Advanced One-Step Synthesis of Nitrogen-Containing Aromatic Heterocyclic Diamines for Industrial Scale-Up
The chemical industry is constantly seeking more efficient pathways to synthesize high-performance monomers, and the recent disclosure in patent CN112679418A represents a significant leap forward in the production of nitrogen-containing aromatic heterocyclic diamines. These specialized compounds serve as critical building blocks for advanced polyimide resins, high-temperature resistant polymers, and sophisticated agrochemical intermediates. Traditionally, accessing these complex heterocyclic structures required laborious multi-step syntheses that were prone to low yields and difficult purification challenges. However, this new methodology introduces a robust one-step Suzuki-Miyaura coupling strategy that directly constructs the carbon-carbon bonds between amino-substituted phenyl rings and nitrogen-containing heterocycles. By leveraging this innovative approach, manufacturers can now access a broad spectrum of diamine derivatives with exceptional purity and structural diversity, addressing the growing demand for reliable nitrogen-containing aromatic heterocyclic diamine suppliers in the global market.
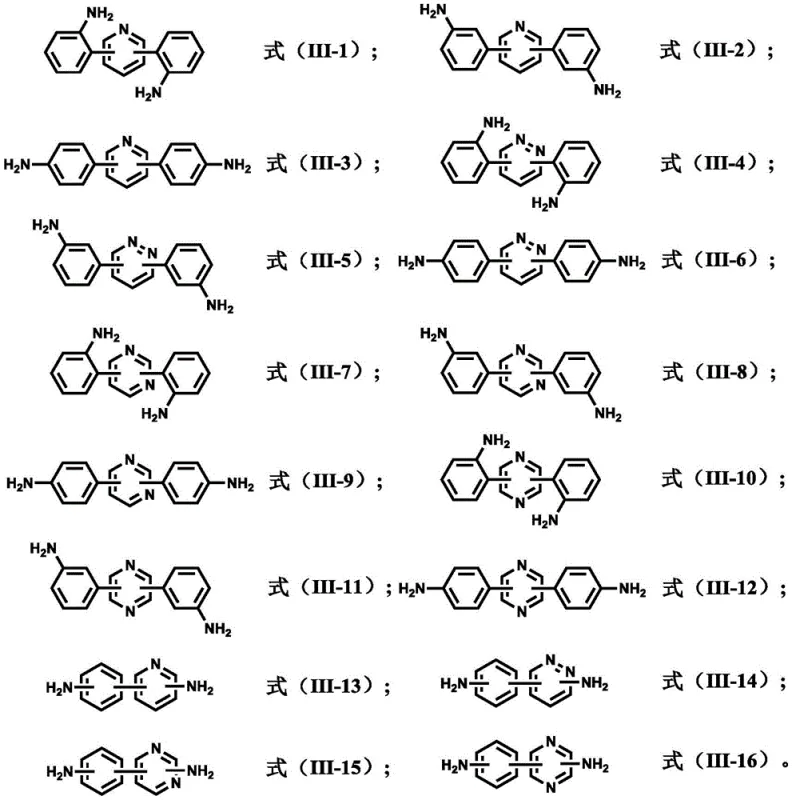
The versatility of this synthetic route is immediately apparent when examining the wide array of structures accessible through this single protocol. As illustrated in the structural library, the method accommodates various heterocyclic cores including pyridine, pyrimidine, pyrazine, and pyridazine, allowing for fine-tuning of the electronic and steric properties of the final polymer or active ingredient. This flexibility is crucial for R&D teams aiming to optimize material properties such as thermal stability, solubility, or mechanical strength in downstream applications. Furthermore, the ability to introduce amino groups at specific positions (ortho, meta, or para) on the phenyl rings provides chemists with precise control over the polymerization behavior of these monomers. Such structural precision is often the differentiating factor between a standard commodity chemical and a high-value specialty intermediate capable of commanding premium pricing in sectors like microelectronics and aerospace materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of nitrogen-containing aromatic heterocyclic diamines was fraught with inefficiencies that hindered both research speed and commercial viability. Conventional routes typically involved lengthy sequences of protection and deprotection steps, harsh reaction conditions that compromised sensitive functional groups, and the use of expensive or toxic reagents that complicated waste management. These multi-step processes not only increased the overall cost of goods sold due to cumulative yield losses but also extended the lead time for high-purity nitrogen-containing aromatic heterocyclic diamines significantly. Additionally, the purification of intermediates often required column chromatography or recrystallization from difficult solvent systems, creating bottlenecks in production capacity. For procurement managers, these complexities translated into volatile supply chains and unpredictable pricing, as any disruption in the supply of a specific intermediate could halt the entire synthesis line. The environmental footprint of these older methods was also substantial, generating large volumes of organic waste that required costly treatment before disposal.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a direct cross-coupling reaction that streamlines the entire manufacturing process into a single operational unit. By reacting an amino-substituted phenylboronic acid derivative directly with a halogenated nitrogen-containing heterocycle, the method eliminates the need for intermediate isolation and purification steps. This telescoped process not only reduces the total number of unit operations but also minimizes solvent usage and energy consumption, aligning perfectly with modern green chemistry principles. The reaction conditions are remarkably mild yet effective, utilizing common palladium catalysts and inorganic bases in biphasic solvent systems like toluene and water. This simplicity allows for a straightforward workup procedure where the product often precipitates out of the reaction mixture upon cooling, enabling recovery via simple filtration. Such operational ease drastically lowers the barrier to entry for commercial scale-up of complex heterocyclic intermediates, making it an attractive option for manufacturers looking to optimize their production lines.
