Advanced Synthesis of Brivaracetam Intermediates via Lewis Acid Catalysis for Commercial Scale-up
Advanced Synthesis of Brivaracetam Intermediates via Lewis Acid Catalysis for Commercial Scale-up
The pharmaceutical landscape for antiepileptic drugs continues to evolve, driven by the need for safer, more efficient manufacturing processes for third-generation therapeutics like Brivaracetam. Patent CN111196771B introduces a transformative approach to synthesizing the critical intermediate required for Brivaracetam production, addressing long-standing challenges in yield and environmental safety. This technology leverages a novel Lewis acid catalyzed ring-opening strategy that bypasses the hazardous halogenation steps typical of legacy methods. By utilizing readily available starting materials such as (R)-4-propyldihydrofuran-2-one and (S)-2-aminobutanamide, the process achieves high optical purity and operational simplicity. For global procurement teams, this represents a significant opportunity to secure a reliable pharmaceutical intermediate supplier capable of delivering consistent quality without the supply chain risks associated with toxic reagents.
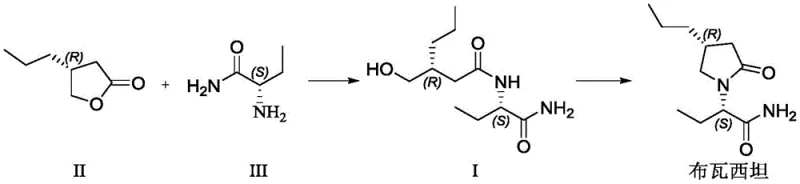
The core innovation lies in the direct formation of the amide linkage followed by cyclization, streamlining what was previously a fragmented and dangerous synthetic pathway. The method demonstrates exceptional control over stereochemistry, ensuring that the chiral integrity of the starting materials is preserved throughout the synthesis. This level of precision is paramount for meeting the stringent regulatory requirements of major markets like the FDA and EMA. Furthermore, the elimination of purification bottlenecks allows for a more continuous and scalable manufacturing flow, directly supporting the commercial scale-up of complex chiral molecules needed for high-volume API production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of Brivaracetam has been plagued by inefficient and hazardous synthetic routes that pose significant risks to both operational safety and cost structures. One prominent prior art method, disclosed in CN1882535B, relies on a condensation-reduction-ammoniation sequence that necessitates multiple column chromatography purification steps. This reliance on chromatography is a major bottleneck for industrial application, as it drastically reduces total yield and increases processing time, making it economically unviable for large-scale cost reduction in API manufacturing. Additionally, another common route described in CN106365986A involves the preparation of 4-halobutyryl chloride using aggressive chlorinating agents.
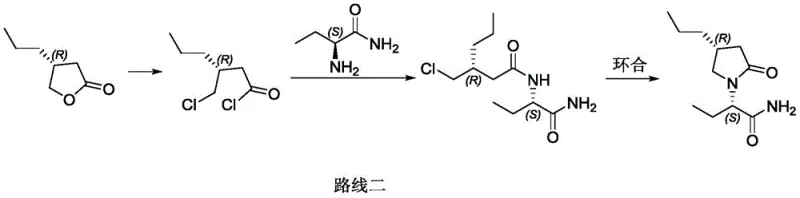
The use of reagents such as phosgene or thionyl chloride in these traditional pathways introduces severe environmental and safety liabilities. These chemicals are highly toxic and corrosive, requiring specialized containment infrastructure and generating substantial amounts of acidic waste gas that must be treated before release. The instability of the resulting halogenated intermediates further complicates storage and transportation, adding hidden logistics costs and supply chain fragility. Moreover, the harsh reaction conditions often lead to lower reaction yields and increased formation of impurities, forcing manufacturers to invest heavily in downstream purification to meet pharmacopeial standards. These factors collectively render conventional methods less attractive for modern, sustainability-focused chemical enterprises.
The Novel Approach
In stark contrast, the novel methodology presented in the patent data offers a streamlined, two-step synthesis that fundamentally redesigns the construction of the Brivaracetam scaffold. Instead of engaging in risky halogenation, the new route employs a direct ring-opening reaction between a cyclic ketone and an amino-amide derivative under mild Lewis acid catalysis. This approach not only simplifies the operational workflow by removing dangerous reagents but also significantly enhances the overall atom economy of the process. The reaction proceeds smoothly in common organic solvents like tetrahydrofuran or acetone at moderate temperatures ranging from 10°C to 35°C, which minimizes energy consumption and thermal stress on the chiral centers.
