy, as illustrated in the chemical diagram below.
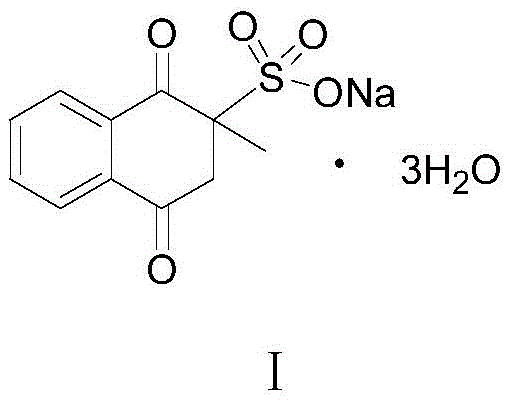
Historically, the industrial synthesis of this vital intermediate has been plagued by severe environmental and economic inefficiencies. Conventional routes often rely on hazardous oxidants like chromic anhydride, generating toxic waste streams that complicate disposal and inflate operational expenditures. The methodology presented in patent CN113185431B represents a paradigm shift, replacing these archaic, pollution-heavy steps with a green catalytic sequence. By leveraging cheap and easily obtained 3,4-dihydro-1(2H)-naphthalenone as the primary feedstock, the new process achieves a total yield of approximately 88.9 percent while virtually eliminating heavy metal contamination. For R&D directors and procurement strategists, this transition offers a compelling value proposition: a robust, scalable, and compliant supply chain for one of the agriculture sector's most critical additives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional manufacturing pathways for Menadione Sodium Bisulfite have long been burdened by intrinsic chemical and logistical flaws that hinder sustainable production. As depicted in prior art such as Scheme 1, the oxidation of 2-methylnaphthalene typically necessitates a chromic anhydride system. This approach is fundamentally flawed due to the generation of substantial volumes of chromium-containing acidic wastewater, posing severe environmental hazards and requiring costly remediation infrastructure. Furthermore, the starting material, 2-methylnaphthalene, is often subject to price volatility and supply chain bottlenecks, creating uncertainty for long-term procurement planning.
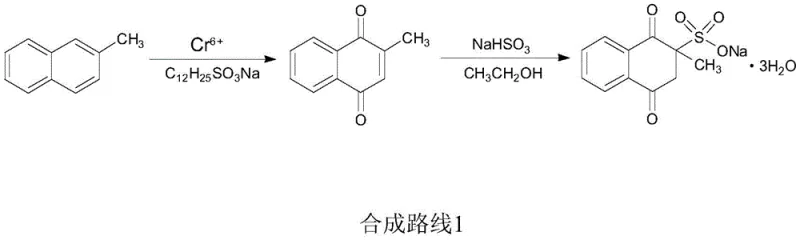
Alternative methods, such as those utilizing m-chloroperoxybenzoic acid (m-CPBA) shown in Scheme 2, attempt to bypass heavy metals but introduce different economic barriers. These organic peroxide-based routes suffer from prohibitively high reagent costs and notoriously low yields, often hovering around 31 percent for the quinone intermediate. Such inefficiencies result in poor atom economy and excessive solvent consumption, rendering these methods commercially unviable for large-scale industrial application. The combination of low selectivity, complex purification requirements, and high raw material costs creates a fragile supply chain that struggles to meet the growing global demand for feed-grade vitamins.
The Novel Approach
In stark contrast, the innovative route disclosed in CN113185431B dismantles these barriers through a cleverly designed four-step sequence that prioritizes both economic efficiency and environmental stewardship. The process initiates with the halogenation and subsequent elimination of 3,4-dihydro-1(2H)-naphthalenone, a readily available ketone derivative, to form 1-naphthol with exceptional specificity. This is followed by a catalytic methylation using methanol and an iron oxide catalyst, avoiding expensive alkylating agents. The crown jewel of this synthesis is the utilization of molecular oxygen (air) for the oxidation of 2-methyl-1-naphthol to the quinone, completely eradicating the need for stoichiometric chemical oxidants.