Mechanistic Insights into Palladium-Catalyzed Suzuki Coupling
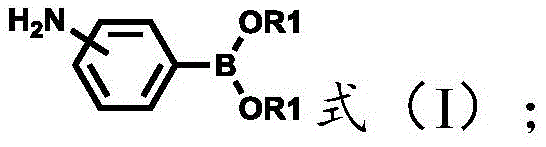
At the heart of this transformative synthesis lies the well-established yet highly adaptable mechanism of the Suzuki-Miyaura cross-coupling reaction. The catalytic cycle begins with the oxidative addition of the palladium(0) species into the carbon-halogen bond of the nitrogen-containing heterocyclic substrate, forming a reactive organopalladium(II) intermediate. This step is critical and is facilitated by the electron-deficient nature of the heterocyclic ring, which activates the halide towards metal insertion. Subsequently, the base present in the reaction mixture, such as potassium carbonate or cesium fluoride, activates the boronic acid ester by forming a boronate species, which is more nucleophilic and capable of transmetallation. The transfer of the aryl group from the boron atom to the palladium center generates a di-organopalladium complex, setting the stage for the final reductive elimination step. It is during this reductive elimination that the new carbon-carbon bond is forged, releasing the desired diamine product and regenerating the active palladium(0) catalyst to continue the cycle. Understanding this mechanistic pathway is essential for troubleshooting and optimizing the reaction, particularly when dealing with sterically hindered substrates or less reactive halides.
Impurity control is another vital aspect where this mechanistic understanding pays dividends, ensuring the delivery of high-purity aromatic diamines required for sensitive applications like semiconductor manufacturing. One common side reaction in Suzuki couplings is homocoupling, where two boronic acid molecules or two halide molecules couple with each other instead of cross-coupling. However, the specific stoichiometry and catalyst loading optimized in this patent—typically using a slight excess of the boronic acid relative to the halide—effectively suppresses homocoupling of the valuable heterocyclic component. Furthermore, the choice of ligand on the palladium catalyst, such as triphenylphosphine, stabilizes the active species and prevents the formation of palladium black, which can lead to catalyst deactivation and metal contamination in the final product. The high selectivity observed in the examples, with yields consistently exceeding 80%, indicates that the reaction conditions effectively minimize the formation of mono-coupled intermediates or dehalogenated byproducts. This level of purity is paramount for polymerization reactions, where even trace impurities can act as chain terminators, severely affecting the molecular weight and mechanical properties of the resulting polyimide or polyamide materials.
How to Synthesize Nitrogen-Containing Aromatic Heterocyclic Diamine Efficiently
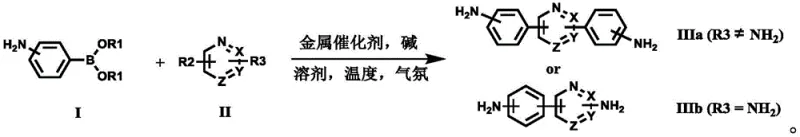
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to the specific parameters outlined in the patent to ensure reproducibility and safety. The process begins with the preparation of the reaction vessel under an inert atmosphere, typically nitrogen or argon, to prevent oxidation of the sensitive palladium catalyst and the boronic acid reagent. The reactants, including the amino-phenylboronic acid ester and the heterocyclic dihalide, are combined with the catalyst and base in a suitable solvent system, often a mixture of an organic solvent like toluene or dioxane with water to facilitate the dissolution of the inorganic base. The reaction mixture is then heated to a temperature range of 80°C to 130°C, depending on the reactivity of the specific substrates involved, and stirred vigorously to ensure efficient mass transfer between the phases. Monitoring the reaction progress via thin-layer chromatography (TLC) is recommended to determine the exact endpoint, ensuring complete consumption of the limiting reagent before proceeding to workup. The detailed standardized synthesis steps见下方的指南。
- Mix amino-substituted phenylboronic acid ester, nitrogen-containing heterocyclic halide, palladium catalyst, and alkali metal compound in a solvent system under inert atmosphere.
- Heat the reaction mixture to temperatures between 50°C and 150°C, monitoring progress via TLC until the halide starting material is fully consumed.
