Advanced Pd-Catalyzed Hydrogenation for High-Purity Doxycycline Manufacturing
Advanced Pd-Catalyzed Hydrogenation for High-Purity Doxycycline Manufacturing
The pharmaceutical industry continuously seeks robust synthetic routes that balance high stereochemical purity with environmental sustainability, particularly for critical antibiotic intermediates. Patent CN102086165A introduces a transformative approach to the production of doxycycline intermediates, specifically addressing the long-standing challenges in the hydrogenation of 11α-chloro-6-methylene oxytetracycline derivatives. This technology leverages a novel polyvinyl chloride polyamine-supported palladium complex (pvc-pp-Pd) to achieve exceptional stereoselectivity without relying on toxic modifying agents. For R&D directors and process chemists, this represents a significant leap forward, offering a pathway to produce α-6-deoxyoxytetracycline sulfosalicylate with yields exceeding 90% and beta-isomer content suppressed to below 1%. The elimination of carcinogenic poisoning agents like methylthiourea not only simplifies the purification workflow but also aligns with increasingly stringent global environmental regulations regarding hazardous waste discharge.
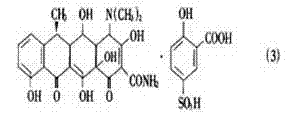
From a commercial perspective, the ability to recycle the heterogeneous catalyst while maintaining high activity addresses two critical pain points: raw material costs and supply chain continuity. Traditional homogeneous catalysts, while selective, often suffer from difficult separation and metal contamination issues that complicate downstream processing. In contrast, this supported system allows for simple filtration and reuse, drastically reducing the operational expenditure associated with precious metal loss. Furthermore, the mild reaction conditions (30-90°C, 0.4-0.7 MPa) ensure that the process is scalable and safe for large-scale manufacturing environments, providing a reliable foundation for consistent API supply.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of doxycycline has relied heavily on palladium on carbon (Pd/C) catalysts or homogeneous rhodium complexes, both of which present significant drawbacks in terms of efficiency and safety. The conventional Pd/C catalyzed hydrogenation suffers from poor stereoselectivity, typically yielding only about 60% of the desired alpha-isomer while generating up to 10% of the biologically inactive beta-isomer. To mitigate this, manufacturers have been forced to introduce toxic poisoning agents such as methylthiourea into the reaction system to artificially enhance selectivity. This practice introduces severe environmental and safety liabilities, as these sulfur-containing compounds are carcinogenic and require complex, costly waste treatment protocols to remove before the final product can meet pharmacopoeial standards. Additionally, homogeneous catalysts like triphenylphosphine rhodium chloride, while offering better selectivity, are notoriously difficult to separate from the product stream, leading to potential heavy metal contamination and the inability to recover expensive rhodium, thereby inflating production costs.
The Novel Approach
The innovative methodology described in the patent circumvents these issues by employing a specially designed polyvinyl chloride polyamine-supported palladium catalyst. This heterogeneous system inherently possesses high catalytic activity and stereocontrol, rendering the addition of external toxic modifiers entirely unnecessary. By utilizing the nitrogen-rich environment of the polyamine support, the catalyst directs the hydrogenation specifically to form the alpha-configuration, achieving yields greater than 90% with beta-isomer levels consistently maintained between 0.15% and 0.8%. This represents a dramatic improvement over the 2% limit often stipulated by pharmacopoeias. Moreover, the solid nature of the catalyst facilitates easy recovery via simple filtration, allowing it to be recycled multiple times without significant loss of activity. This dual benefit of enhanced selectivity and recyclability creates a cleaner, more cost-effective manufacturing process that is highly attractive for modern green chemistry initiatives.
Mechanistic Insights into Polymer-Supported Palladium Catalysis
The success of this hydrogenation process lies in the unique microenvironment created by the polyvinyl chloride polyamine (pvc-pp) support. Unlike simple carbon supports which offer little electronic interaction with the metal center, the polyamine ligands coordinate with the palladium atoms, modulating their electronic density and steric accessibility. This coordination likely stabilizes specific transition states during the hydrogen addition to the exocyclic methylene group at the C-6 position, favoring the formation of the thermodynamically stable alpha-methyl configuration over the beta-epimer. The polymer matrix acts not just as a carrier but as a functional ligand that mimics the selectivity of homogeneous catalysts while retaining the physical properties of a heterogeneous system. This synergy allows the reaction to proceed efficiently under relatively mild hydrogen pressures of 0.4 to 0.7 MPa, reducing the energy input required compared to more forcing conditions often needed for less active catalysts.
Furthermore, the mechanism ensures superior impurity control by minimizing side reactions that typically plague tetracycline chemistry, such as epimerization at other chiral centers or over-reduction. The absence of sulfur-based poisons means there is no risk of sulfur contamination in the final API, a critical quality attribute for regulatory compliance. The robustness of the covalent bonding between the palladium and the polymer support prevents metal leaching, ensuring that the filtrate contains negligible amounts of residual palladium. This inherent purity simplifies the downstream salt formation step with sulfosalicylic acid, resulting in a crystalline product that requires minimal recrystallization to meet stringent purity specifications. For process engineers, this translates to a shorter cycle time and higher overall plant throughput.
