Industrial Scale-Up of 4-Fluoro-2-Methylbenzoic Acid via Novel Friedel-Crafts Acylation
The chemical landscape for producing specialized aromatic acids is constantly evolving, driven by the dual demands of higher purity for electronic applications and lower costs for pharmaceutical intermediates. A pivotal development in this sector is documented in patent CN110903176A, published on March 24, 2020, which outlines a robust synthetic methodology for 4-fluoro-2-methylbenzoic acid. This compound serves as a critical building block, notably functioning as a key raw material for electronic chemical photoinitiators and holding significant potential as a medicinal intermediate in various drug synthesis pathways. The patent introduces a strategic shift away from hazardous and energy-intensive protocols, proposing a streamlined approach that leverages classical Friedel-Crafts chemistry adapted for modern industrial efficiency. By utilizing m-fluorotoluene and trichloroacetyl chloride as foundational feedstocks, the inventors have established a pathway that circumvents the severe operational constraints of previous generations of synthesis, thereby offering a compelling value proposition for global supply chains seeking reliability and cost optimization in the production of fluorinated benzoic acid derivatives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-fluoro-2-methylbenzoic acid has been plagued by significant technical and economic hurdles that hinder efficient commercial scale-up. Traditional routes often rely on the direct hydrolysis of 4-fluoro-2-methylbenzonitrile under strongly basic conditions; while conceptually simple, the precursor nitrile itself involves a complex and costly synthetic sequence that inflates the overall production expense. Alternatively, organometallic approaches utilizing 2-bromo-5-fluorotoluene have been employed, necessitating Grignard reactions or lithiation with butyllithium. These methods impose draconian operational requirements, specifically the need for strictly anhydrous and anaerobic environments coupled with cryogenic cooling down to minus 78 degrees Celsius to manage the reactivity of the organolithium species. Such extreme conditions not only demand specialized, expensive reactor infrastructure but also introduce substantial safety risks associated with handling pyrophoric reagents on a large tonnage scale. Furthermore, these legacy processes often suffer from poor regioselectivity, generating difficult-to-separate isomer mixtures that compromise the final yield and purity, rendering them suboptimal for the high-specification demands of the electronic and pharmaceutical industries.
The Novel Approach
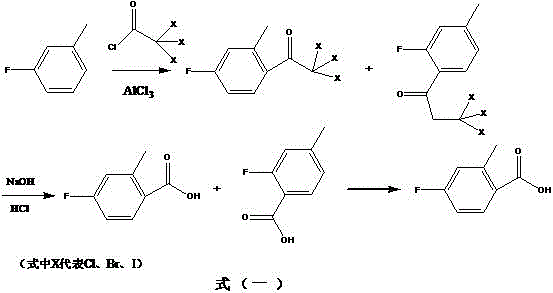
In stark contrast to these cumbersome legacy techniques, the methodology disclosed in the patent presents a transformative solution by employing a Friedel-Crafts acylation strategy that operates under remarkably mild conditions. By reacting m-fluorotoluene with trichloroacetyl chloride in the presence of a Lewis acid catalyst such as anhydrous aluminum chloride, the process achieves the desired carbon-carbon bond formation at temperatures ranging from negative 20 to 50 degrees Celsius, with a preferred operational window of 0 to 10 degrees Celsius. This elimination of cryogenic requirements represents a paradigm shift in process safety and energy efficiency, allowing for the use of standard glass-lined or stainless steel reactors without the need for complex low-temperature cooling loops. The subsequent hydrolysis step utilizes common alkaline solutions like sodium hydroxide to cleave the trichloromethyl ketone intermediate directly into the carboxylic acid, bypassing the need for hazardous dry ice quenching or high-pressure carbonation steps. This streamlined two-stage reaction sequence, followed by a straightforward recrystallization, drastically simplifies the workflow, reduces the number of unit operations, and minimizes the generation of hazardous waste, thereby aligning perfectly with green chemistry principles and modern manufacturing sustainability goals.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The core of this innovative synthesis lies in the precise control of electrophilic aromatic substitution during the initial acylation phase. When m-fluorotoluene is subjected to reaction with trichloroacetyl chloride under the catalytic influence of anhydrous aluminum chloride, the Lewis acid activates the acid chloride to form a highly reactive acylium ion complex. This electrophile selectively attacks the aromatic ring of the m-fluorotoluene substrate. Due to the directing effects of the existing methyl and fluoro substituents, the reaction favors the formation of the para-substituted ketone relative to the methyl group, which corresponds to the ortho-position relative to the fluoro group, yielding 2,2,2-trichloro-1-(4-fluoro-2-methylphenyl)ethanone as the major isomer. The choice of solvent, preferably 1,2-dichloroethane, plays a crucial role in stabilizing the ionic intermediates and ensuring homogeneous reaction conditions, which is vital for maintaining consistent selectivity and preventing the formation of poly-acylated byproducts that could complicate downstream purification efforts.