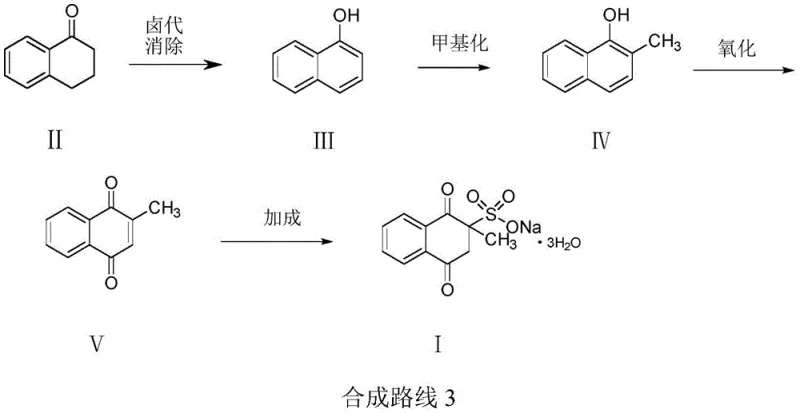
This strategic redesign not only simplifies the operational workflow but also dramatically enhances the purity profile of the final product. By avoiding harsh oxidizing conditions that promote polymerization and side reactions, the new method ensures a cleaner reaction matrix. The final addition of sodium bisulfite proceeds under mild conditions, preserving the integrity of the quinone ring. For supply chain leaders, this translates to a manufacturing process that is safer, easier to control, and capable of delivering consistent quality without the baggage of hazardous waste management, thereby securing a more reliable source of high-purity pharmaceutical intermediates.
Mechanistic Insights into Halogenation-Elimination and Air Oxidation
The success of this green synthesis lies in the precise control of reaction mechanisms at each stage, particularly the initial activation of the naphthalene ring system. The process begins with the halogenation of 3,4-dihydro-1(2H)-naphthalenone, where the carbonyl group activates the ortho-position for electrophilic attack. By carefully selecting halogenating agents such as chlorine gas or hydrochloric acid-hydrogen peroxide systems, the reaction achieves high regioselectivity, preventing unwanted substitution on the aromatic benzene ring. The subsequent elimination step, facilitated by a base like sodium hydroxide, efficiently aromatizes the ring to yield 1-naphthol. This tandem halogenation-elimination strategy is superior to direct hydroxylation methods, as it avoids the formation of complex isomeric mixtures that are difficult to separate.
Following methylation, the oxidation of 2-methyl-1-naphthol to 2-methyl-1,4-naphthoquinone represents a masterclass in green chemistry engineering. Unlike traditional methods that rely on electron transfer from toxic metal centers, this protocol utilizes atmospheric oxygen as the terminal oxidant. The reaction is conducted in solvents like toluene or isopropanol at moderate temperatures, where the phenolic substrate undergoes dehydrogenation. The use of activated carbon for decolorization and recrystallization further refines the product, removing trace polymeric by-products that often plague quinone syntheses. This mechanistic pathway ensures that the final Menadione Sodium Bisulfite meets rigorous purity specifications, essential for its application in sensitive animal feed formulations where contaminant levels are strictly regulated.
How to Synthesize Menadione Sodium Bisulfite Efficiently
The implementation of this novel synthetic route requires careful attention to reaction parameters to maximize yield and minimize impurity formation. The process is divided into distinct operational units: the conversion of the ketone to naphthol, the methylation to the naphthol derivative, the aerobic oxidation to the quinone, and the final bisulfite addition. Each step has been optimized in the patent examples to demonstrate scalability, with specific attention paid to temperature control and reagent stoichiometry. For technical teams looking to adopt this methodology, the following guide outlines the standardized procedural framework derived from the patent data.
- Perform halogenation and elimination on 3,4-dihydro-1(2H)-naphthalenone to generate 1-naphthol.