- Cool the reaction to precipitate the solid product, followed by filtration, washing with water and organic solvents, and vacuum drying to obtain high-purity diamine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this one-step synthesis technology offers tangible benefits that extend far beyond the laboratory bench, directly impacting the bottom line and operational resilience. The simplification of the manufacturing process translates into a significant reduction in capital expenditure and operating costs, as fewer reactors and separation units are required to produce the same volume of product. Moreover, the use of commercially available and relatively inexpensive starting materials, such as substituted boronic acids and halogenated heterocycles, ensures a stable and secure supply chain that is less susceptible to the volatility associated with exotic or custom-synthesized precursors. The robustness of the reaction conditions also means that the process is forgiving of minor variations in raw material quality, further enhancing supply chain reliability. By reducing the complexity of the synthesis, manufacturers can also respond more quickly to fluctuations in market demand, scaling production up or down without the long lead times associated with multi-step campaigns.
- Cost Reduction in Manufacturing: The economic impact of eliminating multiple synthetic steps cannot be overstated, as each skipped step removes the associated costs of solvents, reagents, energy, and labor. In traditional multi-step syntheses, the cumulative cost of purification and isolation after each stage often accounts for a major portion of the total manufacturing expense. By consolidating the synthesis into a single pot reaction, this method drastically cuts down on solvent consumption and waste disposal fees, which are increasingly stringent and costly in the chemical industry. Additionally, the high yields reported in the patent examples suggest that raw material utilization is highly efficient, minimizing the cost per kilogram of the final active pharmaceutical ingredient or polymer monomer. This efficiency allows suppliers to offer more competitive pricing without sacrificing margins, providing a distinct advantage in cost-sensitive markets.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the reliance on complex, multi-vendor supply chains for specialized intermediates. This new method mitigates that risk by utilizing commodity chemicals that are widely produced and stocked by multiple global suppliers. The resilience of the supply chain is further bolstered by the simplicity of the process, which reduces the likelihood of batch failures due to operational errors or equipment malfunctions. With fewer unit operations, there are fewer points of failure in the production line, ensuring a consistent and reliable flow of product to customers. This reliability is particularly valuable for long-term contracts in the pharmaceutical and electronics industries, where supply interruptions can have cascading effects on downstream manufacturing schedules and product launches.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to the plant is notoriously difficult, often requiring complete re-optimization of reaction parameters. However, the straightforward nature of this Suzuki coupling, utilizing standard heating and stirring techniques in common solvents, facilitates a smooth transition to industrial scale. The examples provided in the patent demonstrate successful reactions on scales ranging from grams to over 100 grams, indicating a clear path to ton-scale production. From an environmental perspective, the process aligns with sustainability goals by reducing the E-factor (mass of waste per mass of product) through minimized solvent use and the avoidance of hazardous reagents. The ability to recover and potentially recycle the palladium catalyst further enhances the environmental profile, helping companies meet increasingly rigorous regulatory standards and corporate social responsibility targets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this novel synthesis technology. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for potential partners and clients. Understanding these details is crucial for evaluating the feasibility of integrating this method into existing production workflows or for specifying the correct grade of material for specific applications. We encourage technical teams to review these insights to fully appreciate the capabilities and limitations of the process.
Q: What are the primary advantages of this one-step synthesis method over traditional routes?
A: The primary advantage is the drastic simplification of the synthetic route. Traditional methods often require multiple steps with complex purification between each stage. This patented one-step Suzuki coupling directly links the aromatic rings, significantly reducing operation time, solvent consumption, and waste generation while maintaining high conversion rates and selectivity.
Q: Which catalysts and bases are most effective for this transformation?
A: The patent specifies that palladium-based catalysts such as Pd(PPh3)4, Pd(OAc)2, or PdCl2(dppf) are highly effective. For the base, inorganic carbonates like K2CO3, Cs2CO3, or phosphates like K3PO4 provide optimal results, facilitating the transmetallation step essential for the coupling reaction.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is explicitly designed for industrial mass production. The use of common solvents like toluene and water, combined with a simple workup procedure involving filtration and washing rather than complex chromatography, makes it highly scalable from kilogram to multi-ton quantities without significant loss in efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nitrogen-Containing Aromatic Heterocyclic Diamine Supplier
As the global demand for high-performance polymers and advanced pharmaceutical intermediates continues to surge, the ability to deliver high-quality nitrogen-containing aromatic heterocyclic diamines efficiently is more critical than ever. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging cutting-edge synthetic methodologies like the one described in CN112679418A to provide superior chemical solutions. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volume requirements of even the largest multinational corporations. We are committed to maintaining stringent purity specifications through our rigorous QC labs, employing advanced analytical techniques to verify the identity and purity of every batch before it leaves our facility. This dedication to quality assurance guarantees that our customers receive materials that perform consistently in their downstream applications, whether it be in the fabrication of flexible electronics or the synthesis of life-saving drugs.
We invite you to explore the potential of this innovative synthesis route for your specific project needs and to discuss how our expertise can drive value for your organization. By partnering with us, you gain access not just to a product, but to a comprehensive technical support system dedicated to your success. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your current supply chain. We are ready to provide specific COA data and route feasibility assessments to help you make informed decisions and accelerate your time to market. Let us be your trusted partner in navigating the complexities of fine chemical manufacturing and achieving your strategic goals.
