Advanced Manganese-Catalyzed Hydrosilylation for Commercial Scale Chiral Silane Production
The landscape of organosilicon chemistry is undergoing a transformative shift driven by the urgent need for sustainable and cost-effective catalytic systems. Patent CN111203276B, published in late 2022, introduces a groundbreaking methodology for the preparation of chiral silanes utilizing a manganese-based catalytic system paired with novel chiral bidentate phosphite ligands. This technology addresses the critical limitations of traditional platinum-group metal catalysts, which are not only prohibitively expensive due to their scarcity but also pose significant toxicity risks in pharmaceutical applications. By leveraging abundant manganese sources, this innovation enables the construction of asymmetric carbon-silicon bonds with exceptional efficiency and optical purity. For global procurement leaders and R&D directors, this represents a pivotal opportunity to secure a reliable chiral silane supplier capable of delivering high-value intermediates while drastically simplifying the supply chain and reducing dependency on critical raw materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of organosilicon materials has relied heavily on platinum-based catalysts, such as the Speier and Karstedt catalysts. While effective, these systems suffer from severe economic and environmental drawbacks that hinder long-term scalability. Platinum is an extremely rare metal with a crustal abundance of merely 0.005ppm, leading to volatile pricing and supply chain vulnerabilities that can disrupt production schedules for fine chemical intermediates. Furthermore, the residual platinum in silicone products is toxic, necessitating rigorous and costly purification steps to meet the stringent safety standards required for medical-grade silicones and active pharmaceutical ingredients (APIs). The inability to efficiently recover these precious metals results in substantial waste and increased operational expenditures, making conventional hydrosilylation processes increasingly unsustainable for large-scale commercial manufacturing in the modern green chemistry era.
The Novel Approach
The patented technology offers a robust solution by replacing precious metals with earth-abundant manganese, combined with specifically designed chiral bidentate phosphite ligands (Formula I or II). This novel catalytic system operates under mild reaction conditions, typically ranging from 10°C to 150°C, and demonstrates remarkable versatility across a wide array of substrates. Unlike traditional methods that struggle with regio- and enantio-selectivity for sp3 center chirality, this manganese-ligand complex facilitates the highly selective formation of chiral silanes with yields reaching up to 98% and enantiomeric excess values as high as 98% ee. 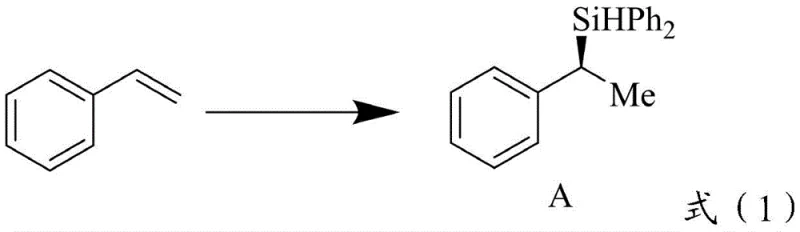 The process is universally applicable to both aliphatic and aromatic olefin derivatives, allowing for the synthesis of complex structures essential for advanced drug discovery without the burden of heavy metal contamination, thereby streamlining the path from laboratory synthesis to commercial scale-up.
The process is universally applicable to both aliphatic and aromatic olefin derivatives, allowing for the synthesis of complex structures essential for advanced drug discovery without the burden of heavy metal contamination, thereby streamlining the path from laboratory synthesis to commercial scale-up.