How to Synthesize α-6-Deoxyoxytetracycline Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for implementing this technology in a pilot or production setting. The process begins with the preparation of the catalyst itself, involving the swelling of PVC resin with polyethylene polyamine followed by metallation with palladium dichloride. Once the catalyst is prepared, the hydrogenation is conducted by suspending the 11α-chloro-6-methylene oxytetracycline substrate in a solvent such as ethanol or methanol. The reaction is monitored via HPLC to ensure complete conversion before the catalyst is filtered off for reuse. The filtrate is then treated with solid sulfosalicylic acid to precipitate the target sulfosalicylate salt. Detailed standardized operating procedures for each of these stages, including specific stoichiometric ratios and workup parameters, are essential for reproducibility.
- Preparation of the pvc-pp-Pd catalyst by swelling PVC with polyethylene polyamine followed by palladium dichloride complexation.
- Hydrogenation of the chloro-methylene precursor in alcohol solvents at 30-90°C and 0.4-0.7 MPa pressure.
- Filtration to recover the catalyst, followed by salt formation with sulfosalicylic acid to isolate the pure alpha-isomer.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this catalytic technology offers substantial strategic benefits beyond mere technical performance. The primary value driver is the significant reduction in manufacturing costs achieved through the elimination of expensive and hazardous auxiliary chemicals. By removing the need for toxic poisoning agents, companies save on both the purchase price of these reagents and the substantial costs associated with their disposal and environmental remediation. Additionally, the recyclability of the palladium catalyst means that the consumption of this precious metal is drastically lowered over time, insulating the production budget from volatility in the global palladium market. This economic efficiency is compounded by the high yield of the reaction, which maximizes the output from every kilogram of starting material, effectively lowering the cost of goods sold (COGS) for the final antibiotic intermediate.
- Cost Reduction in Manufacturing: The process eliminates the recurring expense of purchasing toxic modifiers and reduces the burden on waste treatment facilities, leading to direct operational savings. The ability to recover and reuse the catalyst multiple times further amortizes the initial investment in palladium, creating a leaner cost structure that enhances competitiveness in the generic pharmaceutical market.
- Enhanced Supply Chain Reliability: By simplifying the synthesis route and removing dependency on hard-to-source or regulated toxic reagents, the supply chain becomes more resilient. The robust nature of the heterogeneous catalyst ensures consistent batch-to-batch quality, reducing the risk of production delays caused by failed batches or out-of-specification results that require reprocessing.
- Scalability and Environmental Compliance: The mild reaction conditions and lack of hazardous byproducts make this process highly scalable from pilot plants to multi-ton commercial reactors without requiring major infrastructure upgrades. Furthermore, the green chemistry profile of the process facilitates easier regulatory approval and aligns with corporate sustainability goals, reducing the risk of future regulatory shutdowns due to environmental non-compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented hydrogenation technology. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers are derived directly from the experimental data and claims presented in the patent documentation, ensuring accuracy and relevance for decision-making purposes.
Q: How does the pvc-pp-Pd catalyst improve stereoselectivity compared to traditional Pd/C?
A: Unlike traditional Pd/C which requires toxic poisoning agents like methylthiourea to achieve modest selectivity, the pvc-pp-Pd catalyst utilizes the polyamine support to create a specific coordination environment. This intrinsic stereocontrol yields over 90% conversion with less than 1% of the inactive beta-isomer, eliminating the need for hazardous additives.
Q: Is the supported palladium catalyst recyclable for industrial scale-up?
A: Yes, a key advantage of this heterogeneous system is the ease of separation. The catalyst can be filtered directly from the reaction mixture after hydrogenation and reused, significantly reducing precious metal consumption and waste generation compared to homogeneous rhodium or iridium systems.
Q: What are the typical reaction conditions for this hydrogenation process?
A: The process operates under mild conditions, typically requiring temperatures between 30°C and 90°C and hydrogen pressures ranging from 0.4 to 0.7 MPa. Common solvents include methanol, ethanol, or acetone, making it compatible with standard pharmaceutical reactor setups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Doxycycline Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced catalytic technologies to maintain a competitive edge in the pharmaceutical intermediate market. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the pvc-pp-Pd hydrogenation process can be seamlessly transferred from the lab to the plant. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instruments to verify that every batch of doxycycline intermediate meets the highest global standards for stereochemical purity and residual metal content.
We invite you to collaborate with us to leverage this cutting-edge synthesis route for your supply chain needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this greener, more efficient process can improve your bottom line. Please contact us today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable and cost-effective supply of high-quality antibiotic intermediates.
