Scalable Synthesis of 4-Fluoro-2-Methylbenzoic Acid via Novel Friedel-Crafts Acylation
The chemical industry is constantly seeking more efficient pathways for producing high-value intermediates, and the recent disclosure in patent CN110903182A presents a compelling solution for the synthesis of 4-fluoro-2-methylbenzoic acid. This compound serves as a critical building block for both electronic chemical photoinitiators and advanced pharmaceutical intermediates, yet its historical production methods have been plagued by complexity and high operational costs. The patented methodology introduces a streamlined approach that leverages a classic Friedel-Crafts acylation followed by a haloform oxidation, effectively bypassing the need for hazardous cryogenic operations. By utilizing readily available starting materials such as m-fluorotoluene and acetyl chloride, this process addresses the longstanding economic and safety bottlenecks associated with previous synthetic routes. For technical directors and procurement specialists alike, this represents a significant opportunity to optimize supply chains while maintaining rigorous quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-fluoro-2-methylbenzoic acid has relied on chemically aggressive and operationally demanding techniques that pose significant challenges for industrial scale-up. Prior art, such as the methods described in WO2004050651 and US6342504, often necessitates the use of Grignard reagents or organolithium species like butyllithium, which require strictly anhydrous and anaerobic environments. These reactions must be conducted at extremely low temperatures, typically around -78°C, demanding specialized cryogenic reactor infrastructure that drastically increases capital expenditure and energy consumption. Furthermore, the precursor materials for these routes, such as 2-bromo-5-fluorotoluene or 4-fluoro-2-methylbenzonitrile, involve multi-step syntheses themselves, adding layers of cost and supply chain vulnerability. The handling of pyrophoric reagents also introduces severe safety risks, complicating regulatory compliance and increasing insurance liabilities for manufacturing facilities.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a robust two-step sequence that begins with a Lewis acid-catalyzed acylation and concludes with a straightforward oxidative cleavage. This method operates under significantly milder conditions, with the initial acylation step proceeding efficiently between -5°C and 10°C, a range easily achievable with standard industrial cooling systems rather than expensive cryogenics. The subsequent conversion of the ketone intermediate to the carboxylic acid is achieved using sodium hypochlorite, a common and inexpensive oxidant that eliminates the need for dry ice or high-pressure carbonation steps. This shift in chemistry not only simplifies the reactor setup but also enhances the overall safety profile of the manufacturing process by removing reactive metal species from the workflow. The result is a synthetic pathway that is inherently more adaptable to continuous processing and large-batch production.
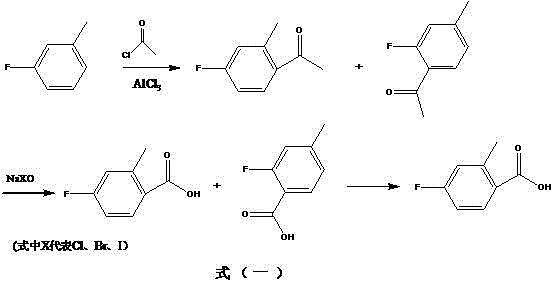
Mechanistic Insights into AlCl3-Catalyzed Acylation and Oxidation
The core of this synthetic strategy relies on the precise control of electrophilic aromatic substitution during the initial Friedel-Crafts acylation phase. In this step, anhydrous aluminum trichloride acts as a potent Lewis acid catalyst, coordinating with the carbonyl oxygen of acetyl chloride to generate a highly reactive acylium ion electrophile. This electrophile attacks the electron-rich aromatic ring of m-fluorotoluene, preferentially targeting positions ortho and para to the activating methyl group, although the fluorine substituent exerts a deactivating influence that helps moderate the reaction rate. The formation of the ketone intermediate, specifically 4-fluoro-2-methylacetophenone alongside its isomers, is a critical juncture where reaction temperature and catalyst loading must be tightly regulated to maximize the desired regioisomer ratio. Following acylation, the process transitions to a haloform reaction mechanism where the methyl ketone moiety undergoes exhaustive halogenation at the alpha-position by the hypohalite species. This poly-halogenated intermediate is then susceptible to nucleophilic attack by hydroxide ions, leading to the cleavage of the carbon-carbon bond and the formation of the carboxylate salt, which is subsequently acidified to yield the final benzoic acid derivative.
Impurity control is intrinsically built into the physicochemical properties of the intermediates and the final purification strategy employed in this patent. While the acylation step inevitably produces a mixture of ortho- and para-isomers due to the directing effects of the substituents on the toluene ring, the downstream processing is designed to handle this complexity without requiring difficult chromatographic separations. The oxidation step converts both isomeric ketones into their corresponding carboxylic acids, which possess distinct solubility profiles that can be exploited during the final workup. The patent specifies a recrystallization protocol using a binary solvent system of toluene and ethyl acetate, which selectively precipitates the target 4-fluoro-2-methylbenzoic acid while leaving soluble impurities and isomeric byproducts in the mother liquor. This reliance on crystallization rather than distillation or column chromatography is a hallmark of a process designed for high throughput and minimal waste generation, ensuring that the final product meets stringent purity specifications of approximately 98.5% as verified by HPLC analysis.