Following the acylation, the transformation of the trichloromethyl ketone into the target carboxylic acid proceeds via a mechanism analogous to the haloform reaction but adapted for synthetic utility. Under alkaline conditions, typically using aqueous sodium hydroxide, the electron-withdrawing trichloromethyl group renders the adjacent carbonyl carbon susceptible to nucleophilic attack by hydroxide ions. This leads to the formation of a tetrahedral intermediate which subsequently collapses, expelling a trichloromethanide anion (which rapidly protonates to form chloroform) and generating the carboxylate salt of the desired acid. Upon acidification with hydrochloric acid, the free 4-fluoro-2-methylbenzoic acid precipitates or separates into the organic phase. This mechanistic pathway is particularly advantageous because it inherently incorporates a purification step; the volatile chloroform byproduct is easily removed, and the differing solubility properties of the ortho and para isomers allow for effective separation during the final recrystallization from toluene, ensuring the isolation of the target isomer with high fidelity.
How to Synthesize 4-Fluoro-2-Methylbenzoic Acid Efficiently
Implementing this synthesis route requires careful attention to stoichiometry and temperature control to maximize the yield of the desired isomer while minimizing side reactions. The process begins with the preparation of the reaction vessel under an inert nitrogen atmosphere to prevent moisture interference with the Lewis acid catalyst. Operators must ensure that the addition of trichloroacetyl chloride is performed dropwise to manage the exotherm effectively, maintaining the internal temperature within the narrow optimal range of 0 to 10 degrees Celsius as specified in the patent examples. Following the acylation, the reaction mixture is quenched into cold dilute hydrochloric acid to decompose the aluminum complexes, followed by phase separation to isolate the organic layer containing the ketone intermediate. For detailed standardized operating procedures regarding exact mixing times, washing protocols, and crystallization parameters, please refer to the structured guide below.
- Perform Friedel-Crafts acylation of m-fluorotoluene with trichloroacetyl chloride using anhydrous aluminum chloride catalyst at 0-10°C.
- Conduct alkaline hydrolysis of the resulting ketone intermediate using sodium hydroxide solution followed by acidification.
- Purify the crude acid product through recrystallization using toluene solvent to achieve high purity specifications.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers profound strategic benefits that extend far beyond simple chemical conversion. The primary advantage lies in the drastic simplification of the manufacturing infrastructure required. By eliminating the need for cryogenic cooling systems and the handling of pyrophoric organolithium reagents, facilities can significantly reduce their capital expenditure on specialized equipment and lower their ongoing operational costs related to energy consumption and safety compliance. The reliance on commodity chemicals like m-fluorotoluene and trichloroacetyl chloride ensures a stable and resilient supply chain, as these feedstocks are produced in massive volumes globally, mitigating the risk of raw material shortages that often plague niche organometallic precursors. Furthermore, the mild reaction conditions translate to shorter batch cycle times and higher throughput, enabling manufacturers to respond more agilely to fluctuating market demands without compromising on product quality or delivery schedules.