- Conduct catalytic methylation of 1-naphthol with methanol using iron oxide to form 2-methyl-1-naphthol.
- Oxidize 2-methyl-1-naphthol using air to obtain 2-methyl-1,4-naphthoquinone.
- React 2-methyl-1,4-naphthoquinone with sodium bisulfite to finalize Menadione Sodium Bisulfite.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain executives, the adoption of this green synthesis route offers transformative advantages that extend far beyond simple regulatory compliance. The shift away from chromium-based oxidation eliminates the need for expensive wastewater treatment facilities dedicated to heavy metal removal, resulting in substantial operational cost savings. Furthermore, the reliance on air as an oxidant removes the recurring cost of purchasing stoichiometric chemical oxidants, which are often priced at a premium due to their specialized nature. This fundamental change in the cost structure allows for a more competitive pricing model for the final Vitamin K3 product, enhancing margin potential for downstream feed manufacturers.
- Cost Reduction in Manufacturing: The replacement of expensive starting materials like 2-methylnaphthalene with cheap 3,4-dihydro-1(2H)-naphthalenone significantly lowers the raw material bill of materials. Additionally, the high selectivity of the halogenation-elimination step reduces the loss of valuable intermediates to side products, improving overall mass balance. The elimination of chromium waste treatment costs further contributes to a leaner manufacturing expense profile, making the process economically robust against fluctuating raw material markets.
- Enhanced Supply Chain Reliability: By utilizing bulk commodity chemicals as feedstocks, the process mitigates the risk of supply disruptions associated with specialty reagents. The simplified reaction conditions, which do not require extreme pressures or cryogenic temperatures, allow for production in a wider range of standard chemical manufacturing facilities. This flexibility ensures a continuous and stable supply of Menadione Sodium Bisulfite, crucial for maintaining uninterrupted livestock feed production schedules globally.
- Scalability and Environmental Compliance: The green nature of this process aligns perfectly with increasingly stringent global environmental regulations. The absence of heavy metal effluents simplifies permitting and reduces the risk of regulatory shutdowns. The process is inherently scalable, as demonstrated by the straightforward workup procedures involving simple filtration and crystallization. This ease of scale-up facilitates rapid capacity expansion to meet surging market demand without the need for complex new infrastructure investments.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this green synthesis technology. These insights are derived directly from the comparative data and experimental results presented in the patent documentation, providing clarity on the practical benefits of switching to this novel manufacturing route.
Q: Why is the new air oxidation method superior to traditional chromium oxidation?
A: Traditional methods utilize toxic chromic anhydride, generating massive amounts of hazardous chromate wastewater that requires expensive treatment. The new method employs air oxidation, eliminating heavy metal pollution entirely and drastically reducing environmental compliance costs while improving reaction selectivity.
Q: What are the cost advantages of using 3,4-dihydro-1(2H)-naphthalenone as a starting material?
A: Unlike 2-methylnaphthalene, which can be costly and supply-constrained, 3,4-dihydro-1(2H)-naphthalenone is a cheap and readily available bulk chemical. This substitution significantly lowers the raw material input cost for the entire synthesis chain.
Q: How does this process ensure high purity for feed-grade applications?
A: The process features high regioselectivity during the halogenation-elimination step and utilizes mild reaction conditions for the final addition. This minimizes side reactions and polymeric by-products, resulting in a final product with exceptional purity suitable for sensitive livestock feed formulations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Menadione Sodium Bisulfite Supplier
The technological advancements detailed in patent CN113185431B underscore the immense potential for optimizing the production of critical vitamin intermediates. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate these laboratory innovations into commercial reality. As a premier CDMO partner, we bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of Menadione Sodium Bisulfite meets the highest international standards for feed and pharmaceutical applications.
We invite forward-thinking partners to collaborate with us to leverage this green synthesis technology for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and comprehensive route feasibility assessments, ensuring that your transition to this superior manufacturing method is seamless, compliant, and economically advantageous.
