Advanced Cu-Pd-CeO2 Catalyst for Efficient Benzopyrazine Manufacturing and Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to synthesize nitrogen-containing heterocycles, particularly benzopyrazine (quinoxaline) derivatives, which serve as critical scaffolds for numerous bioactive compounds ranging from antibiotics to anticancer agents. A significant technological breakthrough in this domain is documented in Chinese Patent CN113828342B, which discloses a novel preparation method for a Cu-Pd-CeO2/γ-Al2O3@NP catalyst and its application in the efficient one-pot synthesis of benzopyrazine compounds. This innovation addresses long-standing challenges in catalytic efficiency and environmental sustainability by introducing a multi-component heterogeneous system that operates under solvent-free conditions. For R&D directors and process chemists, this patent represents a pivotal shift away from expensive, hard-to-separate homogeneous catalysts towards a more industrially viable, recyclable solid catalyst system that maintains high activity and selectivity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of benzopyrazine and its derivatives has relied heavily on the condensation of highly active ortho-dicarbonyl compounds with aromatic 1,2-diamines, a route that often involves expensive starting materials and multi-step purification processes. Alternatively, literature methods utilizing o-nitroanilines and aliphatic diols have employed transition metal complexes such as ruthenium carbonyls, iridium-hydroxypyridyl complexes, or cobalt-phenanthroline systems. While these homogeneous catalytic systems can achieve decent yields, they suffer from severe drawbacks including the necessity for toxic organic solvents like toluene or 1,2-dichloroethane, high reaction temperatures exceeding 140°C, and prolonged reaction times up to 24 hours. Furthermore, the separation of these homogeneous catalysts from the final product is notoriously difficult, often requiring complex chromatographic techniques or extensive washing, which leads to product loss and increased waste generation, making them unsuitable for large-scale commercial manufacturing.
The Novel Approach
In stark contrast, the technology disclosed in patent CN113828342B introduces a groundbreaking heterogeneous catalytic system that streamlines the production of high-purity benzopyrazine intermediates. This novel approach utilizes a specially engineered Cu-Pd-CeO2/γ-Al2O3@NP catalyst that enables a one-pot synthesis directly from o-nitroaniline compounds and fatty diols without the need for any additional organic solvent. The reaction proceeds efficiently under mild conditions, typically between 70-90°C in an air atmosphere, drastically reducing energy consumption compared to traditional high-temperature methods. By eliminating the solvent and utilizing a solid catalyst that can be easily separated via simple filtration, this method not only simplifies the workflow but also significantly enhances the safety profile of the manufacturing process, positioning it as an ideal solution for reliable pharmaceutical intermediates supplier networks aiming for green chemistry compliance.
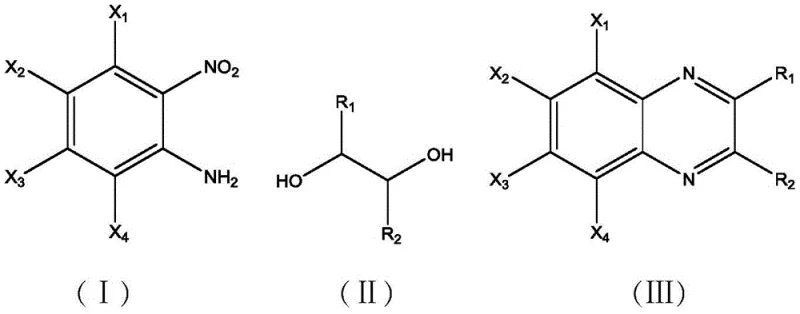
Mechanistic Insights into Cu-Pd-CeO2/γ-Al2O3@NP Catalytic System
The exceptional performance of this catalyst stems from its sophisticated multi-functional design, where each component plays a distinct and synergistic role in the catalytic cycle. The incorporation of Nitrogen (N) and Phosphorus (P) dopants into the γ-Al2O3 support structure is a critical innovation, as it significantly increases the number of basic sites on the catalyst surface. These basic sites are essential for facilitating the initial activation of the reactants and stabilizing intermediate species during the transformation. Simultaneously, the loading of Cerium Oxide (CeO2) serves a dual purpose: it acts as an oxygen storage component and assists in the extraction of protons during the dehydrogenation steps. This proton extraction capability is vital for driving the oxidative dehydrogenation of the diol and the subsequent reduction of the nitro group, ensuring that the reaction proceeds smoothly towards the desired heterocyclic product rather than stalling at partial reduction stages.
Furthermore, the bimetallic combination of Copper (Cu) and Palladium (Pd) creates a highly active surface for hydrogen transfer reactions. The Cu-Pd synergy enhances the overall dehydrogenation activity, which is the rate-limiting step in many similar transformations. This enhanced activity allows the reaction to achieve conversion rates of up to 100% and selectivity as high as 98.3% for the target benzopyrazine products. From an impurity control perspective, the specific pore structure and surface acidity/basicity balance of the @NP modified support help suppress side reactions such as over-reduction or polymerization, which are common pitfalls in nitro-compound chemistry. This precise control over the reaction pathway ensures a cleaner crude product profile, thereby reducing the burden on downstream purification units and contributing to substantial cost savings in the overall manufacturing process.