Following the formation of the linear intermediate, a subsequent acid-catalyzed cyclization step efficiently closes the pyrrolidinone ring to yield the final active pharmaceutical ingredient. This cyclization is performed in solvents such as toluene or isopropyl acetate at temperatures between 80°C and 100°C, conditions that are easily manageable in standard glass-lined or stainless steel reactors. The result is a robust process that delivers high-purity antiepileptic intermediates with yields exceeding 80% for the intermediate step and roughly 77% for the final cyclization. By avoiding the pitfalls of chromatography and hazardous gas generation, this method establishes a new benchmark for efficiency and safety in the production of next-generation neurological therapeutics.
Mechanistic Insights into Lewis Acid Catalyzed Ring-Opening
The success of this synthetic route hinges on the precise activation of the lactone ring in (R)-4-propyldihydrofuran-2-one by a Lewis acid catalyst. In this mechanism, the Lewis acid, such as tetraisopropyl titanate or anhydrous aluminum chloride, coordinates with the carbonyl oxygen of the lactone. This coordination increases the electrophilicity of the carbonyl carbon, making it highly susceptible to nucleophilic attack by the amino group of the (S)-2-aminobutanamide. This activation allows the ring-opening to proceed rapidly under mild thermal conditions, preventing the thermal degradation or racemization that might occur at higher temperatures. The choice of catalyst is critical; experimental data indicates that titanium-based Lewis acids provide an optimal balance of reactivity and selectivity, ensuring that the chiral configuration at the alpha-position remains intact.
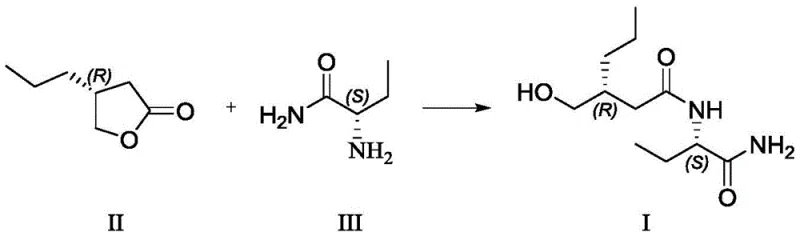
Furthermore, the mechanistic pathway is designed to suppress side reactions that typically plague amide bond formations in complex molecules. The specific stoichiometry, utilizing a molar ratio of Lewis acid to substrate between 1.10:1 and 1.50:1, ensures complete conversion while minimizing the formation of oligomeric byproducts. The subsequent workup involves a straightforward aqueous wash to remove the metal catalyst salts, followed by crystallization which further purifies the intermediate. This inherent ability of the chemistry to self-purify through crystallization is a key factor in achieving the reported chiral purity of over 99.5%. For R&D directors, understanding this mechanism highlights the robustness of the process, as it relies on fundamental coordination chemistry principles that are well-understood and easily controlled in a GMP environment.
How to Synthesize Brivaracetam Intermediate Efficiently
The synthesis of the key intermediate, Formula I, serves as the foundation for the entire Brivaracetam production line and requires strict adherence to optimized reaction parameters to ensure maximum yield and purity. The process begins with the careful selection of anhydrous solvents and high-purity starting materials to prevent premature hydrolysis of the Lewis acid catalyst. Operators must maintain the reaction temperature within the narrow window of 10°C to 35°C to balance reaction kinetics with stereochemical stability. Detailed standard operating procedures regarding the addition rate of the catalyst and the stirring speed are essential to maintain homogeneity and heat transfer throughout the batch. The following guide outlines the critical phases of this synthesis based on the patented methodology.
- Perform ring-opening reaction of (R)-4-propyldihydrofuran-2-one with (S)-2-aminobutanamide using a Lewis acid catalyst like tetraisopropyl titanate in THF at 10-35°C.
- Isolate the intermediate compound Formula I through extraction and crystallization.