- Cost Reduction in Manufacturing: The economic impact of switching to this Friedel-Crafts based route is substantial, primarily driven by the removal of expensive and hazardous reagents. Traditional lithiation methods require butyllithium, which is not only costly to purchase but also requires expensive disposal protocols for quenching residues. In contrast, the new method utilizes aluminum chloride, a ubiquitous and inexpensive industrial catalyst. Additionally, the energy savings from operating at near-ambient temperatures rather than minus 78 degrees Celsius result in a significantly reduced utility burden. The overall process efficiency is further enhanced by the high atom economy of the acylation-hydrolysis sequence, which minimizes waste generation and reduces the costs associated with environmental treatment and disposal of hazardous byproducts, leading to a leaner and more profitable production model.
- Enhanced Supply Chain Reliability: Supply chain continuity is critically dependent on the availability of starting materials and the robustness of the process. This synthesis utilizes m-fluorotoluene, a widely available bulk chemical, unlike the specialized brominated or nitrile precursors required by older methods which may have limited suppliers and longer lead times. The robustness of the reaction against minor variations in conditions means that production is less prone to batch failures or deviations that could disrupt supply. Moreover, the simplified workup procedure, which avoids complex distillation or chromatographic separations in favor of crystallization, allows for faster turnaround times between batches. This agility ensures that inventory levels can be maintained more effectively, reducing the need for large safety stocks and freeing up working capital for other strategic investments within the organization.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to multi-ton production is often fraught with challenges, particularly when dealing with exothermic reactions or unstable intermediates. This patented method demonstrates excellent scalability due to its thermal stability and the use of common solvents like 1,2-dichloroethane which are well-understood in large-scale processing. From an environmental perspective, the process aligns with increasingly stringent regulatory frameworks by avoiding heavy metal catalysts and reducing the volume of hazardous waste. The byproduct chloroform, while regulated, is generated in a contained manner and can be recovered or treated efficiently, unlike the complex salt wastes generated by nitrile hydrolysis. This environmental compatibility facilitates easier permitting for new production lines and enhances the corporate sustainability profile of the manufacturer, which is increasingly important for downstream customers in the pharmaceutical and electronics sectors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 4-fluoro-2-methylbenzoic acid using this advanced methodology. These insights are derived directly from the experimental data and comparative analysis presented in the patent literature, providing a clear understanding of why this route is superior for industrial applications. Understanding these nuances is essential for technical teams evaluating process transfers and for commercial teams negotiating supply agreements based on cost and quality metrics.
Q: What are the primary advantages of this synthesis route over traditional lithiation methods?
A: Unlike traditional methods requiring butyllithium at cryogenic temperatures (-78°C), this patented route operates at mild temperatures (0-10°C) using standard Lewis acid catalysis, significantly reducing energy consumption and safety risks associated with pyrophoric reagents.
Q: What purity levels can be achieved with this manufacturing process?
A: The patent data indicates that through optimized recrystallization steps using toluene, the final product 4-fluoro-2-methylbenzoic acid can achieve a purity of approximately 98.5% as determined by HPLC analysis, meeting stringent requirements for electronic and pharmaceutical applications.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes easily available raw materials like m-fluorotoluene and avoids complex anhydrous/anaerobic conditions required for Grignard reactions, making it highly scalable and cost-effective for commercial manufacturing of fine chemical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Fluoro-2-Methylbenzoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this novel synthesis are fully realized in practice. Our state-of-the-art facilities are equipped to handle the specific requirements of Friedel-Crafts chemistry, including corrosion-resistant reactors and advanced solvent recovery systems. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 4-fluoro-2-methylbenzoic acid meets the exacting standards required for electronic photoinitiators and pharmaceutical intermediates, providing our clients with absolute confidence in product consistency and performance.
We invite global partners to collaborate with us to leverage this cost-effective and scalable technology for their supply chains. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this route can optimize your bill of materials. We encourage you to contact our technical procurement team today to request specific COA data from our pilot runs and to discuss route feasibility assessments for your projects. By partnering with us, you secure not just a chemical supplier, but a strategic ally committed to driving efficiency and innovation in your manufacturing operations.