How to Synthesize Benzopyrazine Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for implementing this technology in a pilot or commercial setting, emphasizing simplicity and reproducibility. The process begins with the meticulous preparation of the catalyst support, followed by the sequential impregnation of active metal species to ensure uniform dispersion. Once the catalyst is activated, the actual synthesis of the benzopyrazine derivative is remarkably straightforward, involving the mixing of the o-nitroaniline substrate, the aliphatic diol, a base promoter, and the catalyst in a standard reactor. The detailed standardized synthesis steps, including specific temperature ramps, stirring rates, and work-up procedures required to achieve the reported high yields, are provided in the technical guide below for immediate reference by process engineers.
- Prepare the CeO2/γ-Al2O3 support by impregnating gamma-alumina with a cerium compound solution, followed by pH adjustment, drying, and calcination at 600-800°C.
- Dope the support with Nitrogen and Phosphorus by treating the carrier with ammonium phosphate solution, drying, and calcining at 400-600°C to form the CeO2/γ-Al2O3@NP carrier.
- Impregnate the N,P-doped carrier with copper and palladium solutions, adjust pH, dry, calcine at 200-300°C, and finally reduce with hydrogen to activate the catalyst.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalytic technology offers transformative benefits that extend far beyond simple yield improvements. The shift from homogeneous to heterogeneous catalysis fundamentally alters the cost structure of benzopyrazine manufacturing by eliminating the need for expensive ligands and complex metal recovery systems. Additionally, the solvent-free nature of the reaction removes the logistical and financial burdens associated with the purchase, storage, and disposal of large volumes of volatile organic solvents. This simplification of the bill of materials directly translates to a more resilient supply chain that is less vulnerable to fluctuations in solvent prices or availability, ensuring consistent production schedules for critical API intermediates.
- Cost Reduction in Manufacturing: The implementation of this solvent-free, heterogeneous catalytic process drives down manufacturing costs through multiple mechanisms. By removing the requirement for organic solvents, facilities can eliminate the capital expenditure and operating costs associated with solvent recovery distillation columns and waste treatment systems. Furthermore, the catalyst's ability to be recycled and reused, as demonstrated in the patent data, reduces the recurring cost of precious metal consumption. The use of inexpensive and readily available raw materials like o-nitroaniline and common diols, instead of costly 1,2-dicarbonyl compounds, further optimizes the raw material spend, resulting in a significantly lower cost of goods sold (COGS) for the final product.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the robustness of the catalyst and the simplicity of the reaction conditions. Operating at moderate temperatures (70-90°C) and under ambient air pressure reduces the stress on reactor equipment, minimizing downtime for maintenance and repairs. The catalyst's stability allows for extended campaign runs without significant loss of activity, ensuring a steady output of product. Moreover, the reliance on commodity chemicals for both the catalyst support (alumina) and the reactants means that sourcing is not dependent on niche suppliers, mitigating the risk of supply disruptions and allowing for more flexible inventory management strategies.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the heterogeneous nature of the catalyst, which is compatible with continuous flow reactors or fixed-bed systems in addition to batch processing. The absence of toxic solvents and the reduction in heavy metal waste align perfectly with increasingly stringent global environmental regulations. This eco-friendly profile not only reduces the regulatory burden and permitting timelines but also enhances the corporate sustainability metrics of the manufacturing entity, making the final product more attractive to environmentally conscious downstream customers in the pharmaceutical and agrochemical sectors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel catalytic system. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on how this technology compares to existing industry standards. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this process into their current manufacturing portfolios to achieve higher efficiency and sustainability goals.
Q: What are the primary advantages of this heterogeneous catalyst over traditional homogeneous methods?
A: Unlike traditional homogeneous catalysts (such as Ru, Ir, or Co complexes) which require complex ligands and difficult separation processes, this Cu-Pd-CeO2/γ-Al2O3@NP catalyst is a solid heterogeneous system. It allows for easy filtration and recycling, eliminates the need for expensive organic solvents, and operates under milder conditions (70-90°C) in air, significantly reducing operational costs and environmental impact.
Q: Can this synthesis method be performed without organic solvents?
A: Yes, the patent explicitly describes a solvent-free one-pot synthesis method. The reaction utilizes the fat diol reactant itself as the medium, which simplifies the downstream processing by removing the need for solvent recovery distillation columns and reduces the generation of hazardous waste streams associated with volatile organic compounds (VOCs).
Q: What is the substrate scope for this benzopyrazine synthesis route?
A: The method demonstrates broad substrate tolerance. It effectively converts various o-nitroaniline derivatives (substituted with methyl, ethyl, or methoxy groups) and aliphatic diols (such as ethylene glycol, 1,2-propanediol, and 2,3-butanediol) into the corresponding benzopyrazine derivatives with high conversion rates (up to 100%) and excellent selectivity (up to 98.3%).
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzopyrazine Supplier
At NINGBO INNO PHARMCHEM, we recognize the immense potential of advanced catalytic technologies like the Cu-Pd-CeO2 system to revolutionize the production of high-value heterocyclic intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are successfully translated into robust industrial realities. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of benzopyrazine or related intermediate we produce adheres to the highest quality standards required by global regulatory bodies.
We invite you to collaborate with us to leverage these cutting-edge synthetic routes for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how switching to this solvent-free methodology can optimize your budget. Please contact us today to request specific COA data for our available intermediates and to discuss route feasibility assessments that can accelerate your time-to-market for new drug candidates.