- Conduct condensation and cyclization of Formula I using concentrated sulfuric acid or phosphoric acid in toluene at 80-100°C to yield Brivaracetam.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers profound strategic advantages that extend beyond simple chemical yield. The primary benefit lies in the drastic simplification of the supply chain for raw materials; by eliminating the need for phosgene, thionyl chloride, and other regulated toxic substances, companies can avoid the rigorous permitting, storage, and handling costs associated with hazardous chemicals. This reduction in regulatory burden translates directly into lower operational overhead and reduced risk of production stoppages due to compliance audits. Furthermore, the stability of the intermediates allows for more flexible inventory management, enabling manufacturers to stockpile key precursors without the fear of rapid degradation, thereby enhancing overall supply continuity.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the elimination of expensive purification technologies and hazardous waste treatment. Traditional methods often require silica gel column chromatography, which is prohibitively expensive and slow at an industrial scale; replacing this with simple crystallization and extraction significantly lowers the cost of goods sold. Additionally, the avoidance of corrosive acid gases reduces the maintenance costs for reactor vessels and ventilation systems, extending the lifespan of capital equipment. The high yield of the reaction means that less raw material is wasted, maximizing the output per batch and improving the overall return on investment for the manufacturing facility.
- Enhanced Supply Chain Reliability: Reliability is bolstered by the use of commodity chemicals and solvents that are widely available in the global market, reducing dependency on niche suppliers of dangerous reagents. The mild reaction conditions reduce the likelihood of runaway reactions or equipment failures, ensuring consistent batch-to-batch performance. This stability allows for more accurate forecasting and planning, which is crucial for meeting the demanding delivery schedules of multinational pharmaceutical clients. By securing a process that is less prone to environmental shutdowns or safety incidents, supply chain leaders can guarantee a steady flow of high-purity pharmaceutical intermediates to their downstream partners.
- Scalability and Environmental Compliance: From an environmental perspective, this route aligns perfectly with green chemistry principles by minimizing waste generation and avoiding the release of ozone-depleting or toxic substances. The process generates significantly less acidic waste compared to halogenation routes, simplifying wastewater treatment and reducing the environmental footprint of the plant. This compliance advantage is increasingly valuable as global regulations on industrial emissions tighten. The scalability of the process is proven by its ability to function effectively in standard reactor setups without requiring exotic high-pressure or cryogenic equipment, facilitating a smooth transition from pilot plant to full commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Brivaracetam synthesis technology. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical implications of adopting this route. Understanding these details is essential for technical teams evaluating the feasibility of technology transfer and for commercial teams assessing the long-term viability of the supply partnership.
Q: How does this new method improve upon traditional halogenation routes?
A: Traditional routes often require hazardous chlorinating agents like phosgene or thionyl chloride, generating toxic acid gas waste. This novel method utilizes a direct Lewis acid catalyzed ring-opening, eliminating the need for dangerous halogenation steps and significantly improving environmental safety and operational simplicity.
Q: What represents the key advantage regarding optical purity in this synthesis?
A: The use of specific Lewis acids, such as tetraisopropyl titanate, under mild temperatures (10-35°C) effectively prevents the racemization of the chiral center. This ensures the final Brivaracetam product maintains high chiral purity (over 99.5% in optimized examples), which is critical for pharmaceutical efficacy.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process avoids complex column chromatography purification steps required by older methods. By relying on standard solvent extractions and crystallizations with high yields (over 80% for the intermediate), the route is highly amenable to multi-kilogram and ton-scale manufacturing without prohibitive cost increases.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Brivaracetam Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to a superior synthetic route requires a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this Lewis acid catalyzed process are fully realized at scale. Our facilities are equipped with state-of-the-art reaction vessels capable of handling the specific solvent systems and temperature profiles required for this synthesis, all while maintaining stringent purity specifications. Our rigorous QC labs utilize advanced chiral HPLC methods to verify the optical purity of every batch, guaranteeing that the final product meets the highest international standards for neurological medications.
We invite potential partners to engage with our technical procurement team to discuss how this innovative methodology can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the specific economic benefits of switching to this non-halogenated route for your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project needs. Let us collaborate to bring safer, more efficient, and high-quality Brivaracetam to the global market, leveraging our shared commitment to excellence in pharmaceutical manufacturing.