Mechanistic Insights into Manganese-Catalyzed Asymmetric Hydrosilylation
The core of this technological breakthrough lies in the unique coordination chemistry between the manganese source and the chiral bidentate phosphite ligand. The ligand, characterized by its rigid binaphthyl or cyclohexane backbone substituted with bulky phosphite groups, creates a highly defined chiral pocket around the manganese center. 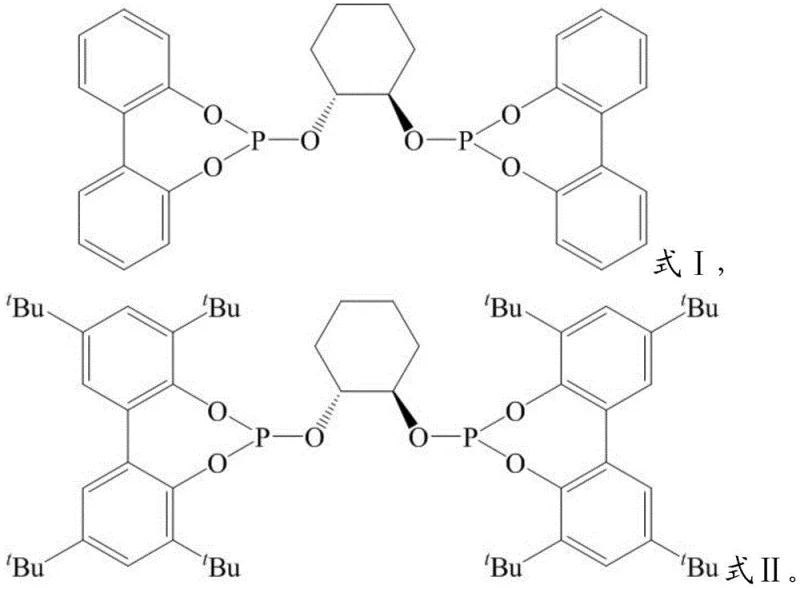 This steric environment is crucial for differentiating the enantiotopic faces of the olefin substrate during the hydrosilylation event. When the manganese salt, such as MnBr2 or MnCl2, complexes with the ligand in a molar ratio ranging from 1:0.4 to 1:1.5, it forms an active catalytic species that activates the Si-H bond of the silane reagent. The subsequent insertion of the olefin into the Mn-H or Mn-Si bond occurs with precise stereocontrol, dictated by the chiral information encoded in the ligand structure, ensuring the formation of the desired enantiomer with minimal formation of the opposite isomer.
This steric environment is crucial for differentiating the enantiotopic faces of the olefin substrate during the hydrosilylation event. When the manganese salt, such as MnBr2 or MnCl2, complexes with the ligand in a molar ratio ranging from 1:0.4 to 1:1.5, it forms an active catalytic species that activates the Si-H bond of the silane reagent. The subsequent insertion of the olefin into the Mn-H or Mn-Si bond occurs with precise stereocontrol, dictated by the chiral information encoded in the ligand structure, ensuring the formation of the desired enantiomer with minimal formation of the opposite isomer.
From an impurity control perspective, this mechanism offers distinct advantages over radical-based or non-chiral transition metal pathways. The concerted nature of the manganese-catalyzed cycle minimizes side reactions such as olefin isomerization or dehydrogenative silylation, which are common pitfalls in less selective systems. The use of protective atmospheres, typically nitrogen or argon, further prevents oxidation of the sensitive manganese-hydride intermediates, ensuring consistent batch-to-batch reproducibility. For R&D teams focusing on purity profiles, this means a cleaner crude reaction mixture that requires less aggressive downstream processing. The ability to tune the electronic properties of the ligand by modifying substituents (such as tert-butyl groups in Formula II) allows for further optimization of reaction rates and selectivity, providing a flexible platform for adapting the synthesis to specific client requirements for high-purity chiral silanes.
How to Synthesize Chiral Silane Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for implementing this technology in a production environment. The process begins with the in situ generation of the catalyst by mixing the chiral ligand and manganese salt in a dry solvent like THF or toluene under inert gas protection. This is followed by the addition of the silane reagent, such as diphenylsilane or phenylsilane, and finally the olefin substrate. The reaction proceeds smoothly within a temperature window of 10°C to 150°C over a period of 1 to 36 hours, depending on the specific substrate reactivity. Detailed standardized synthesis steps see the guide below.
- Prepare the catalyst system by mixing a chiral bidentate phosphite ligand (Formula I or II) with a manganese source (e.g., MnBr2, MnCl2) in a dry Schlenk tube under argon protection.
- Add an organic solvent (such as THF or Toluene) and a silane compound (e.g., diphenylsilane) to the mixture, stirring for approximately 20 minutes to ensure homogeneity.