How to Synthesize 4-Fluoro-2-Methylbenzoic Acid Efficiently
The execution of this synthesis requires careful attention to the addition rates and temperature profiles during the exothermic acylation phase to ensure safety and reproducibility. Operators must maintain the reaction mixture under an inert nitrogen atmosphere to prevent moisture from deactivating the aluminum trichloride catalyst, which is sensitive to hydrolysis. Once the acylation is complete and the ketone intermediate is formed, the transition to the oxidative step involves the controlled addition of sodium hypochlorite solution, where pH management becomes crucial to drive the hydrolysis of the trihalomethyl group to completion. The final isolation involves acidification to precipitate the crude acid, followed by the critical recrystallization step that defines the commercial quality of the material. For a comprehensive breakdown of the specific operational parameters and safety protocols, please refer to the standardized guide below.
- Perform Friedel-Crafts acylation of m-fluorotoluene with acetyl chloride using anhydrous aluminum trichloride at -5 to 10°C.
- Conduct a haloform reaction on the resulting ketone intermediate using sodium hypochlorite aqueous solution at room temperature.
- Purify the crude acid product through recrystallization using a toluene and ethyl acetate solvent system to achieve high purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this patented methodology offers profound advantages by decoupling production from volatile supply chains associated with specialized organometallic reagents. The shift towards commodity chemicals like m-fluorotoluene and acetyl chloride means that procurement managers can leverage existing bulk purchasing agreements and spot market liquidity to secure raw materials at stable prices. This reduces the risk of production stoppages caused by the shortage of niche precursors like brominated fluoro-toluenes or nitriles, which often have limited global suppliers. Furthermore, the elimination of cryogenic requirements translates directly into lower utility costs and reduced maintenance overhead for manufacturing plants, as standard jacketed reactors can be utilized instead of specialized low-temperature vessels. These operational efficiencies compound to create a more resilient and cost-effective supply base for downstream customers in the pharmaceutical and electronics sectors.
- Cost Reduction in Manufacturing: The economic benefits of this route are driven primarily by the substitution of expensive, hazardous reagents with inexpensive, bulk-available commodities. By avoiding the use of butyllithium and dry ice, manufacturers eliminate the high costs associated with the storage, handling, and disposal of pyrophoric materials and cryogens. Additionally, the simplified workup procedure reduces the consumption of solvents and water, leading to lower waste treatment costs and a smaller environmental footprint. The overall process intensity is reduced, allowing for higher throughput per unit of time and equipment, which effectively lowers the fixed cost allocation per kilogram of the final product.
- Enhanced Supply Chain Reliability: The reliance on m-fluorotoluene as a starting material significantly de-risks the supply chain, as this chemical is produced on a massive scale for various fluorination applications globally. Unlike complex intermediates that may be sourced from a single region or manufacturer, m-fluorotoluene benefits from a diversified supplier base, ensuring continuity of supply even during regional disruptions. The robustness of the reaction conditions also means that production can be easily transferred between different manufacturing sites without the need for highly specialized infrastructure, providing flexibility in capacity planning and inventory management.
- Scalability and Environmental Compliance: The environmental profile of this synthesis is markedly improved by the avoidance of heavy metal catalysts and the generation of less hazardous waste streams. The use of sodium hypochlorite generates salt and water as byproducts, which are far easier to treat than the metal-containing sludge typical of Grignard or lithiation quenches. This alignment with green chemistry principles facilitates smoother regulatory approvals and reduces the burden on environmental health and safety teams. The process is inherently scalable, moving seamlessly from pilot plant batches to multi-ton commercial production without the engineering challenges posed by heat transfer limitations in cryogenic systems.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route, derived directly from the patent specifications and comparative analysis. Understanding these details is essential for evaluating the feasibility of adopting this technology within existing manufacturing frameworks. The answers provided reflect the specific advantages and operational parameters disclosed in the intellectual property documentation.
Q: What are the key advantages of this synthesis method over traditional lithiation routes?
A: Unlike traditional methods requiring cryogenic temperatures (-78°C) and pyrophoric reagents like butyllithium, this patented process operates under mild conditions (-5 to 10°C) using stable, commodity chemicals, significantly reducing operational hazards and equipment costs.
Q: How is the isomeric purity managed in this process?
A: The process generates ortho- and para-isomers during acylation, but the final purification step utilizes a specific recrystallization protocol with toluene and ethyl acetate to effectively isolate the target 4-fluoro-2-methylbenzoic acid with purity exceeding 98.5%.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the use of inexpensive starting materials like m-fluorotoluene and acetyl chloride, combined with the elimination of extreme low-temperature requirements, makes this route highly scalable and economically viable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Fluoro-2-Methylbenzoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a stable supply of high-quality intermediates for your most demanding applications. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We are committed to delivering products that adhere to stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify every batch. Our capability to implement the efficient synthesis methods described in recent patents allows us to offer competitive pricing without compromising on the quality essential for pharmaceutical and electronic grade materials.
We invite you to engage with our technical procurement team to discuss how our optimized manufacturing processes can support your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our production efficiencies translate into value for your organization. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your supply chain objectives.