- Introduce the unsaturated organic compound (e.g., styrene or olefin derivatives) and heat the reaction mixture between 10°C and 150°C for 1 to 36 hours, followed by purification via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this manganese-based hydrosilylation technology translates into tangible strategic benefits that extend beyond simple chemical efficiency. The primary advantage lies in the drastic reduction of raw material costs associated with catalyst procurement. By eliminating the need for platinum, which is subject to geopolitical supply constraints and price volatility, manufacturers can stabilize their cost structures and improve margin predictability. Additionally, the removal of toxic heavy metals from the process simplifies regulatory compliance and waste management protocols. This leads to significant cost reduction in pharmaceutical intermediate manufacturing by avoiding expensive metal scavenging resins and complex purification trains required to lower platinum residues to ppm levels acceptable for human consumption.
- Cost Reduction in Manufacturing: The substitution of scarce platinum with abundant manganese fundamentally alters the economic model of silane production. Manganese salts are orders of magnitude cheaper than platinum complexes, and the ligand loading can be kept low (0.01 to 0.1 molar equivalents relative to substrate) while maintaining high activity. This qualitative shift removes the financial burden of precious metal recovery systems and reduces the capital expenditure required for specialized corrosion-resistant equipment often needed for harsher traditional catalysts. Consequently, the overall cost of goods sold (COGS) for chiral silanes is substantially lowered, enhancing competitiveness in the global market.
- Enhanced Supply Chain Reliability: Relying on earth-abundant elements like manganese mitigates the risk of supply disruptions caused by mining bottlenecks or export restrictions on critical minerals. Manganese is widely available globally, ensuring a stable and continuous supply of catalyst precursors. Furthermore, the robustness of the catalytic system allows for the use of common industrial solvents such as toluene and THF, which are readily accessible in bulk quantities. This reliability ensures that production timelines are met consistently, reducing lead time for high-purity chiral silanes and enabling manufacturers to respond agilely to fluctuating market demands without the fear of catalyst shortages.
- Scalability and Environmental Compliance: The mild reaction conditions and high atom economy of this hydrosilylation process make it inherently scalable from kilogram to multi-ton production scales. The absence of toxic platinum residues simplifies the environmental impact assessment and reduces the load on wastewater treatment facilities. Waste streams are easier to treat and dispose of in accordance with strict environmental regulations, lowering the operational overhead associated with environmental health and safety (EHS) compliance. This sustainability profile aligns perfectly with the growing corporate mandate for green chemistry, positioning suppliers who adopt this technology as preferred partners for environmentally conscious multinational corporations seeking to reduce their carbon footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this manganese-catalyzed hydrosilylation technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for evaluating the feasibility of this route for your specific application needs. Understanding these details is crucial for making informed decisions about integrating this advanced synthetic method into your existing manufacturing workflows.
Q: What are the advantages of using manganese catalysts over platinum for hydrosilylation?
A: Manganese catalysts offer a cost-effective and non-toxic alternative to precious platinum group metals. They utilize abundant earth metals, reducing raw material costs and eliminating the risk of toxic heavy metal residues in final pharmaceutical products, which is critical for regulatory compliance.
Q: What level of enantioselectivity can be achieved with this method?
A: The patented method demonstrates excellent enantioselectivity, achieving up to 98% ee (enantiomeric excess) for various substrates. For instance, the synthesis of (S)-diphenyl(1-phenylethyl)silane reached 97% ee with an 88% yield, ensuring high optical purity for chiral drug intermediates.
Q: Is this catalytic system applicable to diverse olefin substrates?
A: Yes, the system exhibits broad substrate scope. It is effective not only for simple olefins like styrene and n-hexene but also for complex derivatives containing functional groups such as methoxy, fluoro, trifluoromethyl, and sulfonyl groups, making it versatile for synthesizing diverse chiral silane structures.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Silane Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of manganese-catalyzed asymmetric hydrosilylation in the production of next-generation pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to industrial manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including precise measurement of enantiomeric excess and residual metal content, guaranteeing that every batch of chiral silane meets the highest international quality standards required by top-tier drug developers.
We invite you to collaborate with our technical team to explore how this cost-effective and sustainable technology can be tailored to your specific project requirements. By leveraging our expertise in process optimization and scale-up, we can help you achieve significant efficiencies in your supply chain. Please contact our technical procurement team to request a Customized Cost-Saving Analysis, specific COA data for our chiral silane portfolio, and comprehensive route feasibility assessments. Let us partner with you to drive innovation and value in your drug development pipeline through superior chemical manufacturing solutions.
